Advanced Industrial Synthesis of Cefoxitin: Optimizing Yield and Purity from 7-ACA
Advanced Industrial Synthesis of Cefoxitin: Optimizing Yield and Purity from 7-ACA
The pharmaceutical landscape constantly demands more efficient pathways for critical antibiotics, and the synthesis of Cefoxitin stands as a prime example of process innovation. Based on the technical disclosures within patent CN101555252A, a robust methodology has been established that utilizes 7-Aminocephalosporanic acid (7-ACA) as the foundational starting material. This approach represents a significant departure from traditional reliance on Cephamycin C, addressing long-standing issues regarding raw material availability and process complexity. By integrating a streamlined sequence of acylation, methoxylation, and deacetoxylation, this technology offers a viable solution for manufacturers seeking to enhance their reliable antibiotic intermediate supplier capabilities. The core innovation lies in the seamless transition between reaction steps without intermediate purification, coupled with the strategic application of solid base catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Cefoxitin has been plagued by significant logistical and chemical hurdles, primarily stemming from the dependence on Cephamycin C as a precursor. Cephamycin C is notoriously difficult to obtain in large quantities with consistent quality, creating a bottleneck that destabilizes the supply chain for downstream pharmaceutical intermediates. Furthermore, conventional synthetic routes often involve multi-step processes with rigorous purification requirements between each stage, leading to extended production cycles and accumulated yield losses. The reliance on aqueous reaction systems in earlier methodologies also generated substantial volumes of wastewater contaminated with organic solvents, posing severe environmental compliance challenges and escalating waste treatment costs. These factors combined to create a high-cost, low-efficiency paradigm that struggled to meet the growing global demand for stable, beta-lactamase-resistant antibiotics.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data revolutionizes the workflow by anchoring the synthesis on the widely available and stable 7-ACA. This method ingeniously combines the acylation and methoxylation steps, allowing the crude cephalothin acid to proceed directly to methoxylation without isolation or purification. This telescoping of reactions not only simplifies the operational procedure but also dramatically improves the single-step yield by minimizing material handling losses. The transition from aqueous to organic phase reactions in the initial acylation step further mitigates the generation of solvent-laden wastewater, addressing critical environmental concerns. By implementing a solid biological base catalyst for the deacetoxylation step, the process achieves a level of efficiency and reusability that conventional chemical methods cannot match, ultimately resulting in a more cost-effective and environmentally sustainable manufacturing protocol.
Mechanistic Insights into Solid Base Catalyzed Deacetoxylation
The chemical elegance of this synthesis is most evident in the introduction of the 7-alpha-methoxy group, a structural feature critical for the antibiotic's stability against beta-lactamases. The mechanism involves the reaction of cephalothin acid with t-butyl hypochlorite and sodium methylate under strictly controlled low-temperature conditions, typically ranging from -85°C to 0°C. This precise thermal control is essential to prevent the degradation of the sensitive beta-lactam ring while ensuring the stereoselective introduction of the methoxy moiety. Following this, the formation of the cyclohexylamine salt serves as a crucial purification and stabilization checkpoint, preparing the intermediate for the subsequent deacetoxylation. The use of a solid biological base catalyst in this stage is particularly noteworthy, as it facilitates the removal of the acetoxy group at the 3-position under mild conditions, preserving the integrity of the delicate cephem nucleus while enabling high conversion rates.
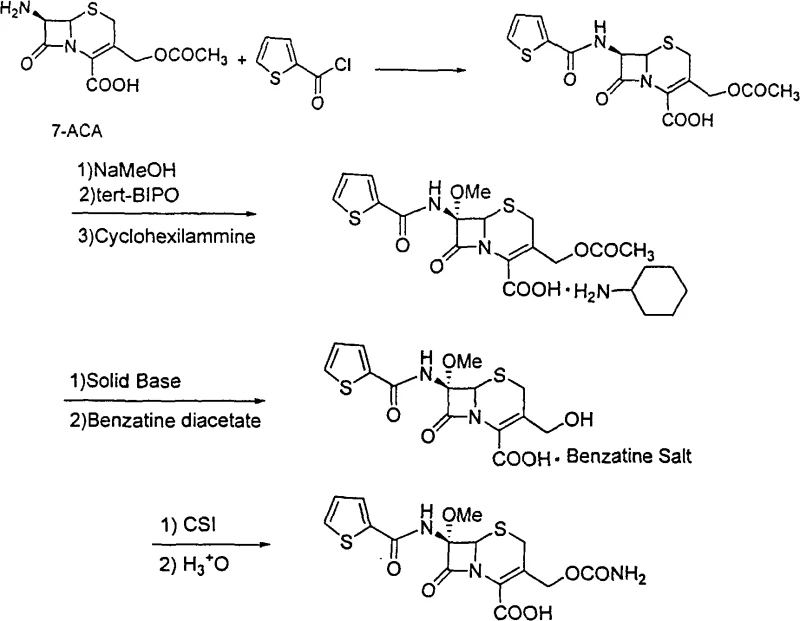
From an impurity control perspective, the decision to forego purification of the cephalothin acid prior to methoxylation is a calculated risk that pays off through process intensification. By maintaining the reaction mixture in an organic solvent system, the formation of hydrolytic byproducts is minimized compared to aqueous alternatives. The solid base catalyst employed in the deacetoxylation step offers a heterogeneous surface that likely restricts non-specific side reactions, thereby enhancing the purity profile of the resulting 7-alpha-methoxy-3-deacetoxy cephalothin benzathine salt. This high level of chemical fidelity is paramount for meeting the stringent purity specifications required for final API production. The final carbamylation using chlorosulfonyl isocyanate is executed with similar precision, ensuring the installation of the carbamate side chain without compromising the stereochemistry established in earlier steps.
How to Synthesize Cefoxitin Efficiently
Executing this synthesis requires strict adherence to the optimized reaction parameters outlined in the patent to ensure maximum yield and safety. The process begins with the dissolution of 7-ACA in an organic solvent such as methylene dichloride, followed by the controlled addition of thiopheneacetyl chloride. Operators must maintain the temperature between -20°C and 50°C during acylation to manage exothermicity effectively. Subsequent steps involve cryogenic conditions for methoxylation and precise pH control during salt formation. For a comprehensive breakdown of the specific reagent ratios, temperature profiles, and workup procedures required to replicate this high-efficiency route, please refer to the standardized synthesis guide below.
- Acylation of 7-ACA with thiopheneacetyl chloride in organic solvent to form cephalothin acid.
- Direct methoxylation of crude cephalothin acid using t-butyl hypochlorite and sodium methylate.
- Deacetoxylation using solid biological base catalyst and benzathine diacetate.
- Final carbamylation with chlorosulfonyl isocyanate to yield Cefoxitin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond simple unit cost metrics. The primary advantage lies in the decoupling of production from the volatile supply of Cephamycin C, replacing it with the robust and globally traded 7-ACA market. This shift significantly enhances supply chain reliability, ensuring that production schedules are not disrupted by raw material shortages. Furthermore, the elimination of intermediate purification steps reduces the overall processing time, allowing for faster turnaround times and improved responsiveness to market demand fluctuations. The ability to recycle solvents and the drastic reduction in wastewater treatment requirements also contribute to a leaner operational model with lower overheads.
- Cost Reduction in Manufacturing: The implementation of a solid biological base catalyst that can be reused for over 500 batches represents a monumental shift in cost structure. Unlike traditional liquid bases that are consumed in stoichiometric amounts and generate salt waste, this reusable catalyst minimizes raw material expenditure per kilogram of product. Additionally, the telescoping of the acylation and methoxylation steps eliminates the need for isolation, filtration, and drying of the cephalothin acid intermediate, thereby saving significant energy and labor costs. These cumulative efficiencies drive down the cost of goods sold (COGS) without compromising the quality of the final high-purity antibiotic intermediate.
- Enhanced Supply Chain Reliability: By utilizing 7-ACA, a commodity chemical with a mature and diversified global supply base, manufacturers mitigate the risk of single-source dependency. The simplified process flow, characterized by fewer unit operations and reduced handling, inherently lowers the probability of batch failures or delays. This operational robustness ensures a consistent flow of material to downstream API manufacturers, fostering stronger long-term partnerships. The stability of the process also means that inventory levels can be optimized, reducing the capital tied up in work-in-progress stock and improving overall cash flow dynamics for the enterprise.
- Scalability and Environmental Compliance: The transition away from aqueous systems and the adoption of solid-phase catalysis align perfectly with modern green chemistry principles. The reduction in wastewater discharge containing organic solvents simplifies the regulatory burden associated with environmental permits and effluent treatment. This makes the process highly scalable, as facilities do not need to invest heavily in expanded waste treatment infrastructure to increase capacity. The ease of scale-up from laboratory to commercial production ensures that the commercial scale-up of complex antibiotic intermediates can be achieved rapidly, supporting the rapid deployment of generic versions of this critical medication.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cefoxitin synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating technology transfer and for procurement officers assessing vendor capabilities. The focus remains on the practical implications of the solid base catalysis and the 7-ACA starting material strategy.
Q: What are the key advantages of the 7-ACA route over Cephamycin C for Cefoxitin?
A: The 7-ACA route utilizes readily available starting materials compared to Cephamycin C, which is difficult to source. Furthermore, the process simplifies operations by eliminating purification steps between acylation and methoxylation, significantly reducing the production cycle and cost.
Q: How does the solid base catalyst impact production costs?
A: The use of a solid biological base catalyst allows for reuse over 500 batches. This drastically reduces material consumption and waste disposal costs associated with conventional chemical bases, leading to substantial long-term operational savings.
Q: Does this method reduce environmental waste?
A: Yes, the process shifts from aqueous reactions to organic phase reactions where applicable and utilizes reusable solid catalysts. This eliminates the discharge of wastewater containing organic solvents, aligning with stricter environmental compliance standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefoxitin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the pharmaceutical sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101555252A are fully realized in practice. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Cefoxitin intermediate meets the highest international standards. Our commitment to process excellence allows us to deliver consistent quality while navigating the complexities of beta-lactam chemistry.
We invite you to collaborate with us to optimize your supply chain for this vital antibiotic. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply network that prioritizes both economic efficiency and technical superiority, ensuring your production lines remain uninterrupted and profitable.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
