Advancing Pharmaceutical Synthesis: A Green Zinc-Catalyzed Route to High-Purity Cyanoalkynes
Introduction to Novel Cyanoalkyne Synthesis Technology
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the urgent need for greener, safer, and more cost-effective methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN114591198A, which discloses a novel preparation method for cyanoalkyne compounds. These compounds are pivotal structural units in medicinal chemistry, serving as versatile precursors for a vast array of bioactive molecules, including peptide probes like APN-TMPP and antibody prodrugs such as CBTF. The dual functionality of the carbon-carbon triple bond and the cyano group allows for extensive downstream transformations into amines, amides, carboxylic acids, and heterocycles, making them indispensable for modern drug discovery pipelines. This patent introduces a paradigm shift by replacing hazardous cyanating agents with a benign sulfonamide derivative and utilizing earth-abundant zinc as a catalyst, thereby addressing critical safety and environmental concerns inherent in legacy synthetic routes.
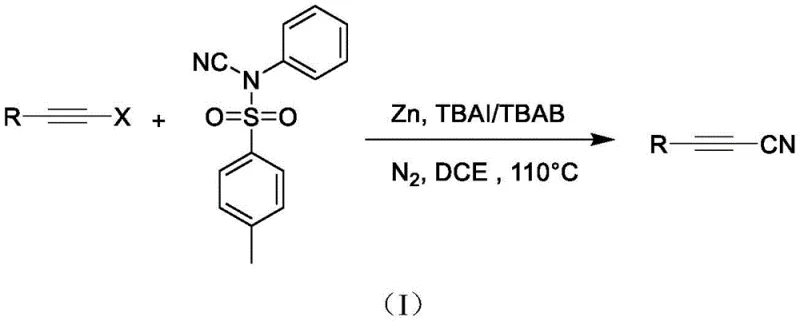
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyanoalkynes has been fraught with significant operational hazards and economic inefficiencies. Conventional strategies typically rely on the direct cross-coupling of terminal alkynes with剧毒 (highly toxic) cyanating agents such as cuprous cyanide (CuCN) or sodium cyanide (NaCN). These reagents pose severe risks to personnel safety and require elaborate containment systems and waste treatment protocols, drastically inflating the operational expenditure of any manufacturing facility. Furthermore, alternative methods involving copper-catalyzed reactions with cyanogen iodide or electrophilic cyanation using 1-cyanobenzotriazole often necessitate excessive amounts of expensive transition metal catalysts and rigorous reaction conditions, such as extreme temperatures or inert atmospheres that are difficult to maintain on a large scale. The generation of heavy metal waste and the potential for residual metal contamination in the final active pharmaceutical ingredient (API) are persistent quality control nightmares that complicate regulatory approval processes.
The Novel Approach
In stark contrast, the methodology outlined in CN114591198A offers a streamlined and environmentally benign alternative that fundamentally alters the economic and safety profile of cyanoalkyne production. By employing N-cyano-N-phenyl-p-toluenesulfonamide as a solid, easy-to-handle cyanide source, the process eliminates the immediate risks associated with volatile or soluble cyanide salts. The use of inexpensive zinc powder as the primary catalyst not only reduces raw material costs but also simplifies the removal of metal residues compared to noble metals like palladium or complex copper systems. As illustrated in the reaction scheme, the transformation proceeds smoothly in 1,2-dichloroethane (DCE) at a moderate temperature of 110°C under a nitrogen atmosphere, utilizing tetrabutylammonium salts as additives to enhance reactivity. This robust protocol ensures high conversion rates while maintaining a safety profile that is compatible with standard industrial hygiene practices.
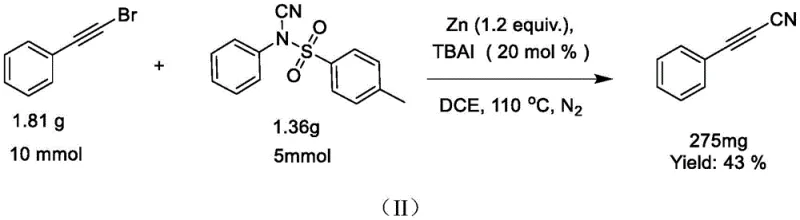
Mechanistic Insights into Zinc-Catalyzed Cyanation
The core of this innovation lies in the unique interaction between the zinc catalyst and the halogenated alkyne substrate, which facilitates a Reformatsky-type reaction mechanism. Unlike traditional nucleophilic substitutions that might struggle with the steric and electronic properties of sp-hybridized carbons, the zinc-mediated pathway likely involves the in situ generation of an organozinc intermediate. This intermediate is sufficiently nucleophilic to attack the cyano group transferred from the N-cyano-N-phenyl-p-toluenesulfonamide reagent, effectively displacing the sulfonamide leaving group. The presence of tetrabutylammonium iodide (TBAI) or tetrabutylammonium bromide (TBAB) acts as a crucial phase-transfer catalyst or halide exchange agent, activating the zinc surface and facilitating the oxidative addition step. This mechanistic nuance allows the reaction to tolerate a wide range of functional groups on the aromatic ring, including electron-withdrawing halogens and electron-donating alkoxy groups, without compromising the integrity of the triple bond.
From an impurity control perspective, this mechanism offers distinct advantages for GMP manufacturing. The byproduct of the reaction is a sulfonamide derivative, which is generally non-toxic and easily separable from the target cyanoalkyne via standard aqueous workups or silica gel chromatography. The absence of free cyanide ions in the reaction mixture minimizes the formation of hazardous side products and simplifies the purification workflow. Moreover, the specificity of the zinc catalyst reduces the likelihood of homocoupling side reactions (Glaser coupling) that often plague copper-catalyzed alkyne chemistries. This high selectivity translates directly into higher isolated yields, as evidenced by the patent data where yields for various substrates consistently reach levels between 63% and 87%, ensuring a reliable supply of high-purity material for downstream synthesis.
How to Synthesize Cyanoalkyne Compounds Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield. The process is designed to be operationally simple, requiring standard laboratory glassware such as Schlenk tubes, yet it is robust enough for kilogram-scale production. The key to success lies in the precise control of the nitrogen atmosphere to prevent oxidation of the zinc catalyst and the careful management of the reaction temperature at 110°C. Detailed standardized operating procedures regarding the addition sequence of reagents and the specific workup protocols are essential for maintaining batch-to-batch consistency. For a comprehensive understanding of the exact molar ratios and purification techniques required to replicate these results, please refer to the detailed synthesis guide below.
- Under a nitrogen atmosphere, combine halogenated alkyne, zinc powder, tetrabutylammonium additive (TBAI or TBAB), and 1,2-dichloroethane in a Schlenk tube and stir at room temperature.
- Add N-cyano-N-phenyl-p-toluenesulfonamide to the mixture, seal the vessel, and heat to 110°C for 48 hours to facilitate the cyanation reaction.
- Cool the reaction, filter through celite, wash with dichloromethane, remove solvent via rotary evaporation, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zinc-catalyzed technology represents a strategic opportunity to optimize the cost structure and resilience of the API intermediate supply chain. The shift away from precious metal catalysts and toxic cyanide sources directly impacts the bottom line by reducing both raw material expenditures and the overhead costs associated with hazardous waste disposal. Furthermore, the simplicity of the post-reaction processing—filtration followed by standard chromatography—reduces the manufacturing cycle time and minimizes the need for specialized equipment, thereby enhancing overall production throughput.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with commodity-grade zinc powder results in a drastic reduction in catalyst costs, which is particularly significant when scaling to multi-ton production volumes. Additionally, the use of a solid, stable cyanating reagent eliminates the need for specialized storage facilities and expensive safety infrastructure required for handling liquid or gaseous cyanides. The simplified purification process further lowers operational costs by reducing solvent consumption and labor hours dedicated to complex metal scavenging steps, leading to substantial overall cost savings in pharmaceutical manufacturing.
- Enhanced Supply Chain Reliability: The raw materials for this process, including zinc powder, haloalkynes, and quaternary ammonium salts, are globally available commodities with stable supply chains, mitigating the risk of production delays caused by raw material shortages. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor fluctuations in temperature or atmosphere compared to sensitive palladium-catalyzed couplings. This reliability ensures consistent delivery schedules for critical drug intermediates, supporting just-in-time manufacturing models and reducing inventory holding costs for downstream partners.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the viability of this method through successful gram-level reactions, proving its potential for seamless scale-up to industrial quantities without loss of efficiency. The green chemistry credentials of the process, characterized by low toxicity and minimal hazardous waste generation, align perfectly with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and enhances the corporate sustainability profile, making the supply chain more attractive to environmentally conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing production workflows.
Q: Why is this zinc-catalyzed method superior to traditional copper-cyanide coupling?
A: Traditional methods often rely on highly toxic reagents like cuprous cyanide or sodium cyanide and require harsh conditions. This novel approach utilizes low-toxicity N-cyano-N-phenyl-p-toluenesulfonamide and cheap zinc powder, significantly improving safety profiles and reducing hazardous waste disposal costs.
Q: What is the substrate scope for this cyanation reaction?
A: The method demonstrates high universality, successfully converting various aryl bromide alkynes and aryl chloride alkynes, including those with electron-donating or withdrawing groups, into the corresponding cyanoalkynes with considerable yields ranging from moderate to excellent.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the patent explicitly validates the method through gram-level reactions, demonstrating that the simple post-treatment and use of inexpensive catalysts make it highly suitable for commercial scale-up and continuous manufacturing of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyanoalkyne Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the zinc-catalyzed cyanation technology described in CN114591198A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in a practical, industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of cyanoalkyne intermediate delivered meets the exacting standards required for global drug registration.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and safe synthesis route for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you secure a sustainable and economical supply of critical cyanoalkyne building blocks for your next generation of life-saving medicines.
