Advanced Synthesis of R-3-Chloroserine Methyl Ester Hydrochloride for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antibiotic intermediates that balance high purity with environmental sustainability. A pivotal advancement in this domain is detailed in patent CN109678739B, which discloses a novel synthetic method for R-3-chloroserine methyl ester hydrochloride, a key precursor for the tuberculosis drug D-cycloserine. This technology represents a paradigm shift from legacy methodologies that were plagued by low yields and severe environmental hazards. By leveraging an N-carboxyanhydride (NCA) intermediate strategy, the process achieves exceptional chemical efficiency, reporting final product purities exceeding 99% and yields approaching 95%. For global procurement leaders and R&D directors, this innovation offers a compelling value proposition: a streamlined, cost-effective route that mitigates the supply chain risks associated with complex, multi-step syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cycloserine intermediates has been hindered by reliance on aggressive chlorinating agents and cumbersome purification steps. As illustrated in prior art, such as the route proposed by Plttener P.A. et al., the conventional approach typically involves the direct chlorination of D-serine methyl ester hydrochloride using phosphorus pentachloride (PCl5). This legacy pathway is fundamentally flawed for modern industrial applications due to several critical deficiencies. Firstly, the use of PCl5 generates substantial quantities of phosphorous acid waste, necessitating expensive and energy-intensive neutralization and disposal protocols that inflate operational expenditures. Secondly, the reaction conditions are often harsh, leading to significant racemization and side-product formation, which drastically reduces the overall yield to approximately 40%. Furthermore, the starting material, D-serine methyl ester hydrochloride, is itself a relatively high-cost commodity, compounding the economic inefficiency of the process. These factors collectively create a bottleneck for manufacturers aiming to scale production while adhering to increasingly stringent environmental regulations.
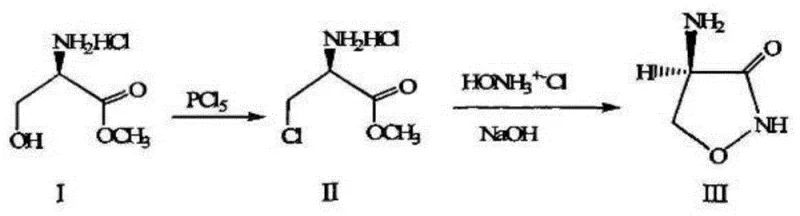
The Novel Approach
In stark contrast to these archaic methods, the technology disclosed in CN109678739B introduces a sophisticated yet operationally simple two-step sequence that circumvents the pitfalls of direct chlorination. The core innovation lies in the initial conversion of D-serine into D-serine N-carboxyanhydride (NCA) via reaction with paraformaldehyde. This activation step fundamentally alters the electronic environment of the molecule, rendering the hydroxyl group significantly more susceptible to nucleophilic substitution in subsequent steps. By bypassing the need for expensive protected serine derivatives and avoiding the use of PCl5, the new route utilizes thionyl chloride (SOCl2) under mild conditions (15-25°C). This strategic modification not only simplifies the reaction setup but also dramatically improves atom economy. The result is a process that is not only chemically superior but also logistically advantageous, as it relies on readily available, commodity-grade reagents that ensure consistent supply chain continuity.
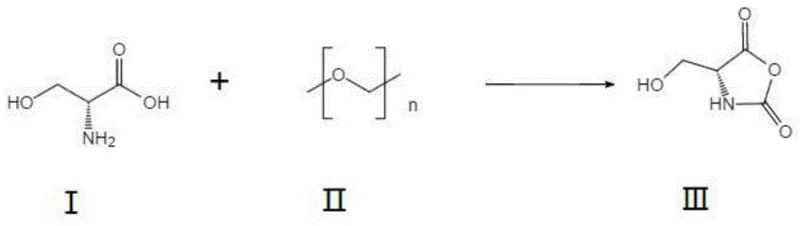
Mechanistic Insights into NCA-Mediated Chlorination
The mechanistic elegance of this synthesis lies in the formation and reactivity of the N-carboxyanhydride intermediate. In the first stage, D-serine reacts with paraformaldehyde to form a cyclic NCA structure. This cyclization protects the amine functionality while simultaneously activating the beta-hydroxyl group. The NCA ring strain and the electron-withdrawing nature of the carbonyl groups facilitate the subsequent nucleophilic attack by chloride ions derived from thionyl chloride. Unlike the traditional route where the free amine might interfere or require protection, the NCA framework directs the reaction specificity towards the desired 3-chloro substitution. Following the chlorination, the reaction medium (methanol) acts as a nucleophile to open the anhydride ring, effectively performing an in-situ esterification to yield the methyl ester. This tandem chlorination-esterification cascade is highly efficient, minimizing the number of unit operations and reducing the potential for product loss during isolation.
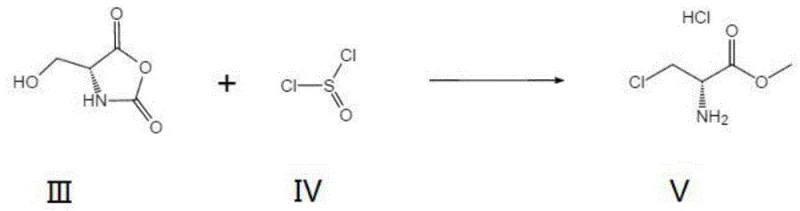
From an impurity control perspective, this mechanism offers distinct advantages. The mild reaction temperatures (15-25°C) suppress thermal degradation pathways that often lead to polymeric byproducts or racemization in harsher acidic environments. Moreover, the crystallization strategy employed in the final step—using petroleum ether as an anti-solvent—is highly selective for the target hydrochloride salt. This allows for the effective rejection of unreacted starting materials and minor side products, ensuring that the final API intermediate meets rigorous purity specifications without the need for chromatographic purification. For quality assurance teams, this implies a more robust and predictable impurity profile, simplifying regulatory filings and batch release testing.
How to Synthesize R-3-Chloroserine Methyl Ester Hydrochloride Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction parameters to maximize the reported yields of >94%. The process begins with the preparation of the NCA intermediate in anhydrous tetrahydrofuran, followed by the critical chlorination step in methanol. Operators must maintain strict temperature control during the addition of thionyl chloride to prevent exothermic runaway, although the reaction is generally manageable at ambient conditions. The final isolation via petroleum ether crystallization is a key determinant of product quality. For a detailed breakdown of the specific molar ratios, reaction times, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- React D-serine with paraformaldehyde in anhydrous tetrahydrofuran at room temperature to form D-serine N-carboxyanhydride (NCA).
- Treat the isolated D-serine NCA with thionyl chloride in methanol at 15-25°C to effect simultaneous chlorination and esterification.
- Recover the solvent and induce crystallization using petroleum ether to obtain the final product with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthetic route translates into tangible operational efficiencies and risk mitigation. The elimination of phosphorus pentachloride removes a major hazardous material from the supply chain, thereby reducing the costs associated with hazardous waste disposal and regulatory compliance. Furthermore, the switch to commodity reagents like paraformaldehyde and thionyl chloride ensures that raw material sourcing is stable and less susceptible to market volatility compared to specialized protected amino acid derivatives.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic increase in chemical yield. By improving the yield from the historical baseline of roughly 40% to over 94%, the effective cost per kilogram of the active intermediate is more than halved, excluding raw material savings. Additionally, the simplified workup procedure, which relies on solvent recovery and crystallization rather than complex extractions or column chromatography, significantly lowers utility consumption and labor hours per batch. The avoidance of expensive chiral resolving agents, as the process starts directly from inexpensive D-serine, further contributes to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The reliance on D-serine, a fermentation-derived bulk chemical, ensures a secure and scalable raw material base. Unlike routes dependent on custom-synthesized precursors with long lead times, D-serine is widely available from multiple global suppliers. This diversification of the supply base reduces the risk of production stoppages due to single-source vendor failures. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, enhancing the consistency of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is markedly superior. The replacement of PCl5 with thionyl chloride simplifies off-gas treatment, as SO2 and HCl are easier to scrub and neutralize than phosphorous waste. The process operates at near-ambient temperatures, reducing the energy load required for heating or cryogenic cooling. These factors make the technology highly amenable to scale-up from pilot plant to multi-ton commercial production, allowing manufacturers to meet growing global demand for cycloserine without incurring prohibitive capital expenditure on waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the feasibility and benefits for potential licensees or manufacturing partners.
Q: Why is the NCA route superior to the traditional PCl5 method?
A: The traditional method utilizes phosphorus pentachloride (PCl5), which generates hazardous acidic waste and requires complex neutralization. The patented NCA route uses thionyl chloride under mild conditions, significantly reducing environmental impact and simplifying waste treatment while boosting yield from ~40% to over 94%.
Q: What is the expected purity and yield of this synthesis?
A: According to patent CN109678739B, the optimized process achieves a final product purity of up to 99.5% and a chemical yield ranging from 91.93% to 94.72%, depending on specific molar ratios and reaction times.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids expensive raw materials and difficult-to-handle reagents, utilizing common solvents like methanol and tetrahydrofuran. The simple crystallization workup facilitates easy isolation, making it highly viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-3-Chloroserine Methyl Ester Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antibiotics like D-cycloserine. Our technical team has thoroughly analyzed the pathway described in CN109678739B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient process to market. We are equipped with rigorous QC labs and state-of-the-art reactors capable of handling the specific solvent systems and exothermic profiles of this NCA-mediated synthesis, ensuring that every batch meets stringent purity specifications of >99%.
We invite pharmaceutical manufacturers and procurement directors to collaborate with us to leverage this advanced technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to obtain specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the next generation of antibiotic production.
