Advanced Synthesis of Benzenesulfonamide Hydroxyl Derivatives for High-Purity PET Imaging Probes
Advanced Synthesis of Benzenesulfonamide Hydroxyl Derivatives for High-Purity PET Imaging Probes
The landscape of oncological diagnostics is rapidly evolving, driven by the critical need for non-invasive technologies capable of detecting tumor hypoxia with high precision. Patent CN101585816A introduces a groundbreaking class of benzenesulfonamide hydroxyl derivatives that function as potent Carbonic Anhydrase IX (CAIX) inhibitors, addressing the urgent demand for reliable molecular imaging agents. These compounds exhibit superior hydrophilicity and reduced non-specific absorption in the digestive system, making them ideal candidates for Positron Emission Tomography (PET) probes. The disclosed technology not only provides the final active pharmaceutical ingredient but also details a robust synthetic pathway for key intermediates, ensuring that reliable pharmaceutical intermediate suppliers can meet the rigorous purity standards required for clinical imaging applications. By leveraging this intellectual property, manufacturers can access a streamlined production method that avoids the pitfalls of traditional high-energy synthesis.
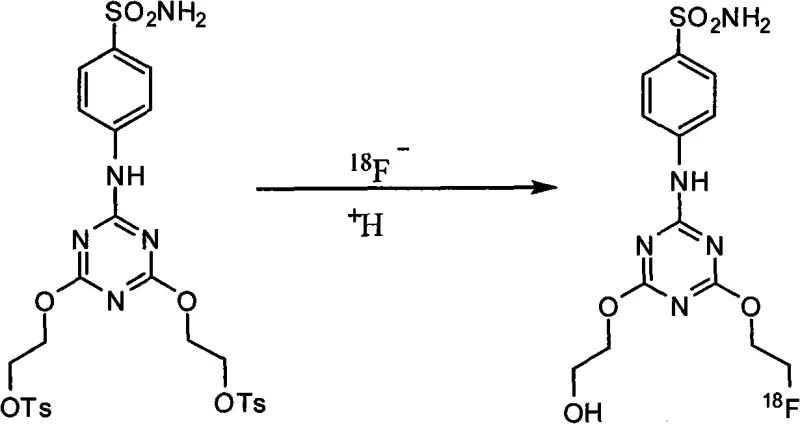
Tumor hypoxia remains a formidable barrier in cancer therapy, often leading to resistance against radiotherapy and chemotherapy. The ability to map hypoxic regions in vivo using CAIX-targeted PET probes offers clinicians a powerful tool for treatment planning. The compound described in this patent, specifically the 18F-labeled variant, represents a significant leap forward in diagnostic capability. Unlike earlier generations of sulfonamides which suffered from poor solubility or metabolic instability, this new derivative maintains strong enzyme inhibition activity while optimizing pharmacokinetic properties. For procurement teams and R&D directors alike, understanding the synthesis of this molecule is crucial, as it dictates the feasibility of large-scale production and the ultimate cost-effectiveness of the diagnostic agent in a competitive healthcare market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonamide-based CAIX inhibitors has been plagued by complex multi-step sequences that require harsh reaction conditions and expensive protecting group strategies. Conventional routes often involve the use of sensitive reagents that necessitate strict anhydrous environments and cryogenic temperatures, driving up operational costs and complicating waste management protocols. Furthermore, traditional methods frequently struggle with regioselectivity on the triazine ring, leading to difficult-to-separate isomeric impurities that compromise the specific activity of the final radiotracer. These inefficiencies create bottlenecks in the supply chain, resulting in extended lead times and inconsistent batch quality that can delay critical clinical trials. The reliance on high-pressure hydrogenation or transition metal catalysts in older methodologies also introduces the risk of heavy metal contamination, requiring additional purification steps that further erode overall yield and profitability.
The Novel Approach
In stark contrast, the methodology outlined in patent CN101585816A presents a remarkably elegant and efficient synthetic strategy that circumvents these historical challenges. The novel approach utilizes a stepwise nucleophilic substitution on a cyanuric chloride core, allowing for precise control over the functionalization of the triazine ring under mild alkaline conditions. As illustrated in the comprehensive reaction scheme below, the process begins with the condensation of sulfanilamide and cyanuric chloride, followed by sequential substitutions with ethylene glycol and 2-fluoroethanol. This route eliminates the need for exotic catalysts and operates at atmospheric pressure, significantly simplifying the engineering requirements for commercial manufacturing. The use of inexpensive, commodity-grade starting materials ensures that cost reduction in pharmaceutical intermediate manufacturing is achievable without sacrificing the high purity necessary for medical applications.
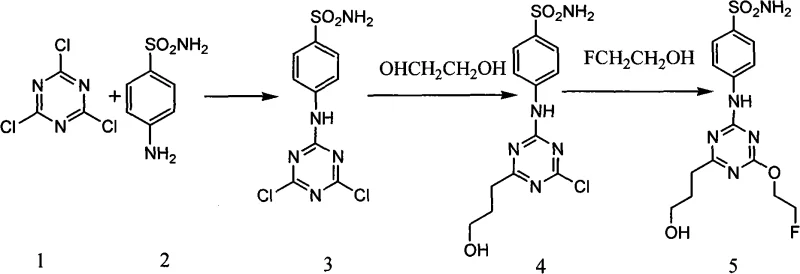
The strategic advantage of this novel approach lies in its modularity and scalability. By decoupling the introduction of the hydrophilic hydroxyethyl group and the fluoroethoxy moiety into distinct steps, chemists can optimize reaction parameters for each transformation independently. For instance, the substitution with ethylene glycol is conducted at a moderate 30-50°C, preventing thermal degradation while ensuring complete conversion. This level of control minimizes the formation of side products, thereby reducing the burden on downstream purification processes such as chromatography. For supply chain managers, this translates to a more predictable production schedule and a reduced risk of batch failure. The simplicity of the workup procedures, often involving mere filtration and washing, further enhances the throughput potential, making this technology highly attractive for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nucleophilic Aromatic Substitution on Triazine
The core chemical transformation driving this synthesis is the nucleophilic aromatic substitution (SnAr) on the electron-deficient 1,3,5-triazine ring. The mechanism relies on the differential reactivity of the chlorine atoms on the cyanuric chloride scaffold, which can be selectively displaced based on temperature and the nucleophilicity of the incoming reagent. In the first step, the amino group of sulfanilamide attacks one of the chlorines at low temperature (0°C), forming a stable C-N bond while leaving the other two chlorines intact for subsequent modification. This selectivity is governed by the inductive withdrawal of the nitrogen lone pair into the ring, which deactivates the remaining positions slightly but keeps them susceptible to stronger nucleophiles or higher temperatures. Understanding this electronic nuance is vital for R&D teams aiming to replicate the process, as deviations in temperature can lead to di-substitution at the wrong positions or hydrolysis of the chloride groups by trace moisture.
Impurity control is intrinsically built into the mechanistic design of this pathway. The sequential addition of reagents ensures that the hydroxyethyl group is installed before the fluoroethoxy group, a sequence dictated by the relative nucleophilicity and steric bulk of ethylene glycol versus 2-fluoroethanol. The patent specifies the use of inorganic bases like sodium hydroxide or potassium hydroxide to deprotonate the alcohol nucleophiles in situ, generating the reactive alkoxide species necessary for the displacement of the chloride leaving groups. By maintaining the reaction pH within a specific range and monitoring progress via TLC, operators can prevent the formation of bis-substituted byproducts where both remaining chlorines are replaced by the same alcohol. This rigorous control over the reaction trajectory ensures that the final product possesses the exact structural configuration required for high-affinity binding to the CAIX enzyme, which is critical for the sensitivity of the PET imaging probe.
How to Synthesize Benzenesulfonamide Hydroxyl Derivative Efficiently
The practical execution of this synthesis requires careful attention to stoichiometry and thermal management to maximize yield and minimize impurity profiles. The process is divided into three distinct stages, beginning with the formation of the dichloro-intermediate, followed by the introduction of the hydrophilic tail, and concluding with the installation of the fluorine-containing moiety. Each stage has been optimized in the patent examples to demonstrate reproducibility across different scales, providing a clear roadmap for process chemists. The following guide summarizes the critical operational parameters derived from the experimental data, serving as a foundational reference for establishing standard operating procedures in a GMP environment. For detailed standardized synthesis steps, please refer to the guide below.
- React cyanuric chloride with sulfanilamide in acetone under ice-water bath conditions with sodium hydroxide to form the dichloro-intermediate.
- Perform nucleophilic substitution on the dichloro-intermediate using ethylene glycol and inorganic base at 30-50°C to introduce the hydroxyethyl group.
- Complete the synthesis by reacting the mono-substituted intermediate with 2-fluoroethanol and base at 80-100°C to yield the final hydroxyl derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic benefits for organizations looking to secure a stable supply of high-value imaging agents. The primary advantage stems from the drastic simplification of the raw material portfolio; by relying on bulk chemicals like cyanuric chloride and ethylene glycol, manufacturers can insulate themselves from the volatility associated with specialized fine chemical markets. This accessibility directly contributes to cost reduction in pharmaceutical intermediate manufacturing, as the elimination of proprietary catalysts and complex protecting groups lowers the bill of materials significantly. Furthermore, the ambient pressure conditions reduce the capital expenditure required for reactor infrastructure, allowing existing facilities to be repurposed for this production without major retrofitting investments.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the high atom economy of the substitution reactions and the avoidance of expensive transition metal catalysts. Traditional cross-coupling methods often require palladium or copper catalysts which are not only costly but also require rigorous removal to meet regulatory limits for residual metals. By utilizing a metal-free SnAr pathway, this method eliminates the need for scavenger resins or extensive charcoal treatments, thereby reducing both material costs and processing time. Additionally, the solvents used, such as acetone and ethylene glycol, are inexpensive and easily recoverable, further enhancing the overall process efficiency and margin potential for large-scale producers.
- Enhanced Supply Chain Reliability: Supply chain resilience is a critical factor for pharmaceutical intermediates, especially those destined for time-sensitive diagnostic applications. The starting materials for this synthesis are commodity chemicals produced globally in massive quantities, ensuring that supply disruptions are highly unlikely. This abundance allows procurement managers to negotiate favorable long-term contracts and maintain safety stock levels without tying up excessive working capital. Moreover, the robustness of the reaction conditions means that the synthesis is less susceptible to minor variations in utility quality, such as cooling water temperature fluctuations, ensuring consistent output even in diverse manufacturing locations. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates and meeting the just-in-time delivery requirements of radiopharmacies.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant often reveals hidden hazards, but this route is inherently safe due to the absence of exothermic runaway risks associated with high-energy reagents. The moderate temperatures (30-100°C) and atmospheric pressure operations simplify the engineering controls needed for heat dissipation and containment. From an environmental standpoint, the aqueous workups and lack of halogenated solvents in the main reaction steps align well with modern green chemistry principles, facilitating easier waste stream treatment and regulatory compliance. This environmental compatibility reduces the overhead costs associated with hazardous waste disposal and permits, making the process sustainable for long-term commercial operation and attractive to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzenesulfonamide derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. These insights are intended to clarify the operational feasibility and strategic value of implementing this technology within your existing manufacturing framework.
Q: What are the critical temperature controls for the triazine substitution steps?
A: The synthesis requires precise temperature staging: the initial condensation occurs at 0°C to prevent polysubstitution, the ethylene glycol substitution proceeds at 30-50°C, and the final fluorination requires 80-100°C.
Q: How does this method improve supply chain reliability for PET probe precursors?
A: By utilizing commercially available starting materials like cyanuric chloride and sulfanilamide without requiring high-pressure equipment, the process ensures consistent batch-to-batch availability and reduces dependency on exotic reagents.
Q: Is the final compound suitable for 18F radiolabeling?
A: Yes, the hydroxyl derivative serves as a reference standard, while the tosylate precursor derived from this route is specifically designed for efficient nucleophilic substitution with 18F- ions for PET imaging applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzenesulfonamide Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented technology in advancing oncological diagnostics. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate these laboratory-scale successes into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch. Whether you require the cold reference standard for assay validation or the GMP-grade intermediate for radiolabeling, our commitment to quality assurance guarantees that your projects proceed without regulatory hindrance.
We invite you to collaborate with us to unlock the full commercial potential of this CAIX inhibitor platform. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By partnering with us, you gain access to deep process knowledge that can further optimize yields and reduce time-to-market. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can serve as a cornerstone for your supply chain strategy in the competitive field of molecular imaging.
