Advanced Ruthenium-Catalyzed Synthesis of (S)-Duloxetine Key Intermediate for Commercial Scale-Up
Advanced Ruthenium-Catalyzed Synthesis of (S)-Duloxetine Key Intermediate for Commercial Scale-Up
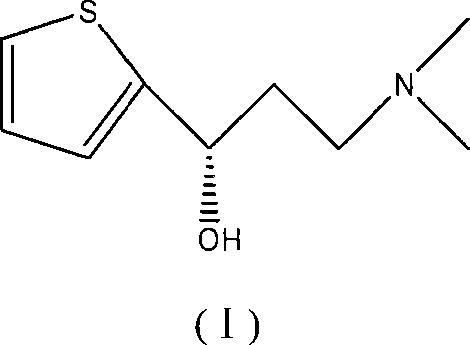
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of high-value active pharmaceutical ingredients (APIs) and their precursors. Patent CN101104614A introduces a transformative approach to synthesizing (S)-N,N-dimethyl-3-hydroxy-3-(2-thienyl)propylamine, a critical chiral intermediate in the manufacturing of Duloxetine hydrochloride, widely known as an antidepressant and analgesic agent. This innovation leverages asymmetric transfer hydrogenation utilizing a chiral monosulfonamide-ruthenium catalyst system, marking a significant departure from conventional resolution techniques. The method utilizes formate salts as a convenient hydrogen source in common organic solvents, eliminating the need for high-pressure hydrogen gas or strictly anaerobic conditions. By achieving high yields and exceptional enantioselectivity under mild conditions, this technology addresses key bottlenecks in the supply chain for nervous system disorder medications. The structural integrity of the target molecule, featuring a chiral hydroxyl group adjacent to a thiophene ring, is paramount for downstream biological activity, and this catalytic route ensures precise stereochemical control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of (S)-duloxetine intermediates has relied heavily on classical resolution strategies or complex hydrogenation protocols that present substantial economic and operational drawbacks. Traditional chemical resolution methods, such as those employing (S)-(+)-mandelic acid, are inherently inefficient because they possess a maximum theoretical yield of only 50%, resulting in the wastage of the unwanted (R)-isomer which is difficult to recycle effectively. Furthermore, alternative catalytic routes utilizing Rhodium-Duanphos complexes, while effective, suffer from extreme sensitivity to oxygen, necessitating rigorous and costly inert gas protection systems throughout the entire manufacturing process. The synthesis of the chiral phosphine ligands required for these rhodium systems is also notoriously difficult and expensive, adding a significant layer of cost to the final API. Additionally, some prior art methods require high hydrogen pressures, such as 7 atmospheres, which imposes stricter safety regulations and requires specialized high-pressure reactor infrastructure that increases capital expenditure for manufacturing facilities.
The Novel Approach
The methodology disclosed in the patent data offers a paradigm shift by employing a chiral monosulfonamide-ruthenium catalyst that operates efficiently under ambient pressure and without the need for nitrogen protection. This novel route utilizes 3-dimethylamino-1-(2-thienyl)-1-propanone hydrochloride as the starting material, subjecting it to asymmetric transfer hydrogenation using formate salts as the hydrogen donor. The reaction proceeds smoothly in solvents like methanol at moderate temperatures ranging from 20°C to 80°C, drastically simplifying the operational complexity compared to high-pressure hydrogenation. The use of a ruthenium-based system with a monosulfonamide diamine ligand provides a robust catalytic cycle that tolerates minor variations in reaction conditions better than sensitive rhodium counterparts. This approach not only improves the atom economy by avoiding the 50% loss inherent in resolution but also streamlines the workflow by removing the requirement for expensive ligand synthesis and strict anaerobic environments, thereby enhancing the overall feasibility for large-scale industrial application.
Mechanistic Insights into Ru-Catalyzed Asymmetric Transfer Hydrogenation
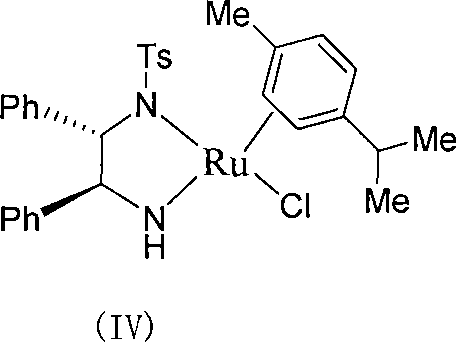
The core of this technological advancement lies in the specific coordination chemistry of the ruthenium catalyst, which facilitates the stereoselective reduction of the prochiral ketone. The catalyst is generated in situ or pre-formed by reacting a ruthenium precursor, such as [RuX2(arene)]2, with a chiral monosulfonamide diamine ligand in the presence of a base like triethylamine. This formation step typically involves refluxing the components in a solvent for 1 to 3 hours to ensure the complete coordination of the ligand to the metal center, creating the active catalytic species depicted in the general formula. The chiral environment created by the sulfonamide and diamine moieties directs the approach of the hydride species derived from the formate hydrogen source, ensuring that the reduction occurs exclusively on one face of the ketone carbonyl group. This precise spatial arrangement is critical for achieving the high enantiomeric excess values observed, often exceeding 90% ee under optimized conditions.
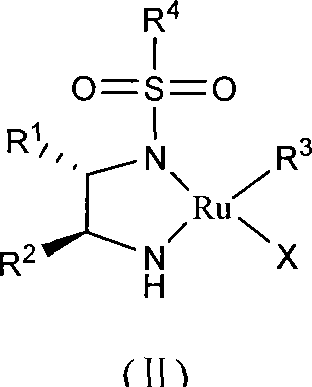
Furthermore, the specific embodiment of the catalyst, such as the complex shown in structure (IV) where the arene ligand is a p-cymene derivative and the diamine backbone features phenyl groups, plays a pivotal role in stabilizing the transition state. The mechanism involves a six-membered pericyclic transition state where the hydride is transferred from the ruthenium center to the carbonyl carbon while a proton is simultaneously transferred from the nitrogen of the ligand to the carbonyl oxygen. This concerted metal-ligand bifunctional catalysis is highly efficient and avoids the formation of free radical intermediates that could lead to racemization or side reactions. The robustness of this mechanism allows the reaction to proceed without strict exclusion of air, as the ruthenium-hydride species formed during the cycle is sufficiently stable to resist rapid oxidation by trace oxygen, unlike many phosphine-based systems. This mechanistic stability translates directly to process reliability, ensuring consistent product quality and minimizing the risk of batch failures due to environmental exposure during scale-up operations.
How to Synthesize (S)-Duloxetine Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for laboratories and manufacturing plants to produce the target chiral amine with high fidelity. The process begins with the preparation or procurement of the specific chiral ruthenium catalyst, followed by the dissolution of the ketone substrate in a polar protic solvent like methanol. The reaction is initiated by the addition of the catalyst and a stoichiometric amount of sodium formate, which serves as the safe and easy-to-handle hydrogen source. The mixture is then maintained at a controlled temperature, preferably between 35°C and 50°C, for a duration of 4 to 8 days to allow the reaction to reach completion with maximal stereocontrol. Detailed standardized synthetic steps, including precise molar ratios, workup procedures involving pH adjustment and extraction, and purification methods, are essential for replicating the high yields reported in the examples.
- Prepare the chiral ruthenium catalyst by refluxing the precursor with chiral monosulfonamide diamine ligands in the presence of triethylamine.
- Dissolve the ketone substrate (3-dimethylamino-1-(2-thienyl)-1-propanone hydrochloride) in a suitable organic solvent such as methanol.
- Add the catalyst and sodium formate hydrogen source, then react at mild temperatures (35-50°C) for several days to achieve high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ruthenium-catalyzed transfer hydrogenation method presents a compelling value proposition driven by significant operational efficiencies and cost structure improvements. The elimination of the resolution step fundamentally alters the cost basis of the intermediate, as the theoretical yield doubles from 50% to nearly 90%, effectively halving the raw material consumption per kilogram of final product. This drastic improvement in atom economy means that less starting material needs to be sourced, stored, and processed, leading to substantial reductions in upstream logistics costs and inventory holding requirements. Moreover, the removal of the need for expensive chiral resolving agents, such as mandelic acid, and the avoidance of complex recycling processes for the unwanted isomer further streamline the bill of materials. The simplicity of the reaction conditions also implies lower utility costs, as there is no need for energy-intensive high-pressure hydrogenation equipment or extensive nitrogen purging systems.
- Cost Reduction in Manufacturing: The transition from resolution-based methods to this asymmetric catalytic route eliminates the inherent 50% yield loss, effectively doubling the output from the same quantity of starting ketone substrate. By utilizing inexpensive formate salts as the hydrogen source instead of high-pressure hydrogen gas or complex silanes, the reagent costs are significantly minimized. The catalyst loading can be kept relatively low while maintaining high turnover, and the absence of expensive rhodium-based ligands reduces the overall catalyst cost burden. These factors combine to create a much leaner cost structure, allowing for more competitive pricing in the bulk drug market while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: The robustness of the ruthenium catalyst system against oxygen exposure removes a critical point of failure in the supply chain, as it negates the need for specialized inert atmosphere handling equipment that can be prone to leaks or maintenance issues. The use of common solvents like methanol and readily available formate salts ensures that raw material sourcing is not dependent on niche suppliers with long lead times. This accessibility of reagents enhances supply continuity, reducing the risk of production stoppages due to material shortages. Furthermore, the mild reaction temperatures reduce thermal stress on equipment, potentially extending the lifespan of reactors and lowering maintenance frequency, which contributes to more predictable production schedules.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the safety hazards associated with high-pressure hydrogen gas, making it easier to obtain regulatory approval for larger reactor volumes. The simplified workup procedure, which involves standard aqueous extraction and crystallization, generates less hazardous waste compared to processes requiring heavy metal scavengers or complex chromatographic separations. This aligns well with modern green chemistry principles and environmental regulations, reducing the cost and complexity of waste treatment and disposal. The ability to run the reaction without strict nitrogen protection also simplifies the engineering controls required for scale-up, facilitating a smoother transition from pilot plant to commercial tonnage production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for duloxetine intermediates. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing manufacturing lines or new project pipelines.
Q: What are the advantages of this Ru-catalyzed method over traditional resolution?
A: Unlike traditional resolution which has a maximum theoretical yield of 50%, this asymmetric transfer hydrogenation method achieves yields up to 88% with high enantiomeric excess (95% ee), effectively doubling the output from the same amount of starting material.
Q: Does this process require strict inert gas protection?
A: No, a significant advantage of this patented method is that it does not require nitrogen protection, simplifying the operational requirements and reducing equipment costs compared to oxygen-sensitive Rh-Duanphos systems.
Q: What represents the optimal reaction conditions for this synthesis?
A: The optimal conditions involve using methanol as the solvent, sodium formate as the hydrogen source, and reacting at 35-50°C for 4-8 days, which balances reaction time with high yield and stereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Duloxetine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and scalable synthetic routes for high-value pharmaceutical intermediates like (S)-N,N-dimethyl-3-hydroxy-3-(2-thienyl)propylamine. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and reliable. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of the manufacturing process. Our capability to implement advanced catalytic technologies, such as the ruthenium-mediated transfer hydrogenation described herein, positions us as a leader in providing cost-effective and high-quality solutions for the global pharmaceutical market.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific volume requirements and quality standards. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalytic route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable manufacturing strategy available.
