Advanced Manufacturing of Aminophenyl Beta-Hydroxyethyl Sulfone Sulfate for Reactive Dyes
Introduction to Advanced Intermediate Synthesis
The production of high-performance reactive dye intermediates has long been challenged by complex multi-step syntheses and significant environmental burdens. Patent CN101525309B introduces a transformative methodology for producing aminophenyl beta-hydroxyethyl sulfone sulfate, a critical precursor for vinyl sulfone reactive dyes widely used in the textile industry. This innovation shifts the synthetic paradigm from the traditional, hazardous acetanilide sulfonation route to a more streamlined process starting from p-nitrochlorobenzene and mercaptoethanol. By leveraging specific catalytic systems, including sodium tungstate for oxidation and palladium carbon for hydrogenation, this technology addresses the dual imperatives of economic efficiency and environmental compliance. For global procurement leaders and R&D directors, understanding this shift is vital, as it represents a move towards sustainable chemical manufacturing that does not compromise on purity or scalability. The detailed reaction engineering described in the patent offers a robust framework for reliable reactive dye intermediate supplier operations, ensuring consistent quality for downstream dye synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of p-amino phenyl-beta-hydroxyethyl sulfone sulfate has relied on a cumbersome pathway initiating with acetanilide and chlorosulfonic acid. This legacy process is fraught with operational hazards and inefficiencies, primarily due to the exothermic nature of the sulfonation reaction which requires rigorous temperature control below -10°C using ice-salt solutions. The generation of substantial quantities of hydrogen chloride gas necessitates complex four-stage water absorption systems to produce industrial hydrochloric acid as a by-product, adding layers of equipment complexity and safety risks. Furthermore, the subsequent reduction and condensation steps involve sodium pyrosulfite and ethylene oxide, generating massive volumes of high-salt wastewater and spent acid. Data indicates that the conventional route produces approximately 6-8 tons of wastewater per ton of product, alongside significant amounts of abraum salt acid and sulfur waste acid, creating a heavy burden on waste treatment facilities and inflating the overall cost reduction in textile chemical manufacturing.
The Novel Approach
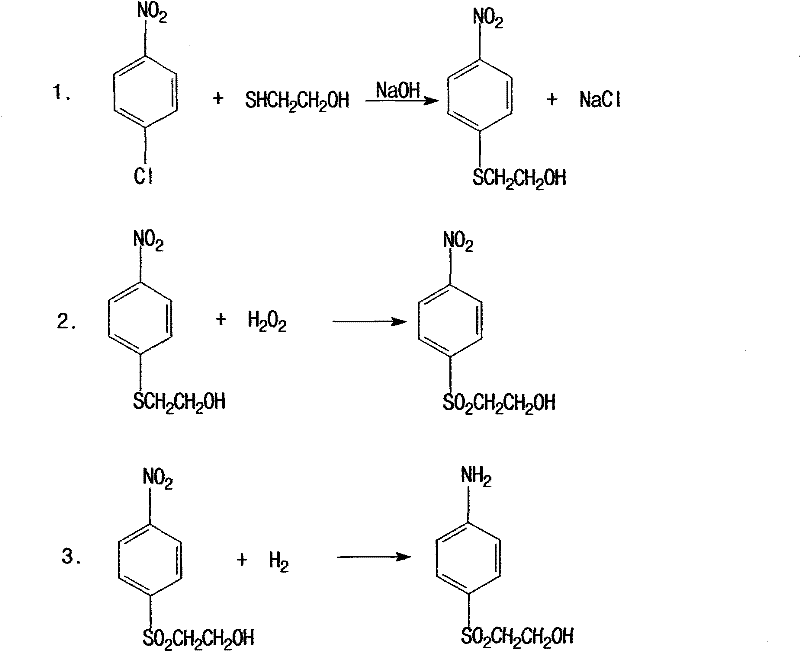
In stark contrast, the novel approach detailed in the patent utilizes a direct nucleophilic substitution between p-nitrochlorobenzene and mercaptoethanol in a dimethylformamide (DMF) solvent system. This strategy eliminates the need for hazardous chlorosulfonic acid handling and the associated cryogenic cooling requirements, allowing reactions to proceed at moderate temperatures between 50-80°C. The process flow is significantly simplified, replacing the multi-stage sulfonation and hydrolysis with a single substitution step followed by a highly selective catalytic oxidation. By avoiding the generation of large volumes of inorganic salts and spent acids inherent in the old route, this method achieves a dramatic reduction in effluent volume, lowering wastewater output to just 1-2 tons per ton of product. This streamlined workflow not only enhances operator safety but also improves the commercial scale-up of complex dye intermediates by reducing the footprint and complexity of the required reactor infrastructure.
Mechanistic Insights into Tungstate-Catalyzed Oxidation and Hydrogenation
The core chemical innovation lies in the selective oxidation of the thioether intermediate to the corresponding sulfone using hydrogen peroxide catalyzed by sodium tungstate. In this mechanism, the tungstate species activates the hydrogen peroxide, facilitating the transfer of oxygen atoms to the sulfur center while minimizing over-oxidation or degradation of the sensitive hydroxyethyl side chain. The reaction is conducted at 95-100°C with a controlled addition of 30-35% hydrogen peroxide over 2-5 hours, ensuring high conversion rates and excellent selectivity. Following oxidation, the nitro group is reduced to an amino group using a palladium carbon catalyst under hydrogen pressure of 1.5-2.5MPa. This heterogeneous catalysis allows for easy separation of the catalyst via pressure filtration, enabling its reuse and preventing metal contamination in the final product. The precise control of reaction parameters, such as maintaining the mass ratio of oxidizing material to water at 1:1.3-1.7, is critical for maximizing crystal purity and yield during the subsequent crystallization steps.
Impurity control is rigorously managed through the physical properties of the intermediates and the specificity of the catalysts employed. The initial substitution reaction generates sodium chloride as a solid by-product, which is removed by filtration at temperatures below 40°C before the distillation of the solvent. This early removal of inorganic salts prevents them from carrying over into the oxidation and hydrogenation stages, where they could interfere with catalyst activity or contaminate the final crystal lattice. Furthermore, the vacuum distillation step effectively removes DMF and moisture, yielding a high-purity p-nitrophenyl-beta-hydroxyethyl thioether that serves as a clean substrate for oxidation. The combination of these purification checkpoints ensures that the final aminophenyl beta-hydroxyethyl sulfone sulfate meets stringent purity specifications required for high-quality reactive dye manufacturing, minimizing the presence of color-forming impurities.
How to Synthesize p-Amino Phenyl-Beta-Hydroxyethyl Sulfone Sulfate Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing this valuable intermediate with high efficiency. The process begins with the preparation of the thioether backbone, followed by a carefully controlled oxidation to install the sulfone functionality, which is essential for the reactivity of the final dye molecule. Subsequent hydrogenation reduces the nitro group to the amine without affecting the sulfone or hydroxyl groups, demonstrating the chemoselectivity of the chosen catalyst system. The final esterification with concentrated sulfuric acid converts the hydroxyl group into the sulfate ester, completing the molecular architecture. For process engineers looking to implement this technology, the detailed operating conditions regarding temperature, pressure, and molar ratios provided in the patent serve as a critical foundation for optimizing batch cycles and maximizing throughput in a commercial setting.
- React p-Nitrophenyl chloride with mercaptoethanol in DMF using sodium hydroxide at 50-80°C to form p-nitrophenyl-beta-hydroxyethyl thioether.
- Oxidize the thioether intermediate using hydrogen peroxide and a sodium tungstate catalyst at 95-100°C to obtain the sulfone crystallization material.
- Perform catalytic hydrogenation on the oxidation product using a palladium carbon catalyst at 1.5-2.5MPa pressure to reduce the nitro group.
- Complete the synthesis by esterifying the hydrogenation product with concentrated sulfuric acid at 100-130°C to yield the final sulfate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this novel synthesis route offers profound advantages in terms of cost structure and supply reliability. The elimination of chlorosulfonic acid, a reagent that requires specialized storage and handling due to its corrosive nature and tendency to release toxic fumes, significantly lowers the barrier to entry for production and reduces insurance and safety compliance costs. Moreover, the drastic reduction in wastewater volume translates directly into lower operational expenditures for effluent treatment, a major cost driver in the fine chemical industry. By simplifying the process flow and removing the need for complex acid recovery towers and cryogenic cooling systems, manufacturers can achieve substantial cost savings in both capital investment and daily utility consumption. This efficiency gain allows suppliers to offer more competitive pricing structures while maintaining healthy margins, a key factor for procurement managers negotiating long-term contracts.
- Cost Reduction in Manufacturing: The new process fundamentally alters the cost equation by replacing expensive and hazardous reagents with more benign alternatives like hydrogen peroxide and sodium hydroxide. The removal of the acetanilide protection-deprotection strategy simplifies the material balance, reducing the total mass of raw materials required per unit of output. Additionally, the ability to recover and reuse the DMF solvent and the palladium catalyst further drives down variable costs. These cumulative efficiencies mean that the cost reduction in textile chemical manufacturing is not merely theoretical but is rooted in tangible decreases in raw material intensity and waste disposal fees, providing a robust buffer against market volatility in commodity chemical prices.
- Enhanced Supply Chain Reliability: The reliance on p-nitrochlorobenzene and mercaptoethanol as starting materials leverages a supply chain that is generally more stable and less prone to the regulatory restrictions often placed on chlorosulfonic acid and ethylene oxide. The simplified unit operations reduce the likelihood of unplanned shutdowns caused by equipment failure in complex absorption or cooling systems. Consequently, this leads to reducing lead time for high-purity dye intermediates, as production cycles are shorter and less susceptible to bottlenecks. For supply chain heads, this translates to a more predictable delivery schedule and the ability to respond rapidly to fluctuations in downstream demand from the textile sector without compromising on quality or safety standards.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily manageable in standard stainless steel reactors rather than requiring specialized glass-lined or Hastelloy vessels needed for strong acid service. The significant decrease in three wastes (waste water, waste gas, and solid waste) aligns perfectly with increasingly stringent global environmental regulations, future-proofing the production facility against tighter emission limits. This environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable partner. The ability to scale from pilot batches to 100 MT annual production with minimal process modification ensures that the technology remains viable and compliant as production volumes grow to meet global market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios or for procurement specialists assessing the long-term viability of their supply sources.
Q: How does the new catalytic oxidation method improve yield compared to traditional sulfonation?
A: The novel method utilizing tungstate-catalyzed oxidation achieves a product yield of approximately 85%, significantly surpassing the roughly 70% yield of the conventional acetanilide sulfonation route, while simultaneously reducing raw material consumption.
Q: What are the environmental benefits regarding wastewater generation in this process?
A: This optimized production technique drastically reduces wastewater generation from 6-8 tons per ton of product in the old process to merely 1-2 tons, minimizing the burden of high-salt wastewater treatment and spent acid disposal.
Q: Is the palladium carbon catalyst recoverable in the hydrogenation step?
A: Yes, the process design includes a specific separation step where the palladium carbon catalyst is reclaimed through pressure filtration prior to cooling crystallization, ensuring efficient catalyst recycling and cost management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminophenyl Beta-Hydroxyethyl Sulfone Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of final textile products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify every batch. Our capability to implement the catalytic oxidation and hydrogenation processes described in recent patents allows us to offer a superior product profile with lower impurity levels and consistent batch-to-batch reproducibility, making us a trusted partner for global dye manufacturers.
We invite you to collaborate with us to optimize your supply chain for reactive dye precursors. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our efficient manufacturing processes can lower your total cost of ownership. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project goals and quality standards.
