Advanced Enzymatic Production of Alpha-Ketoglutarate Calcium for Global Supply Chains
Advanced Enzymatic Production of Alpha-Ketoglutarate Calcium for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and safe manufacturing routes for critical metabolic intermediates. A significant breakthrough in this domain is detailed in patent CN112941116A, which discloses a highly efficient method for preparing alpha-ketoglutarate calcium via an enzymatic pathway. This technology represents a paradigm shift from traditional chemical synthesis, leveraging the specificity of L-glutamate oxidase to convert abundant L-glutamic acid directly into the target calcium salt. For R&D directors and procurement strategists, this patent offers a compelling value proposition: it eliminates the use of genotoxic starting materials, simplifies downstream processing by avoiding the isolation of unstable free acids, and ensures a product profile suitable for high-value applications in nutrition and medicine. The structural relationship between the precursor and the final calcium salt is fundamental to understanding the efficiency gains of this route.
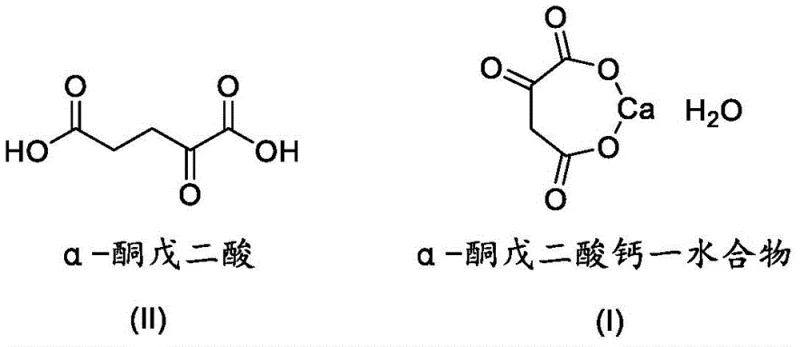
As a reliable pharmaceutical intermediates supplier, understanding the nuances of such proprietary technologies is essential for maintaining a competitive edge. The transition from petrochemical-dependent synthesis to bio-based manufacturing is not merely a trend but a necessity for meeting increasingly rigorous global regulatory standards regarding impurity profiles and environmental impact. This report analyzes the technical merits of patent CN112941116A, providing deep insights into how this enzymatic strategy facilitates cost reduction in pharmaceutical manufacturing while securing a more resilient supply chain for high-purity alpha-ketoglutarate derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of alpha-ketoglutarate calcium has relied heavily on purely chemical synthetic routes or less optimized biological conversions that introduce significant operational bottlenecks. The traditional chemical synthesis, as illustrated in prior art, typically involves the condensation of methyl acrylate with methyl dichloroacetate in the presence of strong bases like sodium methoxide. While this method can achieve high theoretical yields, it suffers from severe drawbacks regarding product safety and environmental compliance. The starting materials, particularly methyl dichloroacetate, are flagged for potential genotoxicity, posing a critical risk for any application intended for human consumption, such as dietary supplements or parenteral nutrition. Furthermore, the chemical route generates substantial halogenated waste streams, complicating wastewater treatment and increasing the overall environmental footprint of the manufacturing facility.
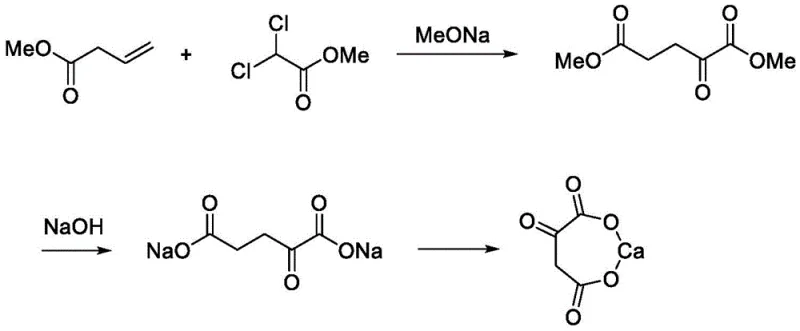
Alternatively, earlier enzymatic methods attempted to produce the free alpha-ketoglutaric acid first, followed by a separate salification step. This two-stage approach is inherently inefficient because alpha-ketoglutaric acid is chemically unstable, especially under the alkaline conditions often required for subsequent salt formation. This instability leads to decarboxylation and the formation of stubborn impurities like succinic acid, which are difficult to remove and degrade the quality of the final API or ingredient. Consequently, these conventional processes require complex purification steps, such as repeated crystallizations or chromatography, which drastically reduce overall recovery rates and inflate production costs, making them less attractive for large-scale commercialization.
The Novel Approach
The methodology described in patent CN112941116A elegantly circumvents these historical challenges by integrating the enzymatic oxidation and salification into a seamless, continuous workflow. Instead of isolating the unstable free acid, the process utilizes L-glutamic acid or its sodium salts as a safe, renewable feedstock. Through the action of L-glutamate oxidase and catalase, the substrate is selectively oxidized to form the alpha-ketoglutarate intermediate in situ. Crucially, the patent teaches that this intermediate solution can be directly treated with calcium chloride to precipitate the target calcium salt. This "one-pot" style progression minimizes the time the sensitive keto-acid spends in solution, thereby suppressing degradation pathways. The result is a process that not only guarantees a non-genotoxic impurity profile but also streamlines the operation, reducing unit operations and energy consumption significantly.
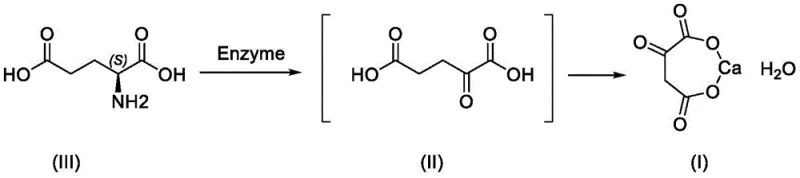
For supply chain heads focused on the commercial scale-up of complex pharmaceutical intermediates, this novel approach offers distinct advantages in terms of process robustness. By operating at mild temperatures ranging from 20°C to 40°C and utilizing air or oxygen as the terminal oxidant, the reaction conditions are far safer and easier to control than the harsh exothermic reactions seen in chemical synthesis. The direct salification step eliminates the need for pH adjustment using carbonates, which can introduce insoluble carbonate impurities, thus ensuring a higher purity product right out of the reactor. This technological leap positions the enzymatic route as the superior choice for manufacturers aiming to reduce lead time for high-purity alpha-ketoglutarate derivatives while adhering to green chemistry principles.
Mechanistic Insights into L-Glutamate Oxidase Catalyzed Oxidation
The core of this innovative synthesis lies in the precise biocatalytic mechanism driven by L-glutamate oxidase. This enzyme facilitates the oxidative deamination of L-glutamic acid, a reaction that involves the removal of the alpha-amino group and the simultaneous oxidation of the alpha-carbon to a ketone group. In the presence of molecular oxygen, the enzyme converts the amino acid substrate into alpha-ketoglutaric acid, ammonia, and hydrogen peroxide. The inclusion of catalase in the reaction system is a critical mechanistic detail; it rapidly decomposes the generated hydrogen peroxide into water and oxygen. This dual-enzyme system prevents the accumulation of peroxide, which could otherwise oxidize sensitive functional groups or inactivate the primary oxidase, thereby maintaining high catalytic turnover and reaction stability over extended periods.
From an impurity control perspective, the specificity of the enzyme is paramount. Unlike chemical oxidants which may attack various sites on the carbon chain, L-glutamate oxidase exhibits high regioselectivity for the alpha-position of the glutamate backbone. This selectivity effectively shuts down the formation of random oxidation byproducts that are common in non-enzymatic routes. Furthermore, by avoiding the isolation of the free acid, the process mitigates the risk of non-enzymatic decarboxylation, a common degradation pathway for alpha-keto acids that leads to succinic acid formation. The mechanistic elegance of coupling the enzymatic step directly with calcium precipitation ensures that the product is locked into its stable salt form immediately upon formation, preserving the integrity of the molecule and simplifying the downstream purification to a mere filtration and washing step.
How to Synthesize Alpha-Ketoglutarate Calcium Efficiently
Implementing this enzymatic protocol requires careful attention to reaction parameters to maximize the conversion efficiency described in the patent examples. The process begins with the preparation of an aqueous solution of the starting material, preferably L-glutamic acid monosodium salt monohydrate, which offers excellent solubility and pH buffering capacity. The reaction environment must be strictly controlled to maintain a neutral pH around 7.0, as deviations can affect enzyme activity and product stability. Oxygen mass transfer is another critical factor; efficient aeration or oxygen sparging is necessary to sustain the oxidative cycle of the enzyme. Once the conversion reaches completion, indicated by HPLC analysis showing minimal residual substrate, the addition of stoichiometric calcium chloride triggers the crystallization of the product. The detailed standardized synthesis steps for replicating this high-yield process are outlined below.
- Prepare an aqueous solution of L-glutamic acid or its sodium salt and adjust pH to neutral conditions suitable for enzymatic activity.
- Introduce L-glutamate oxidase and catalase into the reactor while maintaining temperature between 20-40°C and supplying oxygen or air for oxidative deamination.
- Upon completion of conversion, directly add calcium chloride solution to the reaction mixture to precipitate alpha-ketoglutarate calcium without isolating the free acid intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN112941116A translates into tangible strategic benefits that extend beyond simple unit cost calculations. The shift from petrochemical precursors to fermentation-derived amino acids fundamentally de-risks the supply chain. L-glutamic acid is a commodity chemical produced on a massive global scale for the food industry, ensuring a stable, abundant, and price-resilient supply base. This contrasts sharply with specialized halogenated reagents used in chemical synthesis, which are subject to volatile pricing and stricter regulatory controls due to their hazardous nature. By anchoring production to a widely available biological feedstock, manufacturers can secure long-term supply continuity and mitigate the risks associated with raw material shortages.
- Cost Reduction in Manufacturing: The economic model of this enzymatic process is strengthened by the elimination of costly downstream purification steps. Traditional methods often require extensive solvent extraction, distillation, or chromatographic separation to remove genotoxic impurities and degradation byproducts like succinic acid. By achieving high selectivity and direct salification, this new method drastically simplifies the workflow, reducing solvent consumption, energy usage for heating and cooling, and labor hours. The avoidance of expensive hazard mitigation measures required for handling genotoxic substances further lowers the operational expenditure, resulting in substantial cost savings that can be passed down the value chain.
- Enhanced Supply Chain Reliability: The reliance on L-glutamic acid as a starting material provides a robust foundation for supply chain reliability. As a fermentation product, its production is scalable and less susceptible to the geopolitical and logistical disruptions that often affect petrochemical supply lines. Additionally, the mild reaction conditions (ambient pressure and moderate temperatures) reduce the dependency on specialized high-pressure or cryogenic equipment, allowing for more flexible manufacturing across different facilities. This flexibility ensures that production can be maintained consistently, meeting the rigorous delivery schedules demanded by global pharmaceutical and nutraceutical clients without interruption.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers a clear path to sustainable manufacturing. The absence of halogenated waste streams and genotoxic reagents simplifies waste treatment and disposal, lowering the environmental compliance burden. The use of oxygen from air as the oxidant generates water as the primary byproduct, aligning with green chemistry principles. This eco-friendly profile not only reduces the carbon footprint of the manufacturing process but also enhances the marketability of the final product to environmentally conscious consumers and regulators, facilitating smoother regulatory approvals in key markets like the EU and North America.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the enzymatic production of alpha-ketoglutarate calcium. These answers are derived directly from the experimental data and technical disclosures found within the patent literature, providing a factual basis for evaluating the feasibility of this technology. Understanding these specifics is crucial for technical teams assessing the integration of this route into existing production portfolios.
Q: Why is the enzymatic method preferred over chemical synthesis for food-grade alpha-ketoglutarate calcium?
A: Chemical synthesis often relies on raw materials like methyl acrylate and methyl dichloroacetate, which carry potential genotoxicity risks. The enzymatic method utilizes L-glutamic acid, a safe, natural amino acid, ensuring the final product meets stringent food-grade and pharmaceutical safety standards without toxic residual impurities.
Q: How does the direct salification process improve yield compared to traditional enzyme methods?
A: Traditional enzyme methods isolate the unstable free alpha-ketoglutaric acid, which can decompose into succinic acid under alkaline conditions. By skipping the isolation step and directly reacting the intermediate solution with calcium chloride, this patent prevents decomposition, significantly enhancing overall yield and product purity.
Q: What are the scalability advantages of using L-glutamate oxidase in this process?
A: The process operates under mild conditions (20-40°C) and uses air or oxygen as the oxidant, eliminating the need for hazardous chemical oxidants. This simplifies reactor design and safety protocols, making commercial scale-up from pilot to multi-ton production safer and more energy-efficient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Ketoglutarate Calcium Supplier
The technological advancements detailed in patent CN112941116A underscore the immense potential of biocatalysis in modernizing the production of key metabolic intermediates. At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting such innovative pathways to meet the evolving demands of the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are successfully translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to deliver alpha-ketoglutarate calcium with stringent purity specifications that exceed international pharmacopoeia standards.
We invite procurement leaders and R&D innovators to collaborate with us to leverage this superior enzymatic technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on the most efficient, safe, and cost-effective foundation available in the industry.
