Optimizing Tenofovir Dipivoxil Production via Novel Dehydration Catalysis for Commercial Scale
The global demand for antiretroviral therapies continues to drive the need for robust, scalable manufacturing processes for key active pharmaceutical ingredients (APIs) and their precursors. A pivotal development in this sector is documented in patent CN103880883A, which outlines a significantly modified method for preparing tenofovir dipivoxil, the lipophilic prodrug of tenofovir. This compound is essential for the production of Tenofovir Disoproxil Fumarate (TDF), a cornerstone medication in the treatment of HIV and Hepatitis B. The technical breakthrough described in this patent addresses a longstanding bottleneck in the alkylation of tenofovir: the uncontrollable formation of N-hydroxymethyl substituted impurities caused by moisture. By integrating a specific dehydration strategy using phosphorus pentoxide alongside phase-transfer catalysis, this methodology offers a pathway to higher purity intermediates, directly addressing the critical quality attributes required by regulatory bodies and procurement teams alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tenofovir dipivoxil, as reported in earlier literature such as US5922695, relied on the substitution reaction between tenofovir and chloromethyl isopropyl carbonate in solvents like N-Methyl pyrrolidone (NMP) using triethylamine. However, this conventional approach suffers from a significant chemical vulnerability: the extreme hygroscopicity of the tenofovir starting material. Tenofovir anhydride readily absorbs atmospheric moisture to form a stable monohydrate, which is difficult to remove completely even under standard industrial drying conditions. In the presence of this trace water, the reaction generates a substantial quantity of a specific byproduct known as the N-hydroxymethyl substituted impurity.
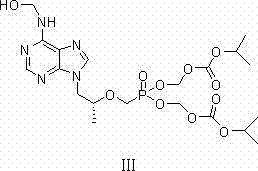
As illustrated in the structural analysis of the side reaction, this impurity typically accumulates to levels between 5% and 10% in traditional batches. This high impurity load not only drastically reduces the overall yield of the desired diester but also complicates downstream purification, requiring extensive chromatography or recrystallization steps that erode profit margins. Furthermore, previous attempts to mitigate this issue involved harsh and energy-intensive dehydration protocols that were time-consuming and difficult to reproduce consistently on a multi-ton scale, leading to batch-to-batch variability that is unacceptable for GMP manufacturing environments.
The Novel Approach
The innovative strategy presented in patent CN103880883A fundamentally alters the reaction landscape by introducing phosphorus pentoxide (P2O5) directly into the reaction matrix as a chemical water scavenger. Unlike physical drying methods, P2O5 reacts irreversibly with trace water—whether it originates from crystal water in the raw material or ambient humidity—thereby creating an essentially anhydrous environment in situ. This modification effectively suppresses the hydrolysis pathway that leads to the N-hydroxymethyl impurity, driving the reaction selectivity toward the desired bis-alkylation product. Concurrently, the inclusion of a phase-transfer catalyst, such as tetrabutyl ammonium bromide or benzyltriethylammonium chloride, enhances the nucleophilicity of the tenofovir species within the organic solvent.

This dual-catalyst system allows the reaction to proceed efficiently at moderate temperatures, typically between 45°C and 55°C, avoiding the thermal degradation risks associated with higher temperature processes. The result is a streamlined workflow where the impurity content is reduced to less than 1%, and the HPLC purity of the crude product often exceeds 97% prior to final crystallization. For a reliable pharmaceutical intermediate supplier, adopting this route translates to a more predictable process with significantly reduced waste generation and higher throughput capacity, aligning perfectly with the goals of cost reduction in API manufacturing.
Mechanistic Insights into P2O5-Mediated Dehydration and Alkylation
To fully appreciate the technical superiority of this method, one must analyze the mechanistic interplay between the dehydrating agent and the phase-transfer catalyst. Tenofovir possesses multiple nucleophilic sites, but the goal is selective O-alkylation to form the dipivoxil ester. In the presence of water, the electrophilic chloromethyl isopropyl carbonate can react with water to form unstable intermediates that subsequently attack the amine nitrogen of the adenine ring or the phosphonate oxygen, leading to the N-hydroxymethyl byproduct. Phosphorus pentoxide acts as a potent Lewis acid and dehydrating agent, sequestering water molecules as metaphosphoric acid species. By maintaining a near-zero water activity coefficient in the reaction medium, the kinetic competition favors the attack of the tenofovir phosphonate oxygens on the alkylating agent.
Furthermore, the phase-transfer catalyst plays a crucial role in solubilizing the ionic species generated by the organic base (such as triethylamine or DIPEA) and the acidic phosphonate groups of tenofovir. The quaternary ammonium cation forms a lipophilic ion pair with the deprotonated tenofovir, transporting it into the organic phase where the concentration of the alkylating agent is highest. This increases the effective collision frequency between reactants, accelerating the reaction rate without necessitating excessive heating. The synergy between the water-scavenging capability of P2O5 and the solubilizing power of the quaternary ammonium salt ensures that the reaction proceeds to completion with minimal formation of mono-ester intermediates or hydrolyzed byproducts, offering a robust solution for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Tenofovir Dipivoxil Efficiently
Implementing this synthesis requires precise control over reagent stoichiometry and addition rates to maximize the benefits of the P2O5 mediation. The process begins by dissolving tenofovir in N-Methyl pyrrolidone (NMP) and warming the mixture to approximately 45°C, followed by the addition of an organic base like triethylamine or DIPEA in a molar ratio of 4:1 to 8:1 relative to the substrate. Once the initial dissolution and neutralization are complete, the critical dehydration and catalysis components are introduced: phosphorus pentoxide flakes and the selected quaternary ammonium salt. The mixture is then heated to 55°C before the slow, dropwise addition of chloromethyl isopropyl carbonate. Detailed standardized synthetic steps for this optimized protocol are provided in the guide below.
- Dissolve Tenofovir in N-Methyl pyrrolidone (NMP) and heat to 45°C, then add an organic base such as triethylamine or DIPEA.
- Introduce a quaternary ammonium phase-transfer catalyst and Phosphorus Pentoxide (P2O5) flakes to the mixture at 55°C to scavenge moisture.
- Dropwise add chloromethyl isopropyl carbonate, monitor via HPLC until monoester content is below 10%, then proceed to filtration and extraction workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the methodology described in CN103880883A offers tangible strategic advantages beyond mere chemical elegance. The primary value proposition lies in the drastic simplification of the purification train. By suppressing the formation of the N-hydroxymethyl impurity at the source, manufacturers can avoid costly and time-consuming remediation steps such as preparative HPLC or multiple recrystallizations that are often required to meet stringent pharmacopeial standards. This streamlining of the downstream processing directly correlates to substantial cost savings in manufacturing, as it reduces solvent consumption, energy usage for evaporation, and labor hours associated with extended purification cycles.
- Cost Reduction in Manufacturing: The elimination of harsh pre-drying steps for the tenofovir starting material represents a significant operational efficiency. Traditional methods require rigorous drying of the hygroscopic raw material to prevent impurity formation, which consumes considerable energy and time. By utilizing P2O5 as an in-situ scavenger, the process tolerates standard commercial grades of raw materials without compromising quality. This flexibility allows procurement teams to source raw materials more broadly and potentially at lower costs, while the increased reaction yield ensures that more product is obtained per unit of input, effectively lowering the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: Process stability is the bedrock of supply chain security. The variability introduced by ambient humidity in conventional syntheses often leads to batch failures or off-spec material, causing delays in delivery schedules. The robust nature of this P2O5-mediated process mitigates the risk of environmental fluctuations impacting production quality. By ensuring consistent impurity profiles below 1%, manufacturers can guarantee shorter lead times for high-purity pharmaceutical intermediates, providing their clients with the certainty needed for their own production planning and regulatory filings.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route is superior. The reduction in solvent usage for purification decreases the volume of hazardous waste requiring disposal. Additionally, the reaction operates at moderate temperatures (45-55°C), reducing the thermal load on plant infrastructure compared to high-temperature alternatives. The ability to scale this process from kilogram to multi-ton quantities without losing selectivity makes it an ideal candidate for long-term supply contracts, ensuring that the supply of this critical antiviral intermediate remains uninterrupted even during periods of surging global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this specific synthetic route. Understanding these parameters is essential for technology transfer teams evaluating the feasibility of adopting this process into their existing manufacturing suites. The answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance.
Q: What is the primary function of Phosphorus Pentoxide in this synthesis?
A: Phosphorus Pentoxide (P2O5) acts as a critical water-absorbing agent. Since Tenofovir is highly hygroscopic and prone to forming stable monohydrates, trace water promotes the formation of N-hydroxymethyl substituted impurities. P2O5 chemically reacts with this moisture, effectively suppressing impurity generation and improving yield.
Q: Which phase-transfer catalysts are suitable for this reaction?
A: The patent specifies various quaternary ammonium salts are effective, including Tetrabutyl ammonium bromide, benzyltriethylammonium chloride, and tetradecyl trimethyl ammonium chloride. These catalysts facilitate the interaction between the organic base and the reactants in the NMP solvent system.
Q: How does this method improve upon conventional US5922695 synthesis?
A: Conventional methods often struggle with 5% to 10% levels of N-hydroxymethyl impurities due to water content. This modified method reduces that impurity to less than 1% without requiring harsh, energy-intensive dehydration pre-treatment steps, resulting in a more stable and scalable industrial process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Dipivoxil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires more than just a patent; it demands deep process engineering expertise. Our technical team has extensively analyzed the P2O5-mediated pathway for tenofovir dipivoxil and is prepared to assist partners in optimizing this route for their specific production needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chemistry are fully realized in a GMP-compliant setting. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards required for antiviral drug substance manufacturing.
We invite global pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this advanced synthesis technology. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your current production bottlenecks. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Let us help you secure a stable, high-quality supply of tenofovir intermediates that drives efficiency and profitability in your antiviral portfolio.
