Advanced Synthesis of 8-Iodoimidazo[1,2-a]pyridine Derivatives for Scalable PET and SPECT Imaging Applications
Advanced Synthesis of 8-Iodoimidazo[1,2-a]pyridine Derivatives for Scalable PET and SPECT Imaging Applications
The pharmaceutical landscape for diagnostic imaging is undergoing a significant transformation driven by the need for more accessible and stable radiotracers. Patent CN100418966C introduces a groundbreaking class of 8-iodoimidazo[1,2-a]pyridine-3-acetamide derivatives designed specifically to overcome the severe logistical bottlenecks inherent in traditional short-lived Carbon-11 radiotracers. While Carbon-11 labeled ligands have demonstrated efficacy in targeting Peripheral Benzodiazepine Receptors (PBR) for tumor and neurodegenerative disease diagnosis, their ultra-short half-life of merely 20.4 minutes restricts their utility to facilities with immediate access to cyclotrons. This patented technology substitutes the transient carbon label with various iodine isotopes, including I-123, I-124, I-125, I-130, and I-131, which offer half-lives ranging from 13 hours to 60 days. This fundamental shift in radionuclide selection not only expands the clinical window for imaging but also revolutionizes the supply chain dynamics for radiopharmaceutical intermediates.
![General chemical structure of 8-iodoimidazo[1,2-a]pyridine-3-acetamide showing the core scaffold and variable Ar group](/insights/img/8-iodoimidazo-pyridine-synthesis-radiopharma-supplier-20260314013656-01.png)
The core innovation lies in the molecular architecture which maintains high affinity for PBR while enabling robust radiochemistry. The structural formula presented in the patent highlights the critical 8-position iodination on the imidazopyridine scaffold, a modification that preserves the biological activity necessary for effective in vivo imaging. By decoupling the production of the tracer from the immediate presence of a particle accelerator, this technology opens new markets for reliable radiopharmaceutical intermediate suppliers who can manufacture precursors centrally and distribute them regionally. For R&D directors and procurement managers in the nuclear medicine sector, this represents a pivotal opportunity to enhance diagnostic capabilities without the prohibitive infrastructure costs associated with short-lived isotope production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the gold standard for PBR imaging has relied heavily on Carbon-11 labeled compounds, such as N-methylaryl amides. However, the reliance on C-11 imposes draconian constraints on the entire value chain of radiopharmaceutical manufacturing. The 20.4-minute half-life means that synthesis, purification, quality control, and administration must all occur within a narrow timeframe, often leading to significant material loss and wasted resources if logistical delays occur. Furthermore, the requirement for an on-site cyclotron limits the deployment of these diagnostic tools to major academic medical centers, excluding a vast majority of community hospitals and regional clinics. From a chemical synthesis perspective, the rapid decay necessitates automated modules that are expensive to maintain and operate, driving up the cost per dose significantly. The inability to stockpile these tracers also creates vulnerability in the supply chain, where any equipment failure results in an immediate cessation of diagnostic services.
The Novel Approach
The methodology outlined in CN100418966C offers a transformative solution by leveraging the favorable nuclear properties of iodine isotopes. Instead of struggling against the clock with C-11, this approach utilizes iododestannylation to introduce isotopes like I-124 (4.2 days) or I-123 (13 hours), which provide ample time for complex synthesis and distribution. The novel route involves a three-step sequence starting from readily available bromo-keto amides, proceeding through a palladium-catalyzed stannylation, and culminating in an electrophilic iodination. This pathway avoids the need for high-temperature or high-pressure reactors, operating instead under mild conditions that preserve the integrity of sensitive functional groups. For procurement teams, this translates to cost reduction in PET tracer manufacturing by simplifying the production workflow and reducing the dependency on specialized, high-maintenance cyclotron infrastructure. The result is a more resilient supply chain capable of supporting widespread clinical adoption of PBR imaging.
Mechanistic Insights into Palladium-Catalyzed Stannylation and Iododestannylation
The chemical elegance of this synthesis lies in its modular design, which separates the construction of the heterocyclic core from the introduction of the radioactive label. The first step involves the condensation of p-chlorophenyl-3-bromo-4-keto amide with 5-chloro-3-bromo-2-amino pyridine. This reaction, typically conducted in polar aprotic solvents like DMF or NMP at temperatures between 20°C and 180°C, forms the stable 8-bromoimidazo[1,2-a]pyridine scaffold. This intermediate serves as a versatile platform, allowing for the installation of various aryl substituents at the amide nitrogen to tune pharmacokinetic properties. The robustness of this condensation ensures high yields and purity, establishing a solid foundation for the subsequent radiolabeling steps. The ability to isolate and characterize this non-radioactive intermediate is crucial for quality control, ensuring that the final radiopharmaceutical meets stringent regulatory standards before the short-lived isotope is even introduced.
The critical radiolabeling event occurs via a two-stage metal-mediated process. First, the 8-bromo intermediate undergoes a palladium-catalyzed cross-coupling with hexabutylditin or similar alkyltin reagents to generate the 8-tributyltin precursor. This stannylation step is highly efficient, utilizing catalysts such as Pd(PPh3)4 to facilitate the exchange of the bromine atom for the tin moiety. Subsequently, the radioactive iodine is introduced through an electrophilic iododestannylation reaction. In the presence of mild oxidizing agents like chloramine-T or peracetic acid, the carbon-tin bond is cleaved and replaced by a carbon-iodine bond. This mechanism is particularly advantageous because it proceeds rapidly at room temperature, minimizing the exposure of the radioactive isotope to harsh conditions that could promote radiolysis. The specificity of this reaction ensures that the radioiodine is incorporated exclusively at the desired 8-position, resulting in the high radiochemical purity observed in the patent examples.
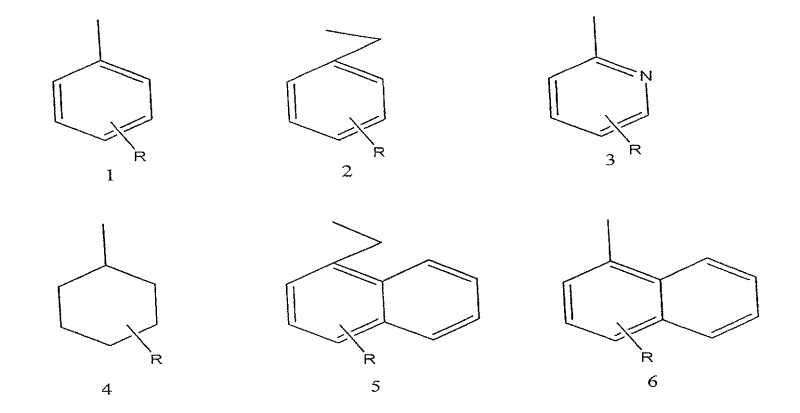
Furthermore, the versatility of this synthetic route is underscored by the diversity of substituents that can be accommodated on the amide nitrogen. As illustrated in the structural variations, the 'Ar' group can range from simple phenyl rings to more complex naphthyl or cyclohexyl moieties. This structural flexibility allows medicinal chemists to optimize the lipophilicity and receptor binding affinity of the final tracer without altering the core radiolabeling strategy. For R&D directors, this means that a single manufacturing platform can support the development of a library of analogues, accelerating the discovery of next-generation imaging agents. The compatibility of the synthesis with various functional groups ensures that the process remains robust even as the molecular complexity increases, providing a scalable pathway for commercial production.
How to Synthesize 8-Iodoimidazo[1,2-a]pyridine Efficiently
The synthesis of these high-value intermediates follows a logical progression designed to maximize yield while minimizing impurities. The process begins with the formation of the imidazopyridine core, followed by the installation of the tin handle, and concludes with the radioactive swap. This standardized approach allows for the decoupling of the cold chemistry from the hot chemistry, enabling the bulk of the work to be done in standard organic synthesis labs. Detailed standard operating procedures for each step, including solvent choices, catalyst loading, and purification methods, are essential for reproducibility. The following guide outlines the critical operational parameters derived from the patent data to ensure successful execution of this pathway.
- Condense p-chlorophenyl-3-bromo-4-keto amide with 5-chloro-3-bromo-2-amino pyridine in an inert solvent to form the 8-bromo intermediate.
- Perform palladium-catalyzed stannylation on the 8-bromo intermediate using alkyltin reagents to generate the 8-tributyltin precursor.
- Execute electrophilic iododestannylation using radioiodine isotopes and an oxidizing agent to yield the final 8-iodo labeled product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, the shift to iodine-based PBR ligands offers profound strategic benefits. The primary advantage is the drastic simplification of the logistics network. Unlike C-11 tracers that must be produced and consumed within the same building, iodine-labeled compounds can be manufactured at a central facility and shipped to multiple clinical sites. This consolidation of production volume allows for economies of scale that are impossible to achieve with decentralized cyclotron models. Additionally, the extended half-life reduces the pressure on scheduling, allowing for more flexible patient appointment times and reducing the rate of missed doses due to production delays. These factors collectively contribute to a more stable and predictable supply chain.
- Cost Reduction in Manufacturing: The elimination of the need for on-site cyclotrons represents a massive capital expenditure saving for end-users, which indirectly boosts demand for the intermediates. From a manufacturing perspective, the use of standard organic solvents and commercially available palladium catalysts keeps raw material costs low. The mild reaction conditions reduce energy consumption and equipment wear, further driving down operational expenses. Moreover, the high radiochemical yields reported in the patent minimize the amount of expensive radioactive starting material required per batch, optimizing the cost-efficiency of every production run.
- Enhanced Supply Chain Reliability: The ability to produce the non-radioactive tin precursor in large batches and store it for extended periods creates a strategic buffer against supply disruptions. This "cold kit" approach ensures that the critical bottleneck is only the final labeling step, which is rapid and efficient. For supply chain heads, this means reducing lead time for high-purity radiopharmaceutical intermediates and ensuring continuous availability even during maintenance cycles of isotope production facilities. The robustness of the chemical synthesis also reduces the risk of batch failures, guaranteeing a consistent flow of materials to the market.
- Scalability and Environmental Compliance: The synthesis route avoids the use of exotic reagents or extreme conditions, making it inherently easier to scale from gram to kilogram quantities. The waste streams generated are typical of standard organic synthesis and can be managed using established protocols, simplifying environmental compliance. The high purity of the final product reduces the burden on downstream purification processes, lowering the overall solvent usage and waste generation. This alignment with green chemistry principles not only meets regulatory standards but also enhances the corporate sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this technology is vital for making informed procurement and development decisions. The following questions address common inquiries regarding the stability, application, and regulatory aspects of these iodine-labeled compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What is the primary advantage of using Iodine-123/124 over Carbon-11 for PBR imaging?
A: Iodine isotopes such as I-123 and I-124 possess significantly longer half-lives ranging from hours to days, compared to the mere 20.4 minutes of Carbon-11. This extended stability allows for centralized production and wider distribution to clinical sites lacking on-site cyclotrons.
Q: How does the iododestannylation method improve radiochemical purity?
A: The electrophilic substitution of the tributyltin group with radioiodine under mild oxidative conditions minimizes side reactions and decomposition. Patent data indicates this method achieves radiochemical purity greater than 98% with high specific activity.
Q: Is this synthesis route scalable for commercial GMP production?
A: Yes, the process utilizes standard organic solvents and avoids extreme high-pressure or high-temperature conditions. The robustness of the palladium-catalyzed steps and simple purification protocols make it highly suitable for scale-up from gram to kilogram levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Iodoimidazo[1,2-a]pyridine-3-acetamide Supplier
As the demand for advanced diagnostic imaging agents continues to grow, partnering with an experienced CDMO is essential for navigating the complexities of radiopharmaceutical production. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready product is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of organometallic chemistry and radioisotope handling, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality intermediates. We understand the critical nature of supply continuity in the healthcare sector and are committed to being a dependable partner in your success.
We invite you to leverage our technical expertise to optimize your supply chain and reduce costs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and logistical needs. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By collaborating with us, you gain access to a wealth of process knowledge that can accelerate your time-to-market and enhance the competitiveness of your diagnostic portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
