Revolutionizing Ortho-Nitrosation: A Mild Silver-Catalyzed Route for High-Purity Pharmaceutical Intermediates
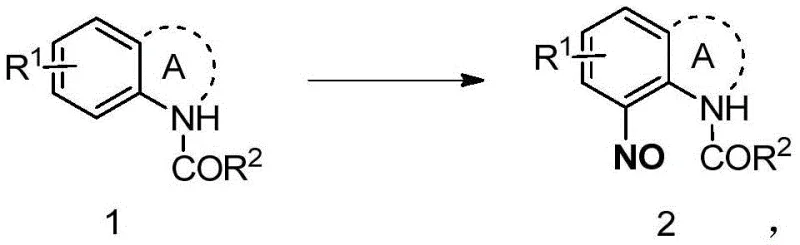
The landscape of fine chemical synthesis is constantly evolving, driven by the need for safer, more selective, and environmentally benign methodologies. A significant advancement in this domain is documented in patent CN113620763A, which discloses a novel synthetic method for acyl-protected ortho-nitrosated arylamine derivatives. This technology addresses a critical bottleneck in the production of aromatic nitroso compounds, which serve as versatile building blocks for various organic transformations including nitrosoaldol reactions, cycloadditions, and the synthesis of indoles and azo dyes. The core innovation lies in the utilization of silver nitrate as a catalyst in conjunction with NOBF4 as the nitrosating agent, enabling the reaction to proceed efficiently under inert atmosphere at room temperature. This approach eliminates the reliance on corrosive strong acids and aggressive oxidants typically associated with traditional nitrosation protocols, thereby offering a pathway to high-purity intermediates with superior functional group tolerance.
For procurement managers and supply chain directors, the implications of this technological shift are profound. By moving away from hazardous reagents, the operational risks associated with storage and handling are drastically minimized, leading to potential reductions in insurance and safety compliance costs. Furthermore, the mild reaction conditions suggest a lower energy footprint compared to high-temperature processes, aligning with modern sustainability goals in chemical manufacturing. As a reliable pharmaceutical intermediate supplier, understanding these underlying process advantages is crucial for evaluating the long-term viability and cost-effectiveness of sourcing such specialized compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic nitroso compounds has been plagued by significant technical challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Conventional strategies often rely on the direct oxidation of anilines or the nitrosation of simple aromatic hydrocarbons using strong mineral acids and powerful oxidizing agents. These harsh conditions frequently result in poor reaction selectivity, where the nitroso group may attach to undesired positions on the aromatic ring, or worse, lead to over-oxidation and decomposition of sensitive functional groups. Additionally, the use of corrosive acids necessitates specialized reactor materials and extensive waste treatment protocols, driving up the capital expenditure and operational costs for manufacturers. The intolerance of these methods towards diverse substituents limits their applicability, forcing chemists to employ lengthy protection-deprotection sequences that erode overall yield and extend production timelines.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a paradigm shift by employing a silver-catalyzed electrophilic nitrosation strategy. This novel approach utilizes NOBF4 as a mild yet effective nitrosating source, activated by a catalytic amount of silver nitrate. The reaction proceeds smoothly at room temperature, circumventing the thermal stress that often degrades complex molecular architectures. Crucially, this method demonstrates exceptional regioselectivity, predominantly yielding the ortho-nitrosated product rather than the thermodynamically favored para-isomer often seen in uncatalyzed reactions. This high degree of control simplifies downstream purification, as the formation of isomeric byproducts is suppressed. For industries seeking cost reduction in fine chemical manufacturing, this translates to higher throughput and reduced solvent consumption during purification, making it a commercially attractive alternative to legacy processes.
Mechanistic Insights into Silver-Catalyzed Electrophilic Nitrosation
To fully appreciate the robustness of this synthesis, one must delve into the mechanistic nuances that govern the transformation. The silver cation acts as a Lewis acid, likely coordinating with the nitrosating agent or the substrate to enhance the electrophilicity of the nitrosonium species. This activation lowers the energy barrier for the electrophilic aromatic substitution, allowing the reaction to occur under ambient conditions. The acyl-protecting group on the nitrogen atom plays a dual role; it not only prevents the oxidation of the amine nitrogen itself but also directs the incoming electrophile to the ortho-position through electronic and steric influences. The coordination between the silver catalyst and the carbonyl oxygen of the acyl group may further rigidify the transition state, enforcing the observed ortho-selectivity. This precise control over the reaction trajectory is vital for R&D directors who require consistent impurity profiles for regulatory filings.
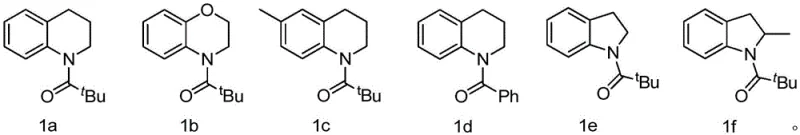
The scope of this reaction is remarkably broad, accommodating various heterocyclic systems which are prevalent in medicinal chemistry. As illustrated in the substrate scope, the method is effective for fused bicyclic systems such as tetrahydroquinolines and benzomorpholines, as well as five-membered rings like indolines. The presence of electron-donating or withdrawing groups on the aromatic ring does not significantly impede the reaction, demonstrating the versatility of the catalytic system. For instance, substrates bearing methyl groups or bulky tert-butyl protecting groups undergo smooth conversion to the corresponding nitroso derivatives. This tolerance suggests that the catalytic cycle is robust against steric hindrance and electronic variation, a key attribute for a platform technology intended for diverse API intermediate synthesis.
How to Synthesize N-pivaloyl-8-nitroso-tetrahydroquinoline Efficiently
The practical implementation of this chemistry is straightforward, requiring standard laboratory equipment and readily available reagents. The process begins with the preparation of the reaction vessel under an inert atmosphere to prevent moisture interference, followed by the dissolution of the substrate and catalyst in acetonitrile. The controlled addition of the nitrosating reagent ensures that the exotherm is managed effectively, maintaining the integrity of the product. Detailed standardized synthesis steps are provided below to guide process chemists in replicating these results.
- Prepare the reaction mixture by combining the acyl-protected arylamine substrate and 20 mol% silver nitrate catalyst in anhydrous acetonitrile under an inert atmosphere.
- Slowly add the nitrosating reagent NOBF4 (2.0 equivalents) dissolved in acetonitrile to the reaction flask at room temperature over approximately 10 minutes.
- Stir the reaction at room temperature for 1 hour, then quench with saturated sodium bicarbonate, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this silver-catalyzed nitrosation method offers tangible benefits that extend beyond mere chemical yield. For procurement teams, the shift to milder reagents implies a reduction in the complexity of the supply chain, as there is no longer a dependency on highly regulated strong oxidants or corrosive acids that require special transport and storage licenses. This simplification can lead to substantial cost savings in logistics and inventory management. Moreover, the high selectivity of the reaction minimizes the generation of difficult-to-separate isomers, which reduces the volume of solvents and silica gel required for purification, directly impacting the cost of goods sold.
- Cost Reduction in Manufacturing: The elimination of strong acids and high-temperature requirements significantly lowers the energy consumption and equipment maintenance costs associated with the production process. By avoiding the need for expensive corrosion-resistant reactors and complex waste neutralization systems, manufacturers can achieve a leaner operational model. The high efficiency of the catalyst means that only small amounts of silver are needed, and the potential for catalyst recovery further enhances the economic viability of the process on a multi-ton scale.
- Enhanced Supply Chain Reliability: The use of stable and commercially available reagents like silver nitrate and NOBF4 ensures a consistent supply of raw materials, mitigating the risk of production delays caused by reagent shortages. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up from pilot plant to commercial production without the need for extensive re-optimization. This reliability is critical for maintaining continuous supply lines to downstream pharmaceutical customers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: Operating at room temperature and avoiding hazardous oxidants aligns perfectly with green chemistry principles, facilitating easier regulatory approval and environmental compliance. The simplified workup procedure, involving basic aqueous quenching and extraction, generates less hazardous waste compared to traditional acidic workups. This environmental friendliness not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for sustainability-conscious global corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrosation technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: Why is silver nitrate preferred over traditional strong acid catalysts for this nitrosation?
A: Traditional methods often require harsh strong acids and oxidants which lead to poor functional group tolerance and safety hazards. The silver nitrate catalyzed method described in patent CN113620763A operates at room temperature without strong acids, significantly improving safety and selectivity for sensitive substrates.
Q: What is the regioselectivity of this nitrosation reaction?
A: The method exhibits excellent ortho-selectivity. Unlike conventional electrophilic substitutions that often favor the para-position or result in mixtures, this silver-catalyzed protocol specifically targets the ortho-position relative to the acyl-protected amine, yielding pure ortho-nitrosated derivatives essential for downstream cyclization reactions.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable. It utilizes common solvents like acetonitrile, operates at ambient temperature reducing energy costs, and avoids hazardous reagents. The simple workup procedure involving aqueous quenching and extraction makes it ideal for large-scale manufacturing of complex heterocyclic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acyl-Protected Ortho-Nitrosated Arylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the silver-catalyzed nitrosation described in CN113620763A for the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this innovative chemistry for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for complex fine chemical synthesis.
