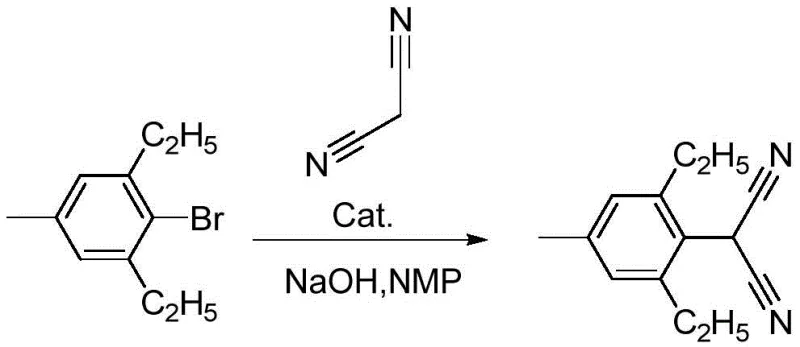
Scalable Nickel-Catalyzed Synthesis of Pinoxaden Intermediate for Global Herbicide Markets
The global demand for high-efficiency graminicides continues to drive innovation in the synthesis of key agrochemical intermediates, specifically those required for the production of Pinoxaden. A pivotal advancement in this domain is detailed in patent CN109320435B, which discloses a novel synthetic methodology for 2-(2,6-diethyl-4-methylphenyl)malononitrile. This compound serves as a critical building block in the manufacturing of phenylpyrazoline herbicides, acting as an acetyl-CoA carboxylase (ACC) inhibitor that effectively disrupts fatty acid synthesis in target weeds. The technical breakthrough presented in this intellectual property lies in the strategic substitution of traditional noble metal catalysts with a more economically viable nickel-based system. By leveraging bis(triphenylphosphine)nickel dibromide under alkaline conditions, the disclosed process achieves yields comparable to palladium-catalyzed routes while significantly mitigating the financial burden associated with precious metal procurement. For R&D directors and procurement strategists alike, this represents a tangible opportunity to optimize the cost structure of herbicide supply chains without compromising on the purity or efficacy of the final active ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(2,6-diethyl-4-methylphenyl)malononitrile has relied heavily on palladium-catalyzed cross-coupling reactions, typically employing triphenylphosphine palladium chloride in xylenes with sodium tert-butoxide as the base. While these legacy methods can achieve respectable yields approaching 92.6%, they are plagued by severe economic and operational inefficiencies that hinder large-scale commercial viability. The primary bottleneck is the exorbitant cost of palladium salts, which can range from 3.5 to 5 million currency units per ton, creating a volatile cost baseline that is difficult to hedge against market fluctuations. Furthermore, conventional protocols often require high catalyst loadings, sometimes up to 16 wt% relative to the substrate, which not only inflates the bill of materials but also complicates downstream purification. The removal of residual palladium from the final product is a notoriously difficult and expensive step, often requiring specialized scavengers or multiple recrystallization cycles to meet stringent regulatory limits for heavy metals in agrochemical products. Additionally, the recovery and reuse of palladium catalysts are technically challenging, leading to significant waste generation and environmental compliance issues that modern green chemistry initiatives seek to eliminate.
The Novel Approach
In stark contrast to the palladium-dependent status quo, the innovative route described in the patent data utilizes a nickel-based catalytic system that fundamentally alters the economic equation of intermediate manufacturing. By employing bis(triphenylphosphine)nickel dibromide, the process capitalizes on the similar electronic properties of nickel and palladium while benefiting from nickel's substantially lower market price and greater abundance. This novel approach allows for a drastic reduction in catalyst loading, with optimal results achieved at ratios as low as 1:0.03 relative to the aryl halide, thereby minimizing the metal burden in the reaction mixture. The use of N-methylpyrrolidone (NMP) as the preferred solvent enhances the solubility of reactants, facilitating rapid mass transfer and enabling the reaction to proceed efficiently at moderate temperatures between 110°C and 120°C. Crucially, this method maintains a high yield of approximately 90%, demonstrating that the transition to base metals does not necessitate a sacrifice in productivity. The simplified workup procedure, involving straightforward acidification and extraction, further underscores the operational superiority of this nickel-catalyzed pathway over its noble metal predecessors.
Mechanistic Insights into Nickel-Catalyzed Cyanomethylation
The mechanistic underpinning of this synthesis involves a nickel-catalyzed cross-coupling reaction, likely proceeding through a cycle of oxidative addition, transmetallation, and reductive elimination, analogous to palladium chemistry but with distinct kinetic profiles. In the presence of a strong base such as sodium hydroxide or potassium hydroxide, the acidic protons of malononitrile are deprotonated to form a nucleophilic carbanion species. This activated nitrile then interacts with the nickel center, which has previously undergone oxidative addition with the 4-methyl-2,6-diethylbromobenzene substrate. The steric environment provided by the triphenylphosphine ligands is critical in stabilizing the nickel intermediates and preventing the formation of inactive nickel black precipitates, a common failure mode in nickel catalysis. The choice of base plays a pivotal role in this mechanism; experimental data indicates that sodium hydroxide and potassium hydroxide provide superior yields compared to weaker or bulkier bases like cesium fluoride or sodium hydride, suggesting that the rate-determining step may involve the efficient generation of the nucleophile or the facilitation of the transmetallation step. Understanding these subtle electronic and steric interactions allows process chemists to fine-tune reaction conditions, ensuring that the catalytic turnover number remains high throughout the 12-hour reaction window.
Impurity control is another critical aspect of the mechanistic landscape, particularly concerning the formation of homocoupling byproducts or hydrolysis of the nitrile groups. The patent data highlights that maintaining a nitrogen atmosphere is essential to prevent oxidation of the nickel catalyst and the sensitive malononitrile anion. Furthermore, the precise control of reaction temperature between 110°C and 120°C is vital; temperatures that are too low result in incomplete conversion, while excessive heat may promote side reactions or decomposition of the catalyst ligand system. The post-reaction treatment involves adjusting the pH to 2-3 with hydrochloric acid, which serves to protonate any remaining basic species and facilitates the partitioning of the organic product into the ethyl acetate phase. This acidic quench also helps to decompose any unstable nickel complexes, ensuring that the final crude product is free from metal contaminants that could interfere with subsequent synthetic steps in the Pinoxaden value chain. The final recrystallization from methanol at 0-5°C effectively removes trace organic impurities, delivering a product with a purity content exceeding 95%, which is suitable for direct use in downstream herbicide synthesis.
How to Synthesize 2-(2,6-Diethyl-4-methylphenyl)malononitrile Efficiently
Implementing this synthesis route requires careful attention to the order of addition and the maintenance of an inert atmosphere to ensure reproducibility and safety. The process begins with the preparation of the nucleophile by mixing malononitrile with the base in the polar aprotic solvent, followed by the introduction of the electrophile and the catalyst. Detailed standard operating procedures regarding specific stoichiometric ratios, heating ramps, and workup parameters are essential for transferring this technology from the laboratory to the pilot plant. For a comprehensive, step-by-step guide on executing this reaction safely and effectively, please refer to the standardized protocol outlined below.
- Prepare the reaction mixture by combining malononitrile, sodium hydroxide, and N-methylpyrrolidone (NMP) under nitrogen protection, maintaining temperature at 25-35°C for 1 hour.
- Introduce 4-methyl-2,6-diethylbromobenzene and the nickel catalyst to the mixture, then heat the system to 110-120°C and react for 12 hours until conversion is complete.
- Quench the reaction with water, adjust pH to 2-3 using hydrochloric acid, extract with ethyl acetate, and recrystallize the crude product from methanol to obtain high-purity material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed synthesis offers a compelling value proposition centered on cost stability and operational resilience. The shift away from palladium eliminates exposure to the volatile pricing of precious metals, allowing for more accurate long-term budgeting and cost forecasting. Moreover, the reduced catalyst loading translates directly into lower raw material consumption per kilogram of product, enhancing the overall atom economy of the process. The use of commodity chemicals like sodium hydroxide and NMP, rather than specialized bases like sodium tert-butoxide, further simplifies the supply chain and reduces the risk of logistical bottlenecks. From a manufacturing perspective, the robustness of the reaction conditions means that the process can be scaled up with minimal re-engineering, reducing the time-to-market for new batches of herbicide intermediates.
- Cost Reduction in Manufacturing: The replacement of high-cost palladium salts with nickel-based catalysts results in a substantial decrease in the direct material cost of the intermediate. Since the catalyst loading is significantly lower and the metal itself is orders of magnitude cheaper, the overall cost of goods sold (COGS) is optimized. Additionally, the simplified purification process reduces the consumption of solvents and energy associated with extensive metal scavenging steps, contributing to further indirect cost savings in the utility and waste management budgets.
- Enhanced Supply Chain Reliability: Relying on nickel instead of palladium diversifies the supplier base, as nickel compounds are more widely available and less subject to geopolitical supply constraints than platinum group metals. The use of common reagents like sodium hydroxide and ethyl acetate ensures that the supply chain is resilient to disruptions, as these materials are standard inventory items for most chemical manufacturing facilities. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major agrochemical companies.
- Scalability and Environmental Compliance: The process generates less hazardous waste due to the absence of heavy palladium residues, simplifying effluent treatment and reducing the environmental footprint of the manufacturing site. The reaction operates at atmospheric pressure and moderate temperatures, eliminating the need for expensive high-pressure reactors or cryogenic cooling systems, which lowers the capital expenditure required for scale-up. These factors combined make the technology highly attractive for contract development and manufacturing organizations (CDMOs) looking to offer sustainable and scalable solutions to their clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is key to evaluating the feasibility of integrating this route into your existing manufacturing portfolio.
Q: Why is the nickel-catalyzed route preferred over palladium for this intermediate?
A: The nickel-catalyzed method described in patent CN109320435B replaces expensive palladium salts with bis(triphenylphosphine)nickel dibromide. This substitution drastically reduces raw material costs while maintaining high yields (approx. 90%) and simplifies catalyst recovery, addressing key economic bottlenecks in large-scale herbicide production.
Q: What are the critical process parameters for maximizing yield?
A: Optimal yields are achieved using N-methylpyrrolidone (NMP) as the solvent due to its superior solubility profile. The molar ratio of substrate to base (NaOH) should be maintained around 1:3, and the catalyst loading is optimized at approximately 3-8% relative to the aryl halide to balance cost and efficiency.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the process utilizes robust conditions (110-120°C) and common reagents like sodium hydroxide and ethyl acetate. The workup involves standard acidification and extraction steps, making it highly amenable to commercial scale-up without requiring specialized high-pressure equipment or cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2,6-Diethyl-4-methylphenyl)malononitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the competitive agrochemical landscape. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies like the nickel-coupled route described above, we help our partners achieve superior cost structures without compromising on quality or regulatory compliance.
We invite you to collaborate with us to optimize your supply chain for Pinoxaden and related herbicides. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
