Efficient One-Step Synthesis of Trifunctional Phosphine Oxide Monomers for High-Performance Polymers
The development of advanced polymeric materials often hinges on the availability of high-performance monomers that can impart specific functional properties such as flame retardancy, thermal stability, and enhanced mechanical strength. In the realm of specialty chemicals, trifunctional phosphine oxide monomers like Tris(4-fluorophenyl)phosphine oxide (TFPPO) and Tris(4-hydroxyphenyl)phosphine oxide (TOHPPO) have emerged as critical building blocks for synthesizing branched or cross-linked polyaryl ether polymers. These materials find extensive applications in demanding sectors ranging from fuel cells and seawater desalination membranes to ionic polymer transducers and electromagnetic shielding technologies. A significant breakthrough in the efficient manufacturing of these intermediates is detailed in patent CN101863922B, which outlines a streamlined synthetic pathway that addresses longstanding inefficiencies in prior art. By leveraging a direct Grignard phosphorylation strategy followed by a robust hydrolysis step, this technology offers a compelling solution for reliable polymer additive supplier networks seeking to optimize their supply chains for next-generation electronic and engineering plastic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of trifunctional phosphine oxide monomers has been plagued by complex multi-step sequences that introduce significant operational burdens and safety hazards. Traditional literature describes the synthesis of TFPPO starting from 4-fluorobromobenzene and phosphorus trichloride, necessitating an initial Grignard reaction to form the phosphine intermediate, followed by a separate oxidation step using hydrogen peroxide to achieve the desired phosphine oxide state. Furthermore, the subsequent conversion to TOHPPO typically involves a methoxy-protected precursor route where 4-bromoanisole is reacted, oxidized, and finally demethylated using boron tribromide at extremely low temperatures around -78°C. This reliance on cryogenic conditions and highly toxic reagents like boron tribromide not only escalates the cost reduction in advanced materials manufacturing but also imposes severe constraints on reactor design and process safety. The cumulative effect of these harsh conditions is a process that is difficult to scale, prone to impurity formation, and economically inefficient due to the high energy consumption required for maintaining ultra-low temperatures.
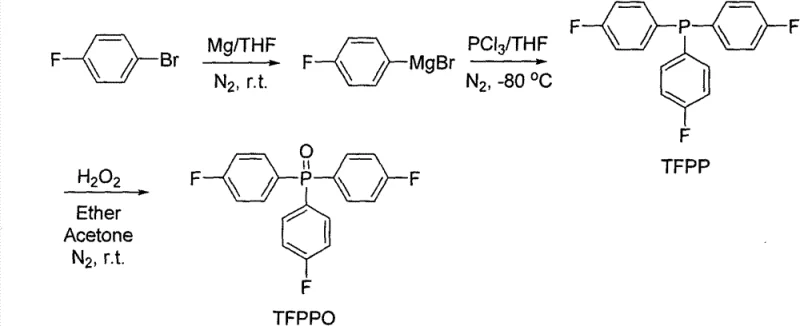
The Novel Approach
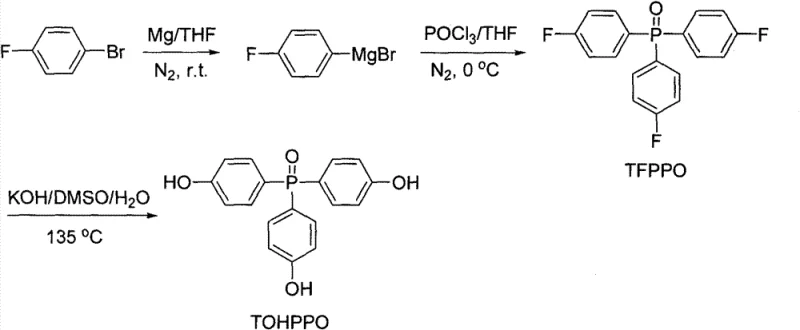
In stark contrast to the cumbersome legacy protocols, the innovative methodology presented in the patent data utilizes a direct and atom-economical approach that drastically simplifies the molecular construction of these valuable monomers. The core of this advancement lies in the direct reaction of fluorine-substituted bromobenzene with phosphorus oxychloride (POCl3) in a tetrahydrofuran solvent system, effectively bypassing the need for a distinct oxidation phase. By carefully controlling the reaction temperature between 0-5°C during the addition of the phosphorylating agent and subsequently allowing the mixture to warm to room temperature, the process achieves high conversion rates directly to the TFPPO intermediate. This consolidation of steps from two down to one represents a paradigm shift in process chemistry, reducing the total processing time and minimizing the exposure of reactive intermediates to potential degradation pathways. The subsequent transformation to TOHPPO is achieved through a straightforward nucleophilic substitution using aqueous potassium hydroxide in dimethyl sulfoxide at elevated temperatures, completely eliminating the need for hazardous demethylating agents and cryogenic cooling systems.
Mechanistic Insights into Direct Grignard Phosphorylation
The chemical elegance of this synthesis lies in the precise orchestration of Grignard reagent formation and its immediate interception by the electrophilic phosphorus center of phosphorus oxychloride. The process initiates with the generation of the aryl magnesium bromide species in situ, where the molar ratio of the halide to magnesium is tightly controlled between 1:1.0 and 1:1.1 to ensure complete consumption of the metal while minimizing side reactions such as Wurtz coupling. Upon the introduction of phosphorus oxychloride, the nucleophilic attack occurs sequentially at the phosphorus atom, displacing chloride ions to form the P-C bonds characteristic of the triarylphosphine oxide structure. The reaction is quenched with ammonium chloride solution, which serves to neutralize excess basicity and facilitate the separation of the organic product. This mechanistic pathway is particularly advantageous because it avoids the formation of unstable phosphine intermediates that require isolation and handling, thereby enhancing the overall safety profile and purity of the crude product before recrystallization.
Furthermore, the hydrolysis step converting TFPPO to TOHPPO operates through a classic nucleophilic aromatic substitution mechanism facilitated by the electron-withdrawing nature of the phosphine oxide group. The use of dimethyl sulfoxide (DMSO) as a polar aprotic solvent is critical here, as it enhances the nucleophilicity of the hydroxide ions generated from the 80-85% potassium hydroxide solution. Heating the reaction mixture to a range of 125-150°C provides the necessary activation energy to overcome the barrier for fluoride displacement, a transformation that is notoriously difficult under milder conditions. The stoichiometry is equally vital, with the patent specifying a molar excess of hydroxide (3-15 times the molar amount of TFPPO) to drive the equilibrium towards the fully substituted tri-hydroxy product. This rigorous control over reaction parameters ensures that the final TOHPPO product exhibits the high purity specifications required for downstream polymerization applications, free from partially substituted byproducts that could compromise the thermal properties of the final polymer matrix.
How to Synthesize Tris(4-hydroxyphenyl)phosphine Oxide Efficiently
Implementing this synthesis protocol requires strict adherence to inert atmosphere techniques and precise thermal management to maximize yield and safety. The procedure begins with the preparation of the Grignard reagent under nitrogen, followed by the controlled addition of the phosphorylating agent at low temperatures to manage the exotherm. Once the TFPPO intermediate is isolated and purified via recrystallization, it serves as the substrate for the high-temperature hydrolysis step in DMSO. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures derived from the patent examples, are outlined below to guide process engineers in replicating this high-efficiency route.
- Prepare the Grignard reagent by reacting fluorine-substituted bromobenzene with magnesium chips in THF at room temperature under nitrogen.
- Add phosphorus oxychloride (POCl3) dropwise at 0-5°C, then warm to room temperature to form Tris(fluorophenyl)phosphine oxide (TFPPO).
- Hydrolyze the TFPPO intermediate using aqueous potassium hydroxide in DMSO at 125-150°C to yield the final TOHPPO product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic benefits that extend beyond simple chemical yield improvements. By fundamentally altering the reaction topology to eliminate distinct oxidation and cryogenic demethylation stages, the process inherently reduces the number of unit operations required, which translates directly into lower capital expenditure for equipment and reduced operational overhead. The removal of boron tribromide, a reagent known for its corrosivity and handling difficulties, significantly lowers the regulatory burden associated with hazardous material storage and waste disposal. This simplification of the chemical bill of materials allows for a more resilient supply chain, as the reliance on specialized, hard-to-source reagents is minimized in favor of commodity chemicals like phosphorus oxychloride and potassium hydroxide. Consequently, this leads to substantial cost savings in raw material procurement and mitigates the risk of production stoppages due to reagent shortages.
- Cost Reduction in Manufacturing: The consolidation of the TFPPO synthesis from a two-step sequence into a single direct phosphorylation event eliminates the need for intermediate isolation and the associated solvent usage, pumping, and filtration costs. Furthermore, the avoidance of the expensive and toxic boron tribromide reagent in the TOHPPO production step removes a significant cost driver from the variable expense sheet. The process operates at near-ambient temperatures for the primary bond-forming step, drastically cutting energy consumption compared to the cryogenic requirements of legacy methods. These factors combine to create a manufacturing footprint that is leaner and more cost-effective, enabling competitive pricing for high-purity polymer additives without sacrificing quality margins.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as fluorobromobenzenes and phosphorus oxychloride, the process decouples production from the volatility of niche reagent markets. The robustness of the reaction conditions, which do not require specialized cryogenic reactors or complex distillation setups for sensitive intermediates, means that production can be scaled across a broader range of manufacturing facilities. This flexibility ensures consistent lead times for high-purity polymer additives, as the risk of batch failure due to temperature excursions or reagent instability is markedly reduced. Suppliers can therefore offer more reliable delivery schedules, supporting just-in-time manufacturing models for downstream polymer producers who depend on steady monomer flows.
- Scalability and Environmental Compliance: The transition away from ultra-low temperature operations and highly toxic demethylating agents aligns perfectly with modern environmental, health, and safety (EHS) standards. The simplified waste stream, devoid of boron-containing byproducts and peroxide residues, facilitates easier treatment and disposal, reducing the environmental impact of the manufacturing process. This green chemistry advantage is increasingly critical for commercial scale-up of complex polymer additives, as it eases the permitting process for new production lines and enhances the sustainability profile of the final product. The ability to run the hydrolysis step in a closed system with standard heating infrastructure further demonstrates the process readiness for multi-ton scale production, ensuring that supply can meet growing global demand for flame-retardant materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms traditional methods in terms of efficiency, safety, and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating these monomers into their existing polymer formulations or supply chains.
Q: How does the new synthesis route improve upon conventional methods for TFPPO?
A: The novel method condenses the traditional two-step synthesis into a single direct phosphorylation step using phosphorus oxychloride, eliminating the need for a separate oxidation stage with hydrogen peroxide and significantly improving overall yield.
Q: What are the safety advantages of producing TOHPPO via this patented method?
A: This process avoids the use of highly toxic demethylating agents like boron tribromide and eliminates the requirement for ultra-low temperature conditions (-78°C), making the operation safer and more suitable for industrial scale-up.
Q: Can this method be adapted for different isomeric forms of the monomer?
A: Yes, the protocol is versatile and supports the use of various fluorine-substituted bromobenzenes, including 2-fluoro, 3-fluoro, and 4-fluoro isomers, allowing for the production of diverse structural variants like 2-TOHPPO and 3-TOHPPO.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tris(4-hydroxyphenyl)phosphine Oxide Supplier
As the global demand for high-performance polymers continues to surge, securing a stable source of critical monomers like TFPPO and TOHPPO is paramount for maintaining competitive advantage in the materials sector. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in process optimization to deliver these complex intermediates with unmatched consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric needs of both pilot-scale research and full-scale industrial manufacturing. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of monomer meets the exacting standards required for advanced electronic and engineering applications.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your product portfolio and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term strategic goals in the advanced materials market.
