Advanced Synthesis of 3-Hydroxy-Cepham Intermediates for Commercial Antibiotic Production
Introduction to Advanced Cephalosporin Intermediate Synthesis
The pharmaceutical industry continuously seeks robust and scalable methods for producing beta-lactam antibiotics, which remain the cornerstone of antimicrobial therapy globally. Patent CN101525341B introduces a groundbreaking preparation method for 3-hydroxy-cepham compounds, addressing critical limitations in traditional synthetic routes. This specific chemical entity serves as a pivotal building block for a wide array of clinically essential cephalosporins, including Cefaclor, Ceftibuten, and Ceftizoxime. The structural integrity of these molecules is paramount, as they exist in a delicate tautomeric equilibrium between the enol and keto forms, a feature that significantly influences their reactivity and stability during downstream processing. Understanding this equilibrium is crucial for optimizing crystallization and purity profiles in large-scale manufacturing environments.
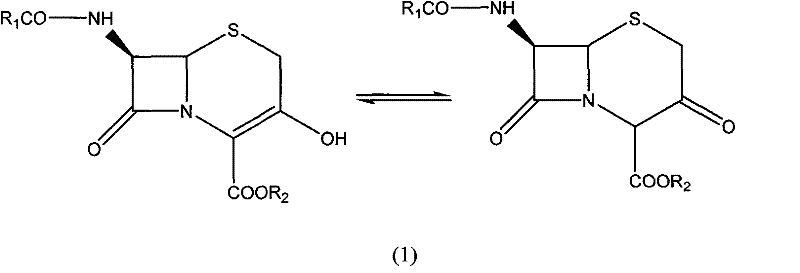
The innovation detailed in this patent represents a significant leap forward in process chemistry, moving away from hazardous reagents and multi-step isolation protocols that have historically plagued this sector. By leveraging a telescoped synthetic strategy, the method achieves superior yields exceeding 75% and purity levels greater than 90%, metrics that are highly attractive for commercial API production. For R&D directors and process chemists, this approach offers a streamlined pathway that minimizes impurity generation while maximizing atom economy. The ability to produce such high-value intermediates with reduced environmental impact and operational complexity positions this technology as a vital asset for any organization aiming to secure a reliable supply chain for next-generation antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-hydroxy-cepham derivatives has been fraught with challenges related to safety, cost, and efficiency. Prior art methods, such as those reported by Yoshioka et al., relied heavily on Methanesulfonyl chloride (MsCl) as the acylating reagent. MsCl is not only highly toxic and corrosive but also prone to generating significant side reactions that complicate purification and reduce overall yield, often capping productivity at around 70%. Furthermore, alternative routes utilizing 7-amino-cephalosporanic acid (7-ACA) as a starting material, while chemically viable, suffer from exorbitant raw material costs and lower transformation efficiencies. These conventional processes typically require the isolation of multiple intermediates, leading to increased solvent consumption, longer cycle times, and a larger environmental footprint due to waste generation.
The Novel Approach
In stark contrast, the methodology outlined in CN101525341B employs a sophisticated four-step sequence that elegantly bypasses these historical bottlenecks. The core innovation lies in the substitution of toxic MsCl with Tosyl chloride (TsCl), a reagent that offers a safer profile and superior reactivity control. This strategic switch effectively mitigates side reactions, allowing for a cleaner transformation of the thiazoline enol ester derivative. Moreover, the process is designed as a "one-pot" synthesis, meaning that the intermediate species generated after sulfonylation, enamine formation, and bromination are not isolated. Instead, the reaction mixture proceeds directly to the subsequent step, drastically reducing unit operations. This continuous flow of chemistry not only enhances throughput but also ensures that the reactive intermediates are consumed immediately, preventing degradation and ensuring high final purity.
Mechanistic Insights into the Telescoped Four-Step Synthesis
The success of this synthetic route hinges on precise control over reaction conditions and reagent selection across four distinct chemical transformations. The process initiates with a sulfonylation reaction where the thiazoline enol ester derivative reacts with Tosyl chloride in a biphasic solvent system, typically dichloromethane and water. The use of ammonia or ammonium hydroxide as the acid binding agent at temperatures between 0°C and 10°C is critical; this specific thermal window prevents the freezing of the aqueous phase while suppressing the formation of sulfonamide by-products. Following this, the reaction mixture undergoes an enamine formation step upon the addition of morpholine. Maintaining the temperature between -25°C and 0°C during this phase is essential to drive the equilibrium towards the desired enamine product without triggering thermal decomposition or polymerization of the sensitive beta-lactam core.
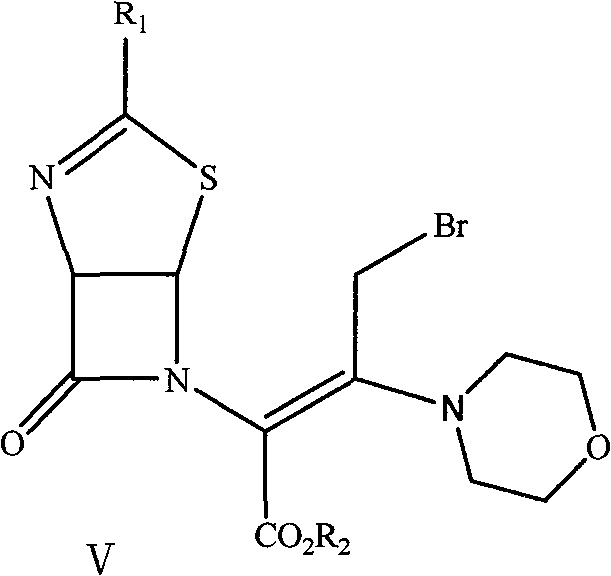
The subsequent bromination and cyclization steps represent the most technically demanding phases of the synthesis. In the bromination stage, liquid bromine is introduced at cryogenic temperatures ranging from -55°C to -15°C in the presence of pyridine. The choice of pyridine as the acid scavenger is a masterstroke of process optimization; it is significantly more cost-effective than the N-methylmorpholine or lutidine used in older methods, yet it provides excellent control over the exothermic bromination. Finally, the cyclization is achieved by treating the brominated intermediate with concentrated hydrochloric acid in methanol. This acid-catalyzed ring closure reconstructs the cephem nucleus, yielding the target 3-hydroxy-cepham compound. The entire sequence, as depicted in the reaction scheme, demonstrates a remarkable level of integration where each step sets up the perfect conditions for the next, minimizing the accumulation of impurities.
How to Synthesize 3-Hydroxy-Cepham Compound Efficiently
Implementing this synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and safety. The procedure begins with the dissolution of the starting thiazoline enol ester in a suitable organic solvent, followed by the controlled addition of Tosyl chloride under basic conditions. Once the sulfonylation is complete, verified by HPLC to ensure less than 0.1% residual starting material, the system is cooled for the enamine step. The subsequent addition of morpholine and base must be managed carefully to maintain the low-temperature regime. Following the enamine formation, the reaction mass is further cooled to sub-zero temperatures for the bromination, where the rate of bromine addition controls the heat release. The final cyclization in acidic methanol completes the transformation, after which standard workup procedures involving extraction, concentration, and crystallization yield the final product.
- Perform sulfonylation of thiazoline enol ester derivative with Tosyl chloride at 0-10°C using ammonia as base.
- Conduct enamine reaction by adding morpholine and triethylamine at -25°C to 0°C without isolating the intermediate.
- Execute bromination using liquid bromine and pyridine at -55°C to -15°C, followed by cyclization in HCl/methanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers tangible benefits that extend far beyond simple chemical yield. The shift from a multi-step isolation process to a telescoped "one-pot" operation fundamentally alters the cost structure of manufacturing. By eliminating the need to isolate and dry intermediate solids, the process significantly reduces the consumption of solvents and the energy required for drying and filtration equipment. This reduction in unit operations translates directly into lower utility costs and a smaller physical footprint for production facilities. Furthermore, the replacement of expensive and specialized reagents like N-methylmorpholine with commodity chemicals like pyridine and ammonia creates a more resilient supply chain that is less susceptible to price volatility of niche fine chemicals.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the simplification of the workflow and the substitution of high-cost inputs. Utilizing Tosyl chloride instead of Methanesulfonyl chloride not only improves safety but also reduces the cost associated with hazardous waste disposal and specialized handling equipment. The high conversion rates observed in the examples, with yields consistently reaching above 75% and purity exceeding 90%, mean that less raw material is wasted per kilogram of final product. This efficiency gain allows manufacturers to offer more competitive pricing for the final API while maintaining healthy margins, a critical factor in the highly price-sensitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Reliability in the supply of key intermediates is paramount for preventing production stoppages in downstream API manufacturing. This method utilizes widely available starting materials such as thiazoline enol ester derivatives and common solvents like dichloromethane and methanol, reducing the risk of supply shortages. The robustness of the reaction conditions, particularly the tolerance for slight variations in temperature during the sulfonylation and cyclization steps, ensures consistent batch-to-batch quality. This consistency is vital for regulatory compliance and reduces the likelihood of batch failures that can disrupt delivery schedules to global pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often reveals hidden inefficiencies, but this route is explicitly designed for scalability. The exothermic nature of the bromination step is managed through controlled addition and cooling, a protocol that scales linearly with reactor size. Additionally, the environmental profile of the process is markedly improved by avoiding toxic reagents and reducing solvent waste through the telescoped design. This alignment with green chemistry principles facilitates easier permitting and compliance with increasingly stringent environmental regulations, ensuring long-term operational continuity without the risk of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this technology for their own production lines or for procurement teams assessing the quality of suppliers utilizing this method.
Q: Why is Tosyl chloride preferred over Methanesulfonyl chloride in this synthesis?
A: Tosyl chloride is less toxic and generates fewer side reactions compared to the highly toxic Methanesulfonyl chloride used in prior art, leading to improved yield and environmental safety.
Q: What is the advantage of the 'one-pot' technique described in the patent?
A: The telescoped process eliminates the need to isolate intermediate products, significantly reducing solvent usage, processing time, and overall production costs while maintaining high purity.
Q: Which antibiotics utilize 3-hydroxy-cepham compounds as key intermediates?
A: This compound is a critical precursor for major cephalosporin antibiotics including Cefaclor, Ceftibuten, Ceftizoxime, Cefpodoxime Proxetil, and Ceftaroline.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-Cepham Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and state-of-the-art infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. We are committed to delivering high-purity 3-hydroxy-cepham compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our capability to manage complex beta-lactam chemistry allows us to consistently deliver material that supports the uninterrupted manufacture of life-saving cephalosporin antibiotics.
We invite global pharmaceutical partners to engage with us to explore how this optimized synthesis can enhance your supply chain resilience. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can drive value for your organization.
