Advanced Solid-Acid Quenching for High-Purity Diphenylphosphine Oxide Hydrogen Manufacturing
Introduction to Next-Generation Organophosphorus Synthesis
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic routes in the fine chemical industry. A pivotal advancement in this domain is detailed in Chinese Patent CN115490726B, which discloses a novel preparation method for diphenylphosphine oxide hydrogen (Ph2P(O)H). This compound serves as a critical building block for a vast array of downstream applications, ranging from ligand synthesis in homogeneous catalysis to the construction of complex pharmaceutical intermediates. The core innovation lies in the strategic replacement of traditional aqueous acid quenching with a solid-state acid neutralization protocol. This shift not only addresses the longstanding challenge of product hydration and instability but also fundamentally restructures the downstream processing workflow. For R&D directors and process chemists, this patent represents a paradigm shift from labor-intensive purification to streamlined isolation, promising a new standard for quality and operational efficiency in the production of high-value phosphorus reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the conversion of triphenylphosphine oxide (Ph3P(O)), a ubiquitous byproduct of Wittig reactions, into diphenylphosphine oxide hydrogen has relied heavily on reductive cleavage followed by aqueous acidic workup. As illustrated in the conventional pathway, the intermediate sodium salt (Ph2P(ONa)) is typically treated with hydrochloric acid or saturated ammonium chloride solutions.  This traditional approach suffers from severe inherent defects that compromise both yield and product integrity. Firstly, the introduction of water inevitably leads to the formation of hydrated species; pure Ph2P(O)H is a solid at room temperature, yet the aqueous method frequently yields hygroscopic oily products that are notoriously difficult to crystallize or purify. Secondly, the P(O) moiety exhibits a strong tendency to form complexes with residual acids, necessitating extensive alkaline washing procedures that further expose the sensitive P-H bond to oxidative degradation. Finally, the requirement for large volumes of organic solvents for extraction not only inflates production costs but also creates significant environmental burdens through waste generation, making the conventional route increasingly untenable for modern sustainable manufacturing standards.
This traditional approach suffers from severe inherent defects that compromise both yield and product integrity. Firstly, the introduction of water inevitably leads to the formation of hydrated species; pure Ph2P(O)H is a solid at room temperature, yet the aqueous method frequently yields hygroscopic oily products that are notoriously difficult to crystallize or purify. Secondly, the P(O) moiety exhibits a strong tendency to form complexes with residual acids, necessitating extensive alkaline washing procedures that further expose the sensitive P-H bond to oxidative degradation. Finally, the requirement for large volumes of organic solvents for extraction not only inflates production costs but also creates significant environmental burdens through waste generation, making the conventional route increasingly untenable for modern sustainable manufacturing standards.
The Novel Approach
In stark contrast, the methodology outlined in CN115490726B introduces a groundbreaking solid-phase quenching strategy that elegantly circumvents the pitfalls of liquid-liquid extraction. The core of this innovation involves the direct reaction of the phosphinate salt (Ph2PONa) with selected solid acids, such as silica gel powder, polymeric organic acids, or inorganic solid acids like zeolites.  By operating within a temperature range of -30°C to 150°C under an inert atmosphere, the solid acid effectively protonates the phosphinate anion to generate the target Ph2P(O)H while converting itself into an insoluble sodium salt. The brilliance of this design lies in the differential solubility properties: the desired product dissolves readily in organic solvents, whereas the spent solid acid and its sodium salts remain completely insoluble. This allows for a simple filtration step to separate the product from the quenching agents, followed by solvent removal to yield anhydrous, high-purity Ph2P(O)H. This approach eliminates the need for aqueous washing entirely, thereby preserving the anhydrous state of the product and drastically simplifying the post-reaction processing workflow for procurement and supply chain teams seeking robust manufacturing solutions.
By operating within a temperature range of -30°C to 150°C under an inert atmosphere, the solid acid effectively protonates the phosphinate anion to generate the target Ph2P(O)H while converting itself into an insoluble sodium salt. The brilliance of this design lies in the differential solubility properties: the desired product dissolves readily in organic solvents, whereas the spent solid acid and its sodium salts remain completely insoluble. This allows for a simple filtration step to separate the product from the quenching agents, followed by solvent removal to yield anhydrous, high-purity Ph2P(O)H. This approach eliminates the need for aqueous washing entirely, thereby preserving the anhydrous state of the product and drastically simplifying the post-reaction processing workflow for procurement and supply chain teams seeking robust manufacturing solutions.
Mechanistic Insights into Solid-Acid Mediated Protonation
To fully appreciate the technical superiority of this method, one must delve into the mechanistic nuances of the reduction and quenching steps. The initial formation of the Ph2PONa intermediate can be achieved through the reaction of Ph3P(O) with metallic sodium. Crucially, the patent highlights that by utilizing non-polar hydrocarbon solvents or specific protic compounds, the stoichiometry of sodium consumption can be optimized. Unlike traditional ether-based reductions that often require excess sodium to drive the formation of phenyl sodium (PhNa) intermediates which subsequently react with solvent, this method leverages radical pathways that favor the direct formation of the phosphinate salt. 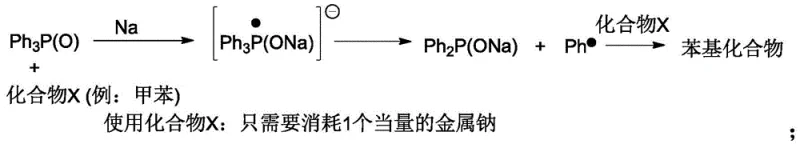 The subsequent quenching step relies on the surface hydroxyl groups present on solid acids like silica gel. These surface -OH groups act as proton donors, effectively neutralizing the basic Ph2PONa. The reaction kinetics are controlled by the accessibility of these active sites and the diffusion of the phosphinate salt into the porous structure of the solid acid. This heterogeneous reaction environment provides a unique advantage: it prevents the localized exotherms often seen in liquid acid additions, thereby minimizing thermal degradation of the sensitive P-H bond. Furthermore, the insolubility of the resulting sodium silicate or sodium salt of the polymeric acid ensures that no ionic contaminants remain in the organic phase, a level of purity that is exceptionally difficult to achieve via aqueous extraction where emulsion formation and ion carryover are common occurrences.
The subsequent quenching step relies on the surface hydroxyl groups present on solid acids like silica gel. These surface -OH groups act as proton donors, effectively neutralizing the basic Ph2PONa. The reaction kinetics are controlled by the accessibility of these active sites and the diffusion of the phosphinate salt into the porous structure of the solid acid. This heterogeneous reaction environment provides a unique advantage: it prevents the localized exotherms often seen in liquid acid additions, thereby minimizing thermal degradation of the sensitive P-H bond. Furthermore, the insolubility of the resulting sodium silicate or sodium salt of the polymeric acid ensures that no ionic contaminants remain in the organic phase, a level of purity that is exceptionally difficult to achieve via aqueous extraction where emulsion formation and ion carryover are common occurrences.
From an impurity control perspective, this mechanism offers unparalleled advantages for the production of high-purity pharmaceutical intermediates. The avoidance of water is paramount, as moisture can lead to the hydrolysis of sensitive intermediates or the promotion of oxidation side reactions that convert Ph2P(O)H into the undesired phosphinic acid (Ph2PO2H). By maintaining a strictly anhydrous environment throughout the quenching and isolation phases, the process inherently suppresses these degradation pathways. Additionally, the solid acid acts as a scavenger for basic impurities, trapping them within the solid matrix during filtration. This dual function of protonation and purification means that the crude organic filtrate is already of high quality, requiring minimal further refinement. For R&D teams focused on impurity profiling, this translates to a cleaner reaction profile with fewer unknown peaks in HPLC analysis, facilitating faster regulatory approval and more consistent batch-to-batch reproducibility in commercial scale-up of complex organophosphorus compounds.
How to Synthesize Diphenylphosphine Oxide Hydrogen Efficiently
The implementation of this synthesis route requires careful attention to atmospheric conditions and reagent selection to maximize the benefits of the solid-acid protocol. The process begins with the generation of the Ph2PONa precursor, which can be accomplished either through direct reduction with sodium metal in non-polar solvents or via reaction with protic compounds like water or alcohols in the presence of sodium. Once the phosphinate salt is prepared, it is suspended in a suitable organic solvent such as toluene, THF, or ethers. The critical step involves the addition of the solid acid—silica gel being a prime example due to its cost-effectiveness and high surface area. The mixture is stirred at controlled temperatures to ensure complete protonation without thermal stress. Following the reaction, the suspension is filtered under inert gas to remove the solid residues. The filtrate, containing the dissolved Ph2P(O)H, is then subjected to rotary evaporation under reduced pressure to remove the solvent, yielding the final anhydrous product as a white solid.
- React Triphenylphosphine Oxide (Ph3PO) with metallic sodium in a non-polar solvent or with a protic compound to generate Sodium Diphenylphosphinate (Ph2PONa).
- Quench the Ph2PONa intermediate with a solid acid (e.g., silica gel, zeolite) at temperatures between -30°C and 150°C under inert atmosphere.
- Filter the mixture to remove insoluble solid acid salts, then remove the organic solvent under reduced pressure to isolate anhydrous Ph2P(O)H.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this solid-acid mediated synthesis offers compelling economic and logistical advantages that extend far beyond simple yield improvements. The elimination of aqueous workup steps fundamentally alters the cost structure of manufacturing this key intermediate. By removing the need for large volumes of water, brine solutions, and the associated wastewater treatment protocols, the process significantly reduces the environmental footprint and operational overhead. Moreover, the simplification of the isolation procedure—from a multi-step extraction and washing sequence to a single filtration and evaporation—drastically shortens the batch cycle time. This efficiency gain allows for higher throughput in existing reactor setups without the need for capital-intensive equipment upgrades, directly contributing to substantial cost savings in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the optimization of reagent stoichiometry and the reduction of solvent consumption. Traditional methods often require excess sodium and large quantities of extraction solvents to recover product from aqueous layers, leading to significant material loss and disposal costs. In contrast, the solid-acid method utilizes stoichiometric amounts of sodium more efficiently and minimizes solvent usage by avoiding partitioning losses. The solid acids themselves, such as silica gel or industrial zeolites, are inexpensive and widely available commodities, ensuring that raw material costs remain low and stable. Furthermore, the ability to produce anhydrous product directly eliminates the energy-intensive drying steps often required to remove trace water from hygroscopic phosphorus compounds, resulting in lower utility costs and a more streamlined production budget.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the new process. The reliance on common, non-hazardous solid acids reduces the dependency on specialized corrosive liquids like concentrated hydrochloric acid, which can face regulatory shipping restrictions and supply volatility. The simplified workflow also reduces the risk of batch failures due to operational errors in complex extraction procedures, ensuring more consistent delivery schedules. For a reliable pharmaceutical intermediate supplier, this means the ability to maintain safety stock levels with greater confidence and respond more agilely to fluctuations in market demand. The reduced processing time per batch further enhances capacity utilization, allowing suppliers to fulfill large-volume orders with shorter lead times, a critical factor for clients managing just-in-time inventory strategies.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces challenges related to heat transfer and mixing, particularly in exothermic quenching steps. The heterogeneous nature of the solid-acid reaction provides a natural buffer against thermal runaways, making the process inherently safer and easier to scale from pilot plant to commercial tonnage. From an environmental compliance standpoint, the drastic reduction in aqueous waste streams simplifies effluent treatment and aligns with increasingly stringent global regulations on industrial discharge. The minimization of organic solvent waste through improved recovery rates further supports sustainability goals. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the brand value of the supply chain, appealing to end-users who prioritize environmentally responsible sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: Why is solid acid quenching superior to traditional aqueous acid methods?
A: Traditional methods using hydrochloric acid or ammonium chloride introduce water, leading to hygroscopic oily products that are difficult to purify. Solid acid quenching produces anhydrous solid products directly, simplifying isolation and preventing hydrolysis.
Q: How does this method improve the purity of Ph2P(O)H?
A: By avoiding aqueous workups, the risk of oxidation to Ph2PO2H is minimized. Furthermore, the insolubility of solid acid salts allows for clean separation via filtration, removing impurities that typically co-extract in liquid-liquid processes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process replaces complex extraction and washing steps with simple filtration and evaporation. This significantly reduces solvent usage and processing time, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenylphosphine Oxide Hydrogen Supplier
The technological advancements detailed in CN115490726B underscore the potential for significant improvements in the production of high-value organophosphorus intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of solid-acid quenching are fully realized in practical application. We operate stringent purity specifications and maintain rigorous QC labs equipped to handle sensitive phosphorus chemistry, guaranteeing that every batch of Diphenylphosphine Oxide Hydrogen meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage these process efficiencies for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can enhance your product quality while optimizing your overall procurement strategy.
