Revolutionizing Chiral Beta-Amino Alcohol Production with Advanced Biocatalytic Technology
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access high-value chiral building blocks. Patent CN114836486B introduces a groundbreaking enzymatic method for synthesizing chiral beta-amino alcohols, a critical structural motif found in numerous active pharmaceutical ingredients including antihypertensives and antimalarials. This technology leverages specific monooxygenases to catalyze the direct asymmetric amino hydroxylation of asymmetric alkenes and amines, bypassing the need for pre-formed chiral epoxides. By utilizing recombinant whole-cell catalysts, this process achieves exceptional enantioselectivity and yield under mild aqueous conditions, representing a significant paradigm shift from traditional chemical synthesis which often relies on hazardous reagents and complex multi-step sequences. The ability to access both (R)- and (S)-configurations simply by switching the enzyme variant offers unparalleled flexibility for process chemists aiming to optimize drug candidate synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino alcohols has been dominated by the nucleophilic ring-opening of epoxides with amines, a method fraught with significant technical and safety challenges. The primary bottleneck lies in the preparation of the chiral epoxide precursors, which typically requires expensive transition metal catalysts, harsh oxidizing agents, and rigorous exclusion of moisture to maintain stereochemical integrity. Furthermore, the ring-opening reaction itself often suffers from poor regioselectivity and requires the use of volatile organic solvents or stoichiometric amounts of Lewis acids, generating substantial chemical waste. Existing biocatalytic attempts have often relied on complex cascade systems involving four different enzymes to achieve similar transformations, creating immense logistical burdens regarding enzyme compatibility, cofactor balancing, and overall process robustness. These conventional limitations result in high production costs, extended lead times, and significant environmental liabilities that are increasingly untenable in modern green chemistry mandates.
The Novel Approach
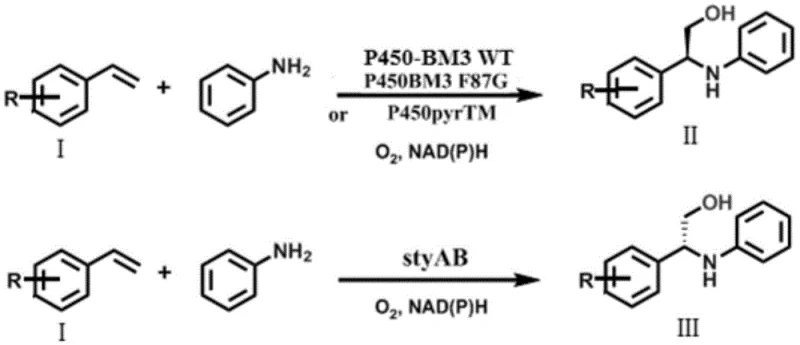
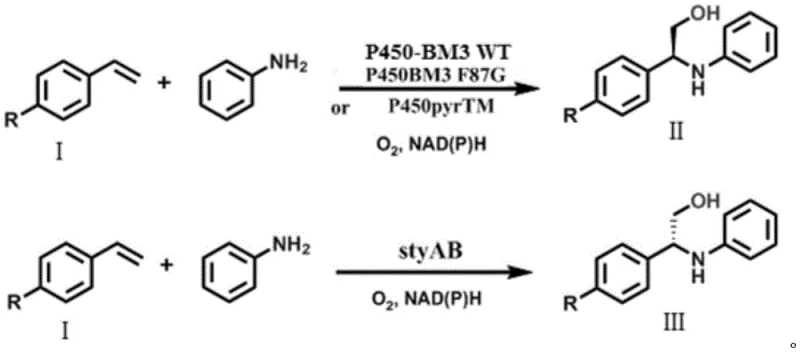
The innovative strategy disclosed in the patent data simplifies this complex landscape by employing a single monooxygenase to catalyze the entire transformation in a one-pot system. This method directly converts readily available bulk chemicals like styrene and aniline into high-purity chiral beta-amino alcohols without isolating unstable epoxide intermediates. The process utilizes engineered Escherichia coli strains expressing specific monooxygenases such as P450-BM3 variants or styAB, which effectively perform the asymmetric oxidation and subsequent amination in situ. Crucially, the system employs glucose as a cheap energy source for NADPH regeneration, eliminating the need for costly external cofactor addition. This streamlined approach not only drastically reduces the number of unit operations but also ensures high optical purity with ee values exceeding 90 percent, demonstrating a clear superiority over prior art methods in terms of both economic efficiency and operational simplicity.
Mechanistic Insights into Monooxygenase-Catalyzed Asymmetric Amino Hydroxylation
The core of this technological breakthrough lies in the precise stereospecificity of the employed monooxygenases, which dictate the configuration of the final product through a sophisticated catalytic cycle. The reaction mechanism involves the initial asymmetric epoxidation of the alkene substrate by the monooxygenase, forming a chiral epoxide intermediate that remains bound or in close proximity within the enzyme's active site or the immediate cellular environment. Subsequently, the amine substrate performs a nucleophilic attack on this transient epoxide, a step that is critically mediated by water molecules which stabilize the transition state and facilitate proton transfer. Computational models suggest that the chirality of the final beta-amino alcohol is strictly determined by the chirality of the enzymatically generated epoxide, ensuring that the stereochemical information is faithfully transferred from the catalyst to the product. This mechanistic understanding allows for the rational selection of enzymes, where P450-BM3 variants favor the formation of (S)-configured products while styAB directs the synthesis towards the (R)-enantiomer, providing a powerful tool for divergent synthesis.
Impurity control in this biocatalytic system is inherently superior due to the high substrate specificity of the enzyme active sites, which reject non-target isomers and side reactions common in chemical catalysis. The use of whole-cell catalysts further enhances purity by sequestering the reaction within the cellular matrix, effectively shielding the sensitive intermediates from bulk solution conditions that might promote racemization or degradation. The patent data highlights that the reaction conditions, specifically the pH range of 6.5 to 9.5 and temperatures between 25 and 45 degrees Celsius, are optimized to maintain enzyme stability while maximizing turnover rates. By avoiding the use of heavy metal catalysts and harsh organic solvents, the process minimizes the formation of metal-complex impurities and solvent-related byproducts, resulting in a cleaner crude reaction mixture that simplifies downstream purification. This inherent cleanliness is a vital attribute for pharmaceutical manufacturing, where strict limits on genotoxic impurities and residual metals must be met.
How to Synthesize Chiral Beta-Amino Alcohols Efficiently
The implementation of this biocatalytic route offers a straightforward protocol for producing high-value chiral intermediates with minimal infrastructure requirements. The process begins with the fermentation of recombinant E. coli strains to generate the biocatalyst, followed by a simple harvesting step to obtain the whole cells. These cells are then resuspended in a buffered aqueous solution containing glucose and the substrates, where the bioconversion proceeds efficiently at ambient pressure. The detailed standardized synthesis steps see the guide below for specific parameters regarding substrate loading and reaction times.
- Construct recombinant E. coli strains expressing specific monooxygenases such as P450-BM3 variants for (S)-selectivity or styAB for (R)-selectivity.
- Cultivate the engineered strains in TB medium, induce expression with IPTG, and harvest whole cells via centrifugation to serve as biocatalysts.
- Perform one-pot whole-cell catalysis in phosphate buffer with glucose for cofactor regeneration, reacting styrene derivatives with aniline at controlled temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this enzymatic technology presents a compelling value proposition by fundamentally altering the cost structure and risk profile of chiral intermediate production. The shift from specialized, hazard-classified epoxide reagents to commodity chemicals like styrene and aniline significantly de-risks the supply chain, ensuring consistent availability and protecting against price volatility associated with niche fine chemicals. The elimination of expensive transition metal catalysts and the reliance on fermentable glucose for cofactor regeneration translate into substantial raw material cost savings, making the process economically attractive even at large scales. Furthermore, the simplified one-pot nature of the reaction reduces the requirement for complex reactor setups and extensive solvent handling systems, lowering both capital expenditure and operational overheads associated with plant maintenance and safety compliance.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the replacement of high-cost chiral epoxides with inexpensive bulk alkenes and the removal of stoichiometric metal catalysts from the bill of materials. By utilizing whole-cell biocatalysis, the need for enzyme purification is obviated, further reducing processing costs and increasing the overall yield per batch. The efficient in situ regeneration of cofactors using glucose ensures that the reaction does not require continuous addition of expensive reducing agents, leading to a leaner and more cost-effective manufacturing process that maximizes resource utilization.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the primary substrates are widely produced petrochemical derivatives with robust global supply networks, unlike specialized chiral auxiliaries that may have single-source dependencies. The biological nature of the catalyst allows for rapid scale-up through standard fermentation techniques, enabling manufacturers to respond quickly to fluctuations in demand without long lead times for catalyst synthesis. This resilience ensures continuity of supply for critical drug intermediates, mitigating the risk of production stoppages due to raw material shortages or logistics disruptions in the fine chemical sector.
- Scalability and Environmental Compliance: The process aligns perfectly with green chemistry principles by operating in aqueous media and generating minimal hazardous waste, which simplifies wastewater treatment and reduces environmental compliance costs. The high selectivity of the enzyme minimizes the formation of byproducts, thereby increasing the mass efficiency of the process and reducing the load on purification units. This environmental friendliness not only meets stringent regulatory standards but also enhances the corporate sustainability profile, making the supply chain more attractive to eco-conscious partners and stakeholders in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this monooxygenase-catalyzed synthesis platform. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for potential adopters of this technology. Understanding these nuances is essential for evaluating the feasibility of integrating this biocatalytic route into existing manufacturing workflows.
Q: What are the advantages of this enzymatic method over traditional chemical synthesis?
A: Traditional methods rely on hazardous chiral epoxides and harsh conditions. This enzymatic approach uses cheap bulk chemicals like styrene and aniline, operates under mild aqueous conditions, and achieves high enantioselectivity without expensive metal catalysts.
Q: How is stereoselectivity controlled in this process?
A: Stereoselectivity is determined by the choice of enzyme. P450-BM3 variants and P450pyrTM produce (S)-configured products, while styAB produces (R)-configured products, allowing flexible access to both enantiomers from the same substrates.
Q: Is this process suitable for industrial scale-up?
A: Yes, the process utilizes robust whole-cell catalysts that can be produced via standard fermentation. It avoids complex multi-enzyme cascades and uses glucose for inexpensive cofactor regeneration, making it highly scalable and economically viable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Amino Alcohol Supplier
As a leader in custom synthesis and process development, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced enzymatic technology for the commercial production of high-purity chiral beta-amino alcohols. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications, providing our clients with a dependable source of critical intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative biocatalytic route can optimize your supply chain and reduce manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into the economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this sustainable and efficient technology into your production portfolio.
