Advanced Synthesis of Thermally Stable Fluorinated Imidazoles for Next-Gen PCB Applications
Advanced Synthesis of Thermally Stable Fluorinated Imidazoles for Next-Gen PCB Applications
The electronics manufacturing industry is currently undergoing a critical transition towards lead-free soldering technologies, driven by stringent environmental regulations and the demand for higher reliability in consumer and industrial devices. This shift imposes severe thermal stress on Organic Solderable Preservative (OSP) treatment liquids, necessitating active ingredients that can withstand multiple high-temperature welding cycles without degradation. Patent CN101074212A introduces a breakthrough in this domain by disclosing a novel diphenyl-imidazole compound containing a fluorine atom, specifically 2-(4'-chlorophenyl)-4-(4'-fluorophenyl)imidazole. This compound represents a significant advancement over traditional halogenated imidazoles, offering exceptional thermal stability and purity profiles essential for high-end electronic chemical applications. The patent details a robust synthetic route that not only improves the physicochemical properties of the final active ingredient but also optimizes the manufacturing process for industrial scalability.
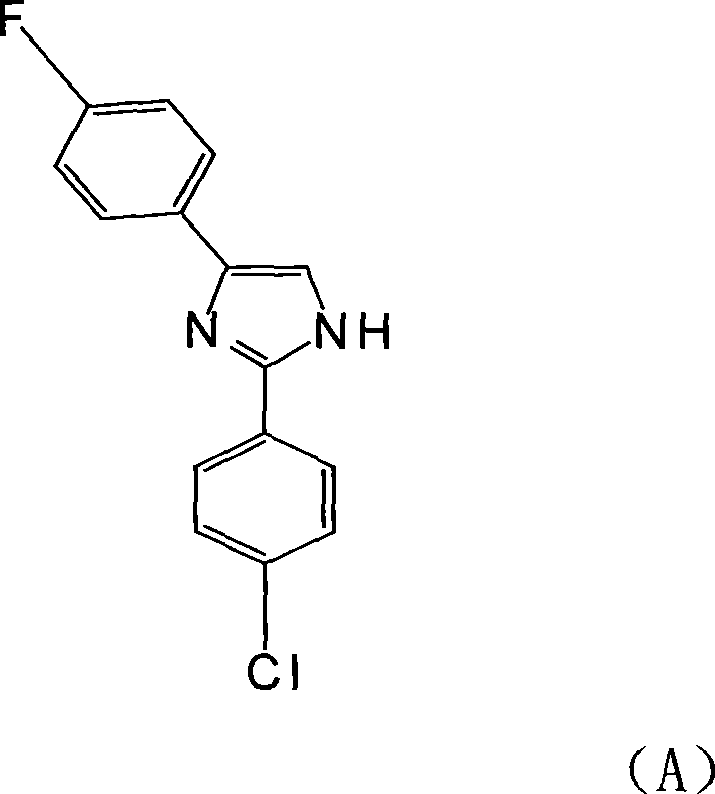
For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, understanding the structural nuances of this molecule is paramount. The strategic placement of the fluorine atom on the phenyl ring enhances the bond dissociation energy compared to chlorine or bromine analogues, directly translating to superior performance in the field. This technical insight report analyzes the proprietary synthesis method described in the patent, highlighting how it resolves long-standing issues regarding yield, safety, and product consistency in the production of complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing diaryl-imidazole compounds, such as those described in Chinese Patent CN1761773A, suffer from significant drawbacks that hinder their suitability for modern, large-scale industrial production. Traditional routes often rely on highly reactive and dangerous reagents, specifically sodium metal or sodium methoxide, to facilitate the condensation reaction. The use of such strong bases introduces severe safety hazards, including the risk of fire and explosion, which complicates logistics and increases insurance and handling costs for chemical manufacturers. Furthermore, these conventional homogeneous phase reactions typically utilize tetrahydrofuran (THF) as a solvent, which is highly flammable and prone to forming explosive peroxides upon storage. From a yield perspective, these older methods are inefficient, often resulting in crude product yields ranging merely from 30% to 71%, with substantial purification losses due to the formation of colored impurities that require extensive decolorization steps.
The Novel Approach
The methodology presented in CN101074212A fundamentally reengineers the synthesis pathway to address these safety and efficiency bottlenecks. The novel approach employs a three-step sequence that culminates in a mild, two-phase condensation reaction. Instead of hazardous sodium bases, the process utilizes potassium carbonate (K2CO3) in a chloroform-water biphasic system. This shift not only mitigates safety risks but also simplifies the workup procedure, as the product can be easily isolated by filtration after cooling. The optimization of reaction conditions, specifically extending the reflux time to 7 hours and adjusting the molar ratio of reactants to 1:1.1, has been shown to dramatically increase the yield to over 80%. This represents a massive improvement in atom economy and resource utilization compared to the legacy processes.
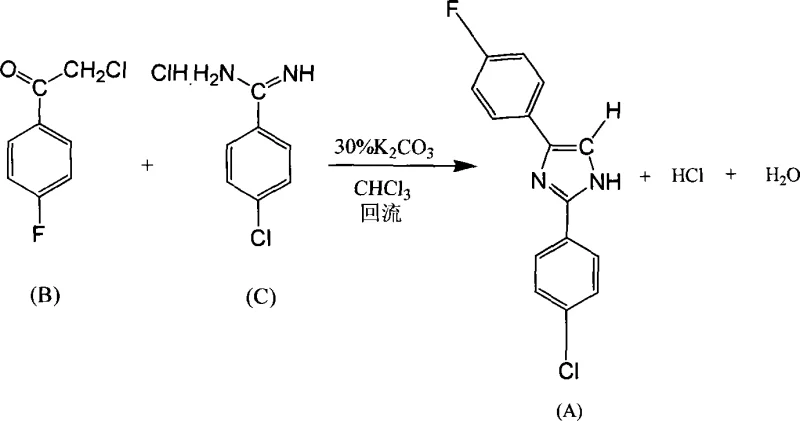
Moreover, the choice of solvents and reagents in this new route facilitates easier recycling and waste management. The use of chloroform in a two-phase system allows for efficient separation of the organic product from inorganic salts, reducing the burden on downstream purification units. For procurement managers focused on cost reduction in electronic chemical manufacturing, this transition from a dangerous, low-yield homogeneous process to a safe, high-yield biphasic process offers a compelling value proposition. It ensures a more consistent supply of high-purity intermediates while minimizing the operational risks associated with handling pyrophoric materials.
Mechanistic Insights into Base-Catalyzed Cyclization and Rearrangement
The formation of the imidazole ring in this specific fluorinated system involves a complex sequence of condensation and rearrangement steps that are critical for achieving the desired regioselectivity and purity. The reaction begins with the nucleophilic attack of the amidine nitrogen on the carbonyl carbon of the acetophenone derivative, facilitated by the mild basicity of the carbonate ion. Unlike simpler imidazole formations, the presence of the fluorine and chlorine substituents influences the electron density of the aromatic rings, requiring precise control over reaction kinetics to prevent side reactions. The initial cyclization forms a dihydro-intermediate, which then undergoes a crucial rearrangement to establish the aromatic imidazole core. This rearrangement is driven by the thermodynamic stability of the aromatic system and the elimination of water and hydrogen chloride.
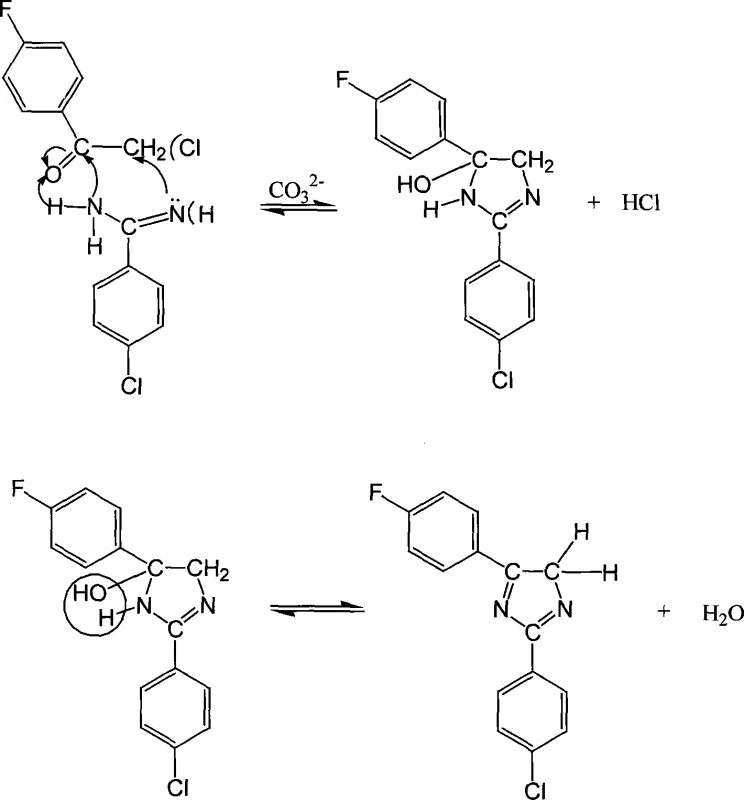
Understanding the impurity profile is essential for R&D teams aiming to replicate this quality. The patent data indicates that the use of stronger bases like NaOH leads to pale brown discoloration, suggesting oxidative degradation or polymerization side reactions. In contrast, the buffered environment provided by the potassium carbonate two-phase system maintains the integrity of the sensitive fluorinated intermediates. The mechanism further reveals that the final aromatization step involves a proton transfer and the restoration of conjugation across the imidazole ring, locking the molecule into its stable, planar configuration. This structural rigidity is what confers the exceptional thermal properties observed in the final product.
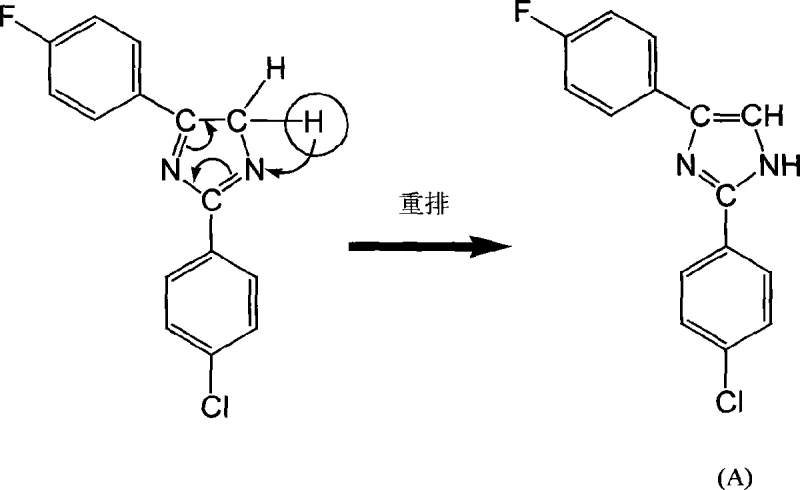
From a quality control perspective, the mechanistic pathway explains why the fluorinated analogue outperforms its chlorinated counterparts. The strong C-F bond resists homolytic cleavage at elevated temperatures where C-Cl or C-Br bonds might begin to degrade. This resistance to thermal decomposition ensures that the active ingredient remains chemically inert during the high-heat phases of PCB assembly, preventing the formation of corrosive byproducts that could damage copper traces. For technical buyers, this mechanistic robustness translates directly into higher reliability for the end-user's electronic components.
How to Synthesize 2-(4'-chlorophenyl)-4-(4'-fluorophenyl)imidazole Efficiently
The synthesis of this high-performance OSP active ingredient is achieved through a streamlined three-step protocol that balances reaction efficiency with operational safety. The process begins with the preparation of the ketone precursor via Friedel-Crafts acylation, followed by the conversion of a nitrile to an amidine salt, and concludes with the key condensation step. Each stage has been optimized to maximize yield and minimize impurity carryover. The detailed standardized synthesis steps, including specific stoichiometry, temperature controls, and workup procedures, are outlined in the guide below to assist process engineers in scaling this technology.
- Perform Friedel-Crafts acylation of fluorobenzene with chloroacetyl chloride using anhydrous AlCl3 to synthesize 2'-chloro-4-fluoroacetophenone.
- Convert 4-chlorobenzonitrile to 4-chlorobenzamidine hydrochloride via Pinner reaction with ethanol/HCl followed by ammonolysis.
- Condense the acetophenone and amidine derivatives in a chloroform-potassium carbonate two-phase system under reflux to form the final imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement officers, the adoption of the synthesis route described in CN101074212A offers distinct strategic advantages beyond mere technical performance. The shift away from hazardous reagents and the implementation of a high-yield biphasic system directly impact the total cost of ownership and supply continuity. By eliminating the need for specialized handling of sodium metal and reducing the complexity of waste treatment, manufacturers can achieve significant operational efficiencies. These improvements contribute to a more resilient supply chain capable of meeting the rigorous demands of the global electronics market.
- Cost Reduction in Manufacturing: The transition to a potassium carbonate-based catalytic system eliminates the expensive and dangerous sodium metal reduction steps found in legacy patents. This substitution drastically simplifies the reactor requirements and safety protocols, leading to substantial cost savings in both capital expenditure and ongoing operational maintenance. Furthermore, the increase in yield from approximately 30% in comparative examples to over 80% in the optimized process means that raw material consumption per kilogram of finished product is reduced by more than half, providing a direct and powerful lever for margin improvement.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as chloroform, potassium carbonate, and readily available fluorobenzene derivatives ensures that the supply chain is not vulnerable to the shortages often associated with specialized organometallic reagents. The robustness of the two-phase reaction system also allows for greater flexibility in batch sizing, enabling manufacturers to scale production up or down rapidly in response to market fluctuations without compromising product quality or safety standards.
- Scalability and Environmental Compliance: The use of a biphasic system facilitates easier solvent recovery and recycling, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing process. The avoidance of THF, a solvent with strict regulatory limits due to peroxide formation and flammability, further simplifies compliance with environmental health and safety (EHS) regulations. This makes the process highly scalable from pilot plant to multi-ton commercial production with minimal regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated imidazole compound. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and process safety.
Q: Why is the fluorinated imidazole superior to chlorinated analogues for OSP applications?
A: The C-F bond provides significantly higher thermal stability. Unlike chloro- or bromo-analogues which discolor or decompose upon melting, the fluorinated compound remains white and stable even after heating to 260°C, ensuring reliability during multiple lead-free soldering cycles.
Q: How does the new synthesis method improve safety compared to prior art?
A: The novel process eliminates the use of hazardous sodium metal or sodium methoxide required in previous methods. Instead, it utilizes mild potassium carbonate in a two-phase system, drastically reducing explosion risks and handling hazards.
Q: What represents the primary cost driver reduction in this manufacturing route?
A: The shift to a two-phase chloroform-water system allows for easier product isolation and solvent recovery. Additionally, the yield increase from approximately 30% to over 80% significantly reduces raw material waste and cost per kilogram.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4'-chlorophenyl)-4-(4'-fluorophenyl)imidazole Supplier
As the demand for high-reliability PCB surface treatment chemicals continues to grow, partnering with an experienced CDMO is essential for securing a stable supply of critical intermediates. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 2-(4'-chlorophenyl)-4-(4'-fluorophenyl)imidazole meets the exacting standards required for advanced electronic applications.
We invite you to engage with our technical team to explore how this patented technology can enhance your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and sourcing strategies effectively.
