Advanced Catalyst-Free Synthesis of 2-Pyridylpyridine Derivatives for Commercial Scale Manufacturing
Advanced Catalyst-Free Synthesis of 2-Pyridylpyridine Derivatives for Commercial Scale Manufacturing
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective manufacturing processes. Patent CN1151132C introduces a groundbreaking methodology for the production of 2-pyridylpyridine derivatives, which serve as critical building blocks in the synthesis of pharmaceuticals, agrochemicals, and advanced electronic materials. This technology represents a paradigm shift away from traditional transition-metal catalyzed couplings, offering a route that is not only environmentally benign but also economically superior for large-scale operations. By leveraging a condensation reaction between acetyl-substituted pyridine derivatives and 3-aminoacrolein compounds, followed by cyclization with ammonia or ammonium salts, this process achieves high yields and exceptional purity without the burden of expensive catalysts. For R&D directors and procurement managers alike, understanding the mechanistic elegance and commercial viability of this patent is essential for securing a competitive edge in the global supply chain of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-pyridylpyridine scaffolds has relied heavily on cross-coupling reactions that pose significant logistical and financial challenges for industrial manufacturers. Traditional approaches, such as the Ullmann reaction between halogenated pyridines, often necessitate the use of stoichiometric amounts of copper or expensive palladium catalysts, which drive up raw material costs substantially. Furthermore, these metal-catalyzed processes frequently suffer from poor selectivity, leading to complex mixtures of regioisomers and homocoupling byproducts that are notoriously difficult to separate. As illustrated in the comparative data associated with this technology, conventional routes can generate significant quantities of unwanted isomers, such as the 2,2'-bipyridine (VIII) and 4,4'-bipyridine (IX) byproducts alongside the desired 2,4'-bipyridine (VII), complicating downstream purification.
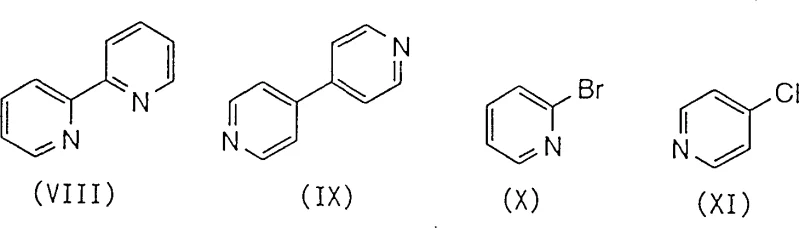
Beyond the issue of selectivity, the reliance on heavy metals introduces severe environmental and regulatory hurdles. The disposal of metal-containing waste streams requires specialized treatment protocols to meet stringent environmental standards, adding hidden costs to the overall production budget. Additionally, methods utilizing Grignard reagents or lithium metal demand rigorous exclusion of moisture and oxygen, necessitating specialized equipment and increasing operational complexity. These factors collectively render many conventional synthetic routes unsuitable for the high-volume, cost-sensitive manufacturing required by the modern pharmaceutical and agrochemical industries, creating a clear demand for alternative technologies that can deliver high purity with simplified processing.
The Novel Approach
The innovative process disclosed in Patent CN1151132C circumvents these traditional bottlenecks by employing a metal-free condensation strategy that utilizes readily available starting materials. Instead of relying on halogenated precursors and transition metals, this method reacts acetyl-substituted pyridine derivatives with 3-aminoacrolein compounds in the presence of a base, followed by cyclization with an ammonium source. This approach fundamentally alters the impurity profile, drastically reducing the formation of difficult-to-remove isomers and eliminating the need for metal scavenging steps. The versatility of this route is demonstrated by its ability to synthesize a variety of valuable derivatives, including 4,4'-dimethyl-2,2'-bipyridine (V), 3-(2-pyridyl)quinoline (VI), and 2,4'-bipyridine (VII), all with high structural regularity.
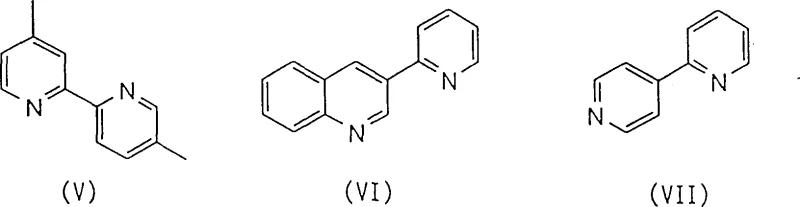
From a commercial perspective, this novel approach offers a streamlined pathway that aligns perfectly with the goals of cost reduction in pharmaceutical intermediate manufacturing. The elimination of precious metal catalysts not only lowers the direct cost of goods sold but also simplifies the supply chain by removing dependency on volatile metal markets. Moreover, the reaction conditions are mild and adaptable, typically proceeding in common solvents like tetrahydrofuran (THF) at moderate temperatures ranging from 40°C to 120°C. This operational simplicity translates directly into enhanced process robustness, allowing manufacturers to scale production from pilot plants to multi-ton commercial facilities with minimal risk and capital expenditure, ensuring a reliable supply of high-quality intermediates for downstream applications.
Mechanistic Insights into Base-Promoted Condensation and Cyclization
The core of this technological breakthrough lies in the precise control of the condensation and subsequent cyclization steps, which proceed through a well-defined mechanistic pathway devoid of radical intermediates often seen in metal-catalyzed systems. The reaction initiates with the deprotonation of the acetyl group on the pyridine ring by a strong base, such as potassium tert-butoxide, generating a reactive enolate species. This nucleophile then attacks the electron-deficient carbon of the 3-aminoacrolein derivative, forming a carbon-carbon bond that establishes the backbone of the future bipyridine system. The use of specific bases like potassium tert-butoxide or sodium hydride is critical, as they provide the necessary basicity to drive the initial condensation without promoting undesirable side reactions, ensuring high conversion rates of the starting materials.
Following the initial condensation, the intermediate undergoes a cyclization reaction upon treatment with ammonia or an ammonium salt, such as ammonium acetate, often in the presence of a weak acid catalyst like acetic acid. This step facilitates the closure of the second pyridine ring, aromatizing the system to yield the final 2-pyridylpyridine derivative. The inclusion of an acid catalyst during the cyclization phase is a key optimization that accelerates the reaction kinetics, allowing the process to reach completion within shorter timeframes, typically between 5 to 12 hours. This mechanistic efficiency is crucial for impurity control, as rapid and complete conversion minimizes the residence time of reactive intermediates that could otherwise degrade or form polymeric byproducts, thereby securing the high purity levels (>98%) observed in the experimental examples.
How to Synthesize 2-Pyridylpyridine Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity, particularly regarding the stoichiometry of the ammonium source and the choice of solvent. The process generally begins by dissolving the acetyl-substituted pyridine and the 3-aminoacrolein derivative in a polar aprotic solvent like THF, followed by the addition of the base at temperatures between 40°C and 80°C. Once the condensation is underway, the ammonium salt and acid catalyst are introduced, and the temperature is elevated to drive the cyclization to completion. Detailed standard operating procedures for optimizing these parameters, including specific molar ratios and workup techniques, are essential for reproducibility.
- React an acetyl-substituted pyridine derivative with 3-aminoacrolein in the presence of a base such as potassium tert-butoxide in a solvent like THF at 40-80°C.
- Add an ammonium salt (e.g., ammonium acetate) and an acid catalyst (e.g., acetic acid) to the reaction mixture to facilitate cyclization.
- Heat the mixture to 60-120°C to complete the reaction, followed by workup involving neutralization, extraction, and recrystallization to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free technology offers profound strategic benefits that extend far beyond simple chemical yield improvements. The most immediate impact is seen in the drastic simplification of the supply chain, as the process eliminates the need for sourcing expensive and often supply-constrained transition metal catalysts like palladium or specialized ligands. This reduction in raw material complexity directly correlates to a more stable and predictable cost structure, shielding the organization from the volatility of the precious metals market. Furthermore, the absence of heavy metals in the reaction mixture removes the necessity for costly purification steps such as activated carbon treatment or specialized resin scavenging, which are standard requirements for API intermediates to meet strict residual metal specifications.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, primarily driven by the elimination of high-cost catalysts and the associated waste treatment expenses. By avoiding the use of palladium, nickel, or copper, manufacturers can achieve substantial cost savings on raw materials while simultaneously reducing the environmental fees associated with hazardous waste disposal. The simplified downstream processing, which often requires only standard extraction and recrystallization rather than complex chromatographic separations, further lowers the operational expenditure per kilogram of product. This efficiency allows for a more competitive pricing strategy in the global market for pharmaceutical and agrochemical intermediates, enhancing profit margins without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity chemicals as starting materials. Acetyl-pyridines and aminoacroleins are widely available from multiple global suppliers, reducing the risk of single-source dependency that often plagues projects relying on proprietary catalysts. The robustness of the reaction conditions, which do not require inert atmospheres or cryogenic temperatures, also means that production can be maintained across a wider range of manufacturing sites, including those with less specialized infrastructure. This flexibility ensures continuous supply continuity even during periods of global logistical disruption, providing a critical safety net for long-term project planning and inventory management.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles, facilitating easier regulatory approval and community acceptance. The absence of toxic heavy metals simplifies the handling of effluent streams, reducing the burden on wastewater treatment facilities and minimizing the environmental footprint of the manufacturing site. The scalability of the process is proven by its ability to operate effectively in standard reactor vessels without the need for specialized equipment like ultrasonic generators or high-pressure autoclaves. This ease of scale-up ensures that production volumes can be rapidly increased to meet market demand, from hundreds of kilograms to multi-ton annual capacities, without encountering the technical barriers typical of more complex synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, drawing directly from the data and examples provided in the patent literature. Understanding these details is vital for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The answers reflect the specific advantages in terms of purity, yield, and operational simplicity that distinguish this method from legacy technologies.
Q: What are the primary advantages of this synthesis method over traditional Ullmann reactions?
A: Unlike traditional Ullmann reactions which require expensive copper or palladium catalysts and generate difficult-to-separate byproducts, this novel process utilizes readily available acetyl-pyridines and aminoacroleins. It eliminates the need for heavy metal catalysts, thereby removing the costly and complex step of metal scavenging and significantly reducing environmental waste treatment costs.
Q: What level of purity can be achieved using this catalyst-free protocol?
A: The process demonstrates exceptional selectivity, achieving product purities exceeding 98% as confirmed by HPLC analysis in multiple embodiments. For instance, 4,4'-dimethyl-2,2'-bipyridine was isolated with 98.0% purity, and 2,4'-bipyridine reached 99.3% purity, indicating a highly controlled reaction pathway with minimal side-product formation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is designed for industrial scale-up. It avoids specialized equipment like high-pressure reactors or ultrasonic apparatuses required by other methods. The use of common solvents like THF and standard heating conditions (40-120°C) ensures that the process can be easily transferred to multi-ton production facilities without significant capital investment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Pyridylpyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst-free synthesis route described in Patent CN1151132C for the production of high-value heterocyclic intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this technology are fully realized in a practical manufacturing setting. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-pyridylpyridine derivatives conforms to the highest industry standards for pharmaceutical and agrochemical applications.
We invite forward-thinking partners to collaborate with us to leverage this efficient and sustainable manufacturing technology for their next-generation projects. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in an increasingly competitive global market.
