Optimized Synthesis of GS-101: Enhancing Purity and Scalability for Tenofovir Prodrug Manufacturing
Optimized Synthesis of GS-101: Enhancing Purity and Scalability for Tenofovir Prodrug Manufacturing
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical antiviral intermediates, particularly those serving as precursors for Hepatitis B treatments. Patent CN113429439A introduces a refined preparation method for GS-101, a pivotal intermediate in the synthesis of tenofovir disoproxil fumarate. Tenofovir, a nucleotide reverse transcriptase inhibitor, faces challenges regarding bioavailability due to its double anionic phosphonic acid group under physiological pH conditions. Consequently, the development of phosphonate prodrugs like GS-101 is essential to enhance cell membrane permeability and therapeutic efficacy. This patent addresses the scarcity of optimized process parameters in prior art, specifically focusing on reaction temperature, time, and catalyst loading to maximize yield and purity.
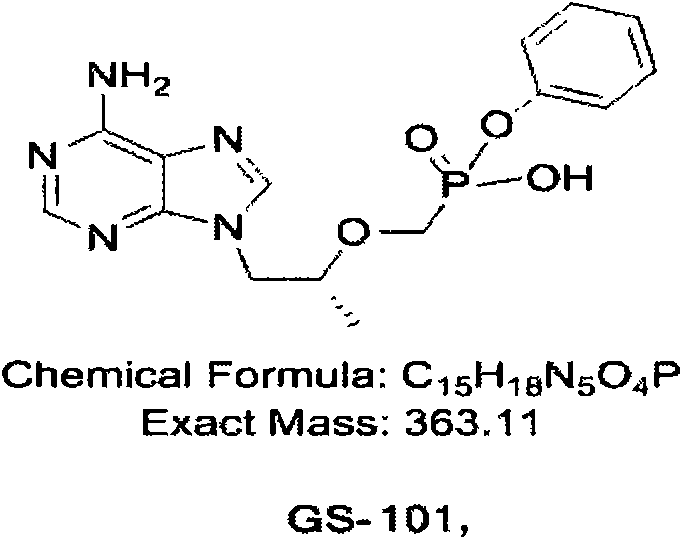
For R&D directors and procurement managers, understanding the structural integrity and synthetic accessibility of GS-101 is paramount. The molecule features a purine base linked to a phosphonate ester moiety, requiring precise chemical handling to avoid degradation or side reactions. The disclosed method offers a reliable pathway for producing high-purity pharmaceutical intermediates, ensuring that downstream processing for the final API remains efficient and compliant with stringent regulatory standards. By optimizing the coupling of the nucleoside analogue with the phosphonate precursor, this technology supports the global supply chain for antiviral medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in this patent, the synthesis of tenofovir prodrug intermediates often suffered from inconsistent yields and poor impurity profiles due to a lack of systematic parameter optimization. Conventional methods frequently failed to account for the sensitivity of the phosphonate esterification to thermal variations and catalyst concentration. For instance, operating at suboptimal temperatures, such as 75°C, resulted in sluggish reaction kinetics where the starting material GS-SM-101 remained largely unconverted even after extended periods. Conversely, excessive heating to 90°C accelerated degradation pathways, leading to a surge in impurity levels that compromised the purity of the final product to unacceptable levels below 90%. Furthermore, imprecise control over the molar ratios of bases like triethylamine often led to incomplete reactions or difficult purification scenarios, creating bottlenecks in commercial scale-up of complex nucleotide analogs.
The Novel Approach
The novel approach presented in CN113429439A overcomes these historical deficiencies through a rigorously optimized catalytic system utilizing DMAP and triethylamine in acetonitrile. By pinpointing a narrow temperature window of 83°C ± 2°C, the process achieves a balance between reaction rate and selectivity, driving the conversion of GS-SM-101 to near completion while suppressing side reactions. The strategic use of DMAP as a nucleophilic catalyst significantly lowers the activation energy for the phosphorylation step, allowing for high yields exceeding 80% under controlled conditions. This method transforms the synthesis from a trial-and-error operation into a predictable, high-efficiency process suitable for industrial manufacturing. The result is a robust protocol that consistently delivers GS-101 with purity levels above 97%, directly addressing the quality requirements of modern pharmaceutical production.
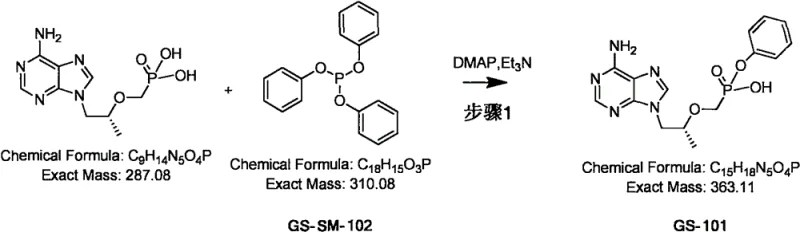
Mechanistic Insights into DMAP-Catalyzed Phosphorylation
The core of this synthetic breakthrough lies in the mechanistic efficiency of the DMAP-catalyzed esterification. In this reaction, 4-dimethylaminopyridine (DMAP) acts as a superior nucleophilic catalyst compared to traditional pyridine derivatives. The mechanism involves the initial attack of the DMAP nitrogen on the phosphorus center of the phosphonate precursor (GS-SM-102), forming a highly reactive phosphonium intermediate. This activated species is then rapidly attacked by the hydroxyl group of the nucleoside analogue (GS-SM-101), facilitating the formation of the P-O-C bond that characterizes the prodrug structure. Triethylamine serves a dual purpose: it acts as a proton scavenger to neutralize the acidic byproducts generated during the reaction and helps maintain the basic environment necessary for the nucleophilic attack to proceed efficiently. This synergistic interaction between the catalyst and the base ensures that the reaction proceeds smoothly without the need for harsh reagents that could damage the sensitive purine ring.
Impurity control is another critical aspect managed by this mechanistic design. The patent data highlights that deviations in temperature or reagent stoichiometry can lead to specific impurity profiles. For example, insufficient base (TEA) leads to incomplete neutralization, stalling the reaction and leaving starting materials that are difficult to separate. Excessive base or incorrect temperatures can promote hydrolysis of the phosphonate ester or degradation of the adenine base. By adhering to the optimized molar ratios—specifically maintaining TEA at approximately twice the molar amount of the starting material—the process minimizes these risks. The subsequent workup, involving acidification to pH 2-3, exploits the solubility differences between the target zwitterionic product and neutral organic impurities, ensuring that the final isolated solid meets the rigorous specifications required for a reliable pharmaceutical intermediate supplier.
How to Synthesize GS-101 Efficiently
The synthesis of GS-101 described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of precise reagent addition and thermal control. The process begins with the charging of the nucleoside starting material and solvent, followed by the sequential addition of the base, phosphonate precursor, and catalyst. Maintaining the reaction temperature at the optimal 83°C is critical for driving the reaction to completion within a 24-hour window. Following the reaction, a specialized workup procedure involving solvent exchange and pH-controlled precipitation is employed to isolate the product in high yield. For a comprehensive understanding of the operational parameters, the detailed standardized synthesis steps are outlined in the guide below.
- Charge GS-SM-101, acetonitrile, triethylamine (TEA), GS-SM-102, and DMAP into a reactor under stirring.
- Heat the reaction mixture to 83°C and maintain stirring for approximately 24 hours until HPLC indicates completion.
- Remove solvent, extract with ethyl acetate, adjust aqueous phase pH to 2-3 with hydrochloric acid to precipitate the product, then filter and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this optimized synthesis route offers substantial strategic benefits beyond mere technical success. The primary advantage lies in the significant yield improvement observed in the optimized examples compared to the comparative data. Achieving yields in the range of 80% versus the 60% seen in suboptimal conditions translates directly to reduced raw material consumption per kilogram of finished product. This efficiency gain is a powerful driver for cost reduction in API manufacturing, as it lowers the effective cost of goods sold (COGS) and minimizes the volume of chemical waste requiring disposal. Furthermore, the use of common, commercially available reagents like acetonitrile, triethylamine, and DMAP ensures that the supply chain remains resilient and不受制于 exotic or scarce catalysts.
- Cost Reduction in Manufacturing: The elimination of inefficient reaction conditions and the maximization of yield inherently reduce the manufacturing cost per unit. By avoiding the need for repeated recrystallizations or complex chromatographic purifications that would be necessary to fix low-purity batches, the process streamlines the production workflow. The qualitative reduction in waste generation also lowers environmental compliance costs, contributing to a more sustainable and economically viable production model. This efficiency allows manufacturers to offer competitive pricing for high-purity pharmaceutical intermediates without compromising on quality margins.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance within the 81-85°C range, reduces the risk of batch failures due to minor thermal fluctuations. This reliability is crucial for maintaining consistent delivery schedules to downstream API manufacturers. Since the starting materials GS-SM-101 and GS-SM-102 are standard intermediates with established supply chains, the risk of raw material shortages is minimized. This stability ensures reducing lead time for high-purity pharmaceutical intermediates, allowing partners to plan their inventory and production cycles with greater confidence and accuracy.
- Scalability and Environmental Compliance: The process utilizes a straightforward workup involving liquid-liquid extraction and acid precipitation, which are unit operations easily scalable from kilograms to metric tons. Unlike processes requiring cryogenic conditions or high-pressure hydrogenation, this method can be implemented in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. The simplified isolation procedure reduces the demand for large volumes of organic solvents during purification, aligning with green chemistry principles. This ease of scale-up facilitates the commercial scale-up of complex nucleotide analogs, ensuring that supply can meet global demand for antiviral therapies effectively.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of GS-101, derived directly from the experimental data and background provided in the patent documentation. These insights are intended to clarify the critical process parameters that define the success of this synthetic route. Understanding these factors is essential for any partner considering the integration of this intermediate into their supply chain.
Q: Why is the reaction temperature critical in GS-101 synthesis?
A: According to patent CN113429439A, maintaining the temperature at 83°C ± 2°C is crucial. Temperatures as low as 75°C result in slow reaction rates and incomplete conversion, while temperatures reaching 90°C significantly increase the risk of impurity formation, reducing both yield and purity.
Q: What role does DMAP play in this synthetic route?
A: DMAP (4-dimethylaminopyridine) acts as a potent nucleophilic catalyst in this esterification/phosphorylation reaction. The patent data indicates that optimizing the molar ratio of DMAP relative to the starting material is essential for achieving high conversion rates within a reasonable timeframe.
Q: How is high purity achieved in the final GS-101 product?
A: High purity (>97%) is achieved through a specific workup procedure involving solvent removal, extraction, and critically, the acidification of the aqueous phase to pH 2-3. This step precipitates the product while leaving soluble impurities in the solution, followed by washing with acetonitrile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GS-101 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates like GS-101 play in the global fight against Hepatitis B. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of large-scale API manufacturers. We are committed to delivering products that adhere to stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific thermal and chemical requirements of nucleotide synthesis, guaranteeing a consistent supply of this vital building block.
We invite potential partners to engage with our technical procurement team to discuss how our optimized GS-101 synthesis can benefit your specific production needs. By requesting a Customized Cost-Saving Analysis, you can evaluate the economic impact of switching to our high-yield process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
