Advanced Hydrogen Peroxide Oxidation Route for High-Purity Cyclotriphosphazene Derivatives
The global demand for high-performance, halogen-free flame retardants has necessitated a rigorous re-evaluation of synthetic pathways for key phosphazene intermediates. Patent CN103467525A introduces a transformative methodology for preparing hexa(4-carboxy-phenoxy)-cyclotriphosphazene (HCPCP), a critical precursor for advanced epoxy resin modifiers and engineering plastics. Unlike conventional approaches that rely on hazardous reagents or generate substantial toxic waste, this invention utilizes a mild, two-step sequence anchored by hydrogen peroxide oxidation. For R&D directors and procurement specialists seeking a reliable plastic additives supplier, this technology represents a paradigm shift towards greener, more economically viable manufacturing. The process replaces dangerous sodium hydride bases with stable potassium salts and substitutes heavy metal oxidants with environmentally benign hydrogen peroxide, directly addressing the industry's pressing need for sustainable cost reduction in polymer additive manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hexa(4-formyl-phenoxy)-cyclotriphosphazene (HAPCP) and its subsequent oxidation to HCPCP has been plagued by significant operational and safety hurdles. Traditional literature, such as reports by Bing Baichun et al., frequently employs sodium hydride (NaH) as the acid-binding agent for the initial nucleophilic substitution. While chemically effective, NaH is pyrophoric, requiring stringent inert atmosphere controls and specialized handling equipment that inflate capital expenditure. Furthermore, the oxidation step in prior art typically relies on potassium permanganate (KMnO4). Although a strong oxidant, KMnO4 generates massive quantities of manganese dioxide (MnO2) sludge, creating a nightmare for downstream processing. The removal of this inorganic residue requires extensive filtration and washing, leading to product entrapment losses and prolonged batch cycles. Additionally, the reaction conditions for KMnO4 oxidation often require elevated temperatures around 60-67°C for extended periods of 24-48 hours, resulting in energy inefficiency and potential thermal degradation of the sensitive phosphazene ring structure.
The Novel Approach
The patented methodology fundamentally重构 s the synthetic landscape by introducing a safer, more efficient protocol that eliminates these bottlenecks. In the first step, the process utilizes anhydrous potassium carbonate (K2CO3) or potassium phosphate as the acid scavenger in anhydrous tetrahydrofuran (THF). This substitution removes the fire hazard associated with NaH, allowing for simpler reactor configurations and reduced safety overheads. The reaction proceeds smoothly at moderate temperatures of 50-80°C over 10-30 hours, yielding the HAPCP intermediate with high consistency. Crucially, the second oxidation step employs 30% hydrogen peroxide in an alkaline medium (pH 9-11), facilitated by a phase transfer catalyst, tetrabutylammonium bromide (TBAB). This system operates effectively at mild temperatures ranging from 15-60°C. The byproduct of this oxidation is merely water and oxygen gas, completely eradicating the heavy metal waste stream. This clean reaction profile not only simplifies the workup to basic layering and washing but also ensures that the final high-purity flame retardant intermediates are free from trace metal contaminants that could compromise the optical or electrical properties of the final polymer application.
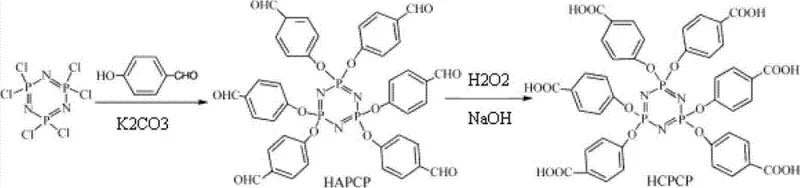
Mechanistic Insights into Hydrogen Peroxide Oxidation with Phase Transfer Catalysis
The core innovation of this technology lies in the sophisticated manipulation of phase transfer catalysis (PTC) during the oxidation phase. In the absence of a catalyst, the reaction between the organic-soluble HAPCP and the aqueous hydrogen peroxide would be kinetically limited by the interface between the two immiscible phases. The addition of TBAB serves as a molecular shuttle, transporting the hydroperoxide anion (HOO-) from the alkaline aqueous phase into the organic THF phase where the HAPCP substrate resides. This dramatically increases the local concentration of the active oxidizing species around the aldehyde groups, accelerating the reaction rate significantly. The mechanism involves the nucleophilic attack of the hydroperoxide anion on the carbonyl carbon of the aldehyde, forming a peroxyhemiacetal intermediate which subsequently rearranges to the carboxylic acid. This pathway is highly selective for aldehydes and leaves the robust phosphazene ring intact, ensuring structural integrity throughout the transformation.
Furthermore, the process exploits a clever solubility switch for purification, a critical factor for achieving the stringent quality standards required in commercial scale-up of complex phosphazene derivatives. The intermediate HAPCP is insoluble in alkaline aqueous solutions, whereas the final product, HCPCP, containing six carboxylic acid groups, forms water-soluble carboxylate salts under the same conditions (pH 9-11). After the oxidation is complete, the reaction mixture naturally separates into phases where unreacted starting material can be filtered off or separated, while the product remains in the aqueous layer. Acidification of this aqueous layer then precipitates the pure HCPCP as a white solid. This intrinsic purification mechanism minimizes the need for column chromatography or extensive recrystallization cycles, directly contributing to reducing lead time for high-purity flame retardants and maximizing overall mass balance efficiency.
How to Synthesize Hexa(4-carboxy-phenoxy)-cyclotriphosphazene Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and phase conditions to replicate the high yields reported in the patent data. The process begins with the careful preparation of the nucleophilic substitution mixture, ensuring that the molar ratio of 4-hydroxybenzaldehyde to the acid binder and HCCP is maintained between (5-10):(5-8):1 to drive the reaction to completion. Following the isolation of the HAPCP intermediate, the oxidation step demands strict pH control to activate the hydrogen peroxide without degrading the catalyst. The detailed standardized operating procedures, including specific addition rates and temperature ramps optimized for industrial reactors, are outlined below to ensure reproducibility and safety.
- Perform nucleophilic substitution of hexachlorocyclotriphosphazene (HCCP) with 4-hydroxybenzaldehyde using anhydrous potassium carbonate in THF at 50-80°C to obtain HAPCP.
- Purify the intermediate HAPCP via filtration, vacuum distillation, and recrystallization using ethanol or ethyl acetate to achieve 98%-99% purity.
- Oxidize the purified HAPCP using 30% hydrogen peroxide and sodium hydroxide (pH 9-11) with TBAB as a phase transfer catalyst at 15-60°C to produce HCPCP.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this hydrogen peroxide-based methodology offers profound strategic benefits beyond simple chemistry. The elimination of hazardous reagents like sodium hydride and potassium permanganate translates directly into reduced regulatory burden and lower insurance premiums. By removing the generation of heavy metal sludge, the facility avoids the high costs associated with hazardous waste disposal and environmental compliance reporting. This streamlined waste profile allows for faster batch turnover and reduces the risk of production stoppages due to waste tank capacity limits. Moreover, the use of commodity chemicals like potassium carbonate and hydrogen peroxide ensures a stable, multi-vendor supply chain, mitigating the risk of raw material shortages that often plague specialty reagent markets.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling when analyzing the total cost of ownership. By replacing expensive and dangerous sodium hydride with inexpensive potassium carbonate, the raw material cost for the acid-binding step is drastically lowered. More significantly, the simplified workup procedure—eliminating the filtration of manganese dioxide sludge—reduces labor hours and solvent consumption. The high selectivity of the hydrogen peroxide oxidation minimizes side reactions, leading to higher crude yields that require less remediation. These factors combine to create a leaner manufacturing process with substantially improved gross margins, making the final product more competitive in the global market without sacrificing quality.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for downstream polymer manufacturers who cannot afford interruptions in their additive supply. This method relies on robust, shelf-stable reagents that are readily available in bulk quantities globally. Unlike sodium hydride, which requires special shipping classifications and has a limited shelf life due to moisture sensitivity, potassium carbonate is stable and easy to store. This stability reduces the risk of production delays caused by degraded raw materials. Furthermore, the milder reaction conditions (15-60°C) place less thermal stress on reactor equipment, potentially extending asset life and reducing maintenance downtime, thereby ensuring a more consistent and reliable delivery schedule to customers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this technology is inherently designed for scalability. The exotherm of the hydrogen peroxide oxidation is manageable under the described conditions, reducing the risk of thermal runaway incidents common in large-scale oxidations. From an environmental perspective, the process aligns perfectly with modern green chemistry principles. The absence of heavy metals in the effluent simplifies wastewater treatment, allowing facilities to meet increasingly strict discharge regulations with standard neutralization protocols. This environmental compatibility future-proofs the manufacturing site against tightening regulations, ensuring long-term operational viability and protecting the brand reputation of partners who prioritize sustainable sourcing.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and quality assurance of this synthesis route. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering transparency into the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains.
Q: Why is hydrogen peroxide preferred over potassium permanganate for HCPCP oxidation?
A: Hydrogen peroxide oxidation generates water and gas as byproducts, eliminating the heavy metal sludge (MnO2) associated with potassium permanganate. This drastically simplifies post-treatment, reduces environmental hazards, and lowers waste disposal costs while maintaining high yields of 85%-92%.
Q: How does the new method improve product purity compared to traditional routes?
A: The process leverages the differential solubility of the intermediate and final product in alkaline solutions. HCPCP dissolves in alkaline water while unreacted HAPCP remains insoluble, allowing for highly effective separation and achieving final purities of 98%-99% without complex chromatography.
Q: What are the safety advantages of using potassium carbonate over sodium hydride?
A: Traditional methods often employ sodium hydride (NaH), which is pyrophoric and hazardous to handle on a large scale. This patented method utilizes anhydrous potassium carbonate or potassium phosphate, which are stable, non-hazardous solids, significantly enhancing operational safety and reducing insurance and handling costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexa(4-carboxy-phenoxy)-cyclotriphosphazene Supplier
The technical superiority of the hydrogen peroxide oxidation route positions HCPCP as a cornerstone material for next-generation flame retardant systems. At NINGBO INNO PHARMCHEM, we recognize that translating patent chemistry into commercial reality requires more than just a recipe; it demands deep process engineering expertise. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific solvent recovery and phase separation requirements of this process, ensuring that every batch meets stringent purity specifications verified by our rigorous QC labs. We understand that consistency is key for polymer additives, and our quality management systems are designed to deliver lot-to-lot reproducibility that your formulation teams can rely on.
We invite you to collaborate with us to optimize your supply chain for high-performance phosphazene derivatives. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener synthesis route can improve your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot runs and to discuss route feasibility assessments for your unique application needs. Let us help you secure a sustainable, cost-effective supply of this critical intermediate.
