Advanced Hybrid Synthesis of Cholecystokinin Octapeptide for Scalable Pharmaceutical Manufacturing
Advanced Hybrid Synthesis of Cholecystokinin Octapeptide for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for producing complex peptide hormones that balance high purity with economic viability. A pivotal advancement in this domain is documented in patent CN102775471B, which discloses a sophisticated method for synthesizing Cholecystokinin Octapeptide (CCK-8), also known as Sincalide. This patent addresses critical bottlenecks in traditional peptide synthesis, specifically the instability of sulfated tyrosine residues and the high costs associated with enzymatic or purely liquid-phase routes. By integrating solid-phase efficiency with liquid-phase precision, this technology offers a viable pathway for the commercial scale-up of complex peptide intermediates. For R&D directors and procurement specialists, understanding this hybrid approach is essential for securing a reliable peptide intermediate supplier capable of delivering diagnostic-grade materials consistently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of CCK-8 has been plagued by significant technical hurdles that impact both yield and supply chain reliability. Traditional enzymatic methods, while specific, rely on expensive proteases such as thermolysin and papain, which are difficult to source in bulk and require complex downstream separation processes to remove proteinaceous impurities. Furthermore, purely liquid-phase chemical synthesis often involves cumbersome protection and deprotection cycles, leading to extended preparation periods and substantial solvent consumption. A major critical failure point in conventional solid-phase synthesis is the use of strong acids like hydrogen fluoride (HF) for final cleavage. HF is not only extremely hazardous to handle, requiring specialized equipment and rigorous safety protocols, but it also poses a severe threat to the integrity of the sulfate ester moiety on the tyrosine residue, leading to desulfation by-products that compromise the biological activity of the final API.
The Novel Approach
The methodology outlined in the patent data revolutionizes this landscape by employing a strategic hybrid of solid and liquid phase techniques. Instead of attempting to synthesize the entire octapeptide on resin, the process isolates the synthesis of the C-terminal dipeptide fragment (H-Asp-Phe-NH2) via liquid phase, ensuring high purity before it ever touches the resin. The remaining hexapeptide sequence is assembled on a 2-chlorotrityl chloride (CTC) resin using Fmoc chemistry. This approach allows for the use of weak acid cleavage conditions (utilizing trifluoroacetic acid mixtures) rather than anhydrous HF. This shift drastically simplifies the operational workflow, eliminates the need for specialized HF cleavage apparatus, and significantly enhances the stability of the sulfated tyrosine bond, resulting in a cleaner crude product profile and higher overall recovery rates suitable for industrial applications.
Mechanistic Insights into Hybrid Solid-Liquid Phase Peptide Synthesis
The core innovation lies in the meticulous selection of protecting groups and resin linkers designed to preserve the labile sulfate group. The synthesis initiates with the liquid-phase preparation of the dipeptide H-Asp(OtBu)-Phe-NH2. Here, Boc-Phe-OH is activated and amidated, followed by coupling with Fmoc-Asp(OtBu)-OH. The use of Fmoc (9-fluorenylmethoxycarbonyl) protection for the aspartic acid allows for orthogonal deprotection strategies that prevent side reactions during the subsequent condensation. This fragment is purified to over 98% purity before being reserved for the final ligation step, acting as a high-quality building block that reduces the burden on the final purification stage.
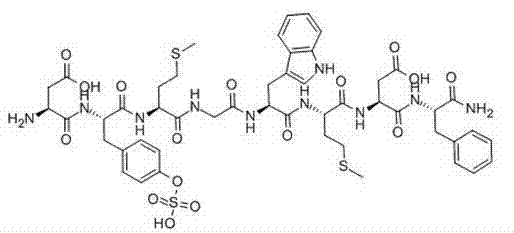
In the solid-phase segment, the choice of CTC resin is mechanistically critical. Unlike standard Wang resins that require 95% TFA or HF for cleavage, CTC resin forms a trityl ester linkage that is stable to piperidine (used for Fmoc removal) but cleavable with very mild acidic conditions (e.g., 1% TFA in DCM). This gentleness is paramount for the high-purity pharmaceutical intermediate production because the Tyr(SO3H) residue is highly acid-sensitive. The hexapeptide sequence (Met-Gly-Trp-Met-Asp-Tyr) is assembled stepwise on this resin. Once the full protected fragment is obtained, it is cleaved from the resin using a weak acid mixture, preserving the sulfate group. This fragment is then condensed with the pre-synthesized dipeptide in the liquid phase using DCC/HOBt activation. The final global deprotection removes the tert-butyl and Boc groups, yielding the crude CCK-8 which is subsequently purified via preparative HPLC to achieve pharmaceutical grade specifications.
How to Synthesize Cholecystokinin Octapeptide Efficiently
Implementing this hybrid protocol requires precise control over reaction stoichiometry and temperature to maximize coupling efficiency while minimizing racemization. The process begins with the rigorous preparation of the dipeptide fragment, followed by the loading of the first methionine onto the CTC resin at a controlled substitution degree of 0.65-0.80 mmol/g to prevent steric hindrance during chain elongation. Each coupling cycle is monitored via the Kaiser test to ensure quantitative reaction completion before proceeding.
- Synthesize the dipeptide fragment H-Asp(OtBu)-Phe-NH2 using liquid phase methods with Boc/Fmoc protection strategies.
- Construct the hexapeptide resin fragment Boc-Asp(OtBu)-Tyr(SO3H)-Met-Gly-Trp-Met-CTC Resin via solid-phase synthesis using Fmoc chemistry.
- Cleave the full-protection fragment using weak acid, condense with the dipeptide fragment in liquid phase, and perform final deprotection and HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this hybrid synthesis method represents a strategic opportunity to optimize cost structures and mitigate supply risks. The elimination of hazardous hydrogen fluoride from the process flow removes a significant bottleneck related to safety compliance and waste disposal. Facilities that are not equipped for HF handling can now produce this high-value intermediate, broadening the potential supplier base and enhancing supply chain resilience. Furthermore, the modular nature of the synthesis—producing fragments separately and condensing them—allows for better inventory management of intermediates and reduces the risk of total batch loss due to a single failed step in a long linear sequence.
- Cost Reduction in Manufacturing: The hybrid approach significantly lowers production costs by replacing expensive enzymatic catalysts with robust chemical reagents and eliminating the capital expenditure associated with HF-resistant equipment. The high yields reported in key steps, such as the 90.6% yield in dipeptide synthesis and over 90% yield in resin cleavage, translate directly into reduced raw material consumption per kilogram of final product. Additionally, the simplified workup procedures, such as ether precipitation instead of complex chromatographic separations at every step, reduce solvent usage and labor hours, driving down the overall cost of goods sold (COGS) for this API intermediate.
- Enhanced Supply Chain Reliability: By utilizing widely available Fmoc-amino acids and standard coupling reagents like DIC and HOBt, the method reduces dependency on specialty enzymes that may have long lead times or supply volatility. The robustness of the CTC resin chemistry ensures consistent batch-to-batch reproducibility, which is critical for maintaining regulatory compliance in pharmaceutical manufacturing. This stability allows suppliers to offer more reliable delivery schedules and maintain safety stock levels without the fear of rapid degradation of sensitive intermediates, thereby reducing lead time for high-purity peptide intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, moving seamlessly from gram-scale optimization to multi-kilogram production. The avoidance of heavy metals and hazardous HF aligns with increasingly stringent global environmental regulations, minimizing the cost and complexity of wastewater treatment. The use of weak acid cleavage generates less toxic waste streams compared to traditional methods, facilitating easier disposal and lowering the environmental footprint of the manufacturing site. This eco-friendly profile not only reduces regulatory risk but also appeals to end-clients seeking sustainable sourcing options for their diagnostic agent supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of CCK-8 using this patented hybrid methodology. These insights are derived directly from the experimental data and process descriptions found in the underlying intellectual property, providing a transparent view of the manufacturing capabilities.
Q: Why is the hybrid solid-liquid phase method preferred for CCK-8 synthesis?
A: The hybrid method combines the high purity of liquid phase synthesis for the C-terminal dipeptide with the efficiency of solid phase synthesis for the main chain. Crucially, it utilizes weak acid cleavage (TFA/DCM) instead of hazardous hydrogen fluoride (HF), significantly improving safety and reducing environmental waste while maintaining the stability of the sensitive sulfated tyrosine bond.
Q: What is the role of CTC Resin in this synthesis protocol?
A: CTC Resin (2-chlorotrityl chloride resin) is selected for its ability to be cleaved under very mild acidic conditions. This is essential for preserving the acid-labile sulfate ester group on the Tyrosine residue (Tyr(SO3H)), which would otherwise decompose under the harsher conditions required by standard Wang or MBHA resins.
Q: How does this method address impurity control in peptide manufacturing?
A: By synthesizing the difficult C-terminal dipeptide fragment separately in the liquid phase with high purity (>98%) before condensation, the method minimizes deletion sequences and racemization often seen in long linear solid-phase runs. Final purification via preparative HPLC ensures the removal of any remaining truncation products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholecystokinin Octapeptide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of diagnostic peptides like CCK-8. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated hybrid solid-liquid phase protocols are executed with precision. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art preparative HPLC and lyophilization systems to deliver material that meets the exacting standards required for pharmaceutical and diagnostic applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can enhance your project's efficiency and reliability.
