Advanced Biphenyl Triphosphine Ligands for High-Selectivity Hydroformylation Manufacturing
The chemical industry is constantly seeking methods to optimize catalytic efficiency, particularly in the massive sector of hydroformylation where millions of tons of aldehydes are produced annually. Patent CN103804413A introduces a groundbreaking advancement in this field by disclosing a series of novel biphenyl triphosphine ligands designed to overcome the inherent limitations of traditional triphenylphosphine (PPh3) systems. These new ligands, characterized by a rigid biphenyl backbone supporting three phosphine donor atoms, offer a unique steric and electronic environment that dramatically enhances reaction selectivity towards linear aldehydes. For R&D directors and process chemists, this represents a significant opportunity to refine impurity profiles and boost throughput in the synthesis of critical fine chemical intermediates. The structural versatility of these ligands allows for extensive tuning of the aryl substituents, enabling precise control over the catalytic pocket.

Furthermore, the patent highlights a pragmatic approach to industrial adoption through a gradual replacement strategy, mitigating the economic shock of switching entirely to expensive novel catalysts. This dual focus on high-performance molecular design and practical implementation makes this technology highly attractive for commercial scale-up of complex polymer additives and pharmaceutical precursors. By addressing both the thermodynamic selectivity issues and the kinetic barriers associated with older catalyst generations, this innovation paves the way for more sustainable and efficient manufacturing processes. The ability to fine-tune the ligand structure means that manufacturers can adapt the catalyst specifically for difficult substrates, ensuring consistent quality in high-value production runs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the hydroformylation industry has relied heavily on triphenylphosphine (PPh3) as the standard ligand for rhodium-catalyzed processes due to its availability and low cost. However, from a technical standpoint, PPh3 suffers from significant drawbacks that impact the bottom line and product quality. Its relatively small cone angle and weak electron-donating capability often result in poor regioselectivity, yielding a mixture of linear and branched aldehydes that requires energy-intensive separation downstream. Moreover, the separation of the PPh3-Rh catalyst complex from the product stream is notoriously difficult, leading to metal contamination in the final product and loss of expensive rhodium. In large-scale operations, these inefficiencies translate into higher purification costs and reduced overall yield, creating a bottleneck for cost reduction in fine chemical manufacturing. The inability to effectively tune the steric bulk of PPh3 limits its utility in synthesizing highly specific linear intermediates required for advanced materials.
The Novel Approach
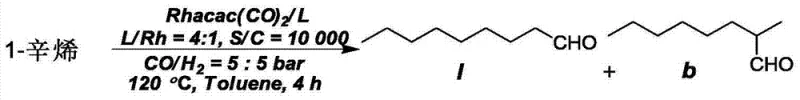
The novel biphenyl triphosphine ligands described in the patent offer a transformative solution by introducing a tridentate coordination mode that fundamentally alters the geometry of the active rhodium species. Unlike the monodentate or bidentate binding of conventional ligands, the tridentate nature of these new molecules creates a highly defined and rigid coordination sphere around the metal center. This rigidity sterically favors the formation of the linear alkyl-rhodium intermediate, thereby drastically increasing the linear-to-branched (l/b) ratio of the aldehyde products. As demonstrated in the patent data, the gradual introduction of these ligands into an existing PPh3 system can push selectivity to exceptional levels without necessitating a complete process redesign. This approach not only improves the purity of the high-purity OLED material precursors or plasticizers but also simplifies the workup procedure. The enhanced stability of the tridentate complex also reduces ligand dissociation, minimizing catalyst degradation and extending the operational life of the catalytic system.
Mechanistic Insights into Tridentate Phosphine-Rhodium Catalysis
The superior performance of the biphenyl triphosphine ligands stems from their ability to enforce a specific coordination geometry that dictates the outcome of the catalytic cycle. In the hydroformylation mechanism, the ligand occupies three coordination sites on the rhodium center, leaving limited space for the substrate and syngas (CO/H2). This crowding effect creates a strong steric bias that disfavors the formation of the branched alkyl intermediate, which requires more space than the linear counterpart. Consequently, the reaction pathway is funneled almost exclusively towards the linear aldehyde product. Electronic effects also play a crucial role; the electron-rich phosphine centers stabilize the electron-deficient rhodium species during the oxidative addition step, accelerating the overall reaction rate. This synergy between steric bulk and electronic donation ensures that the catalyst remains active and selective even under the demanding conditions of industrial reactors. For process engineers, understanding this mechanism is key to optimizing parameters such as temperature and pressure to maximize the turnover number (TON).
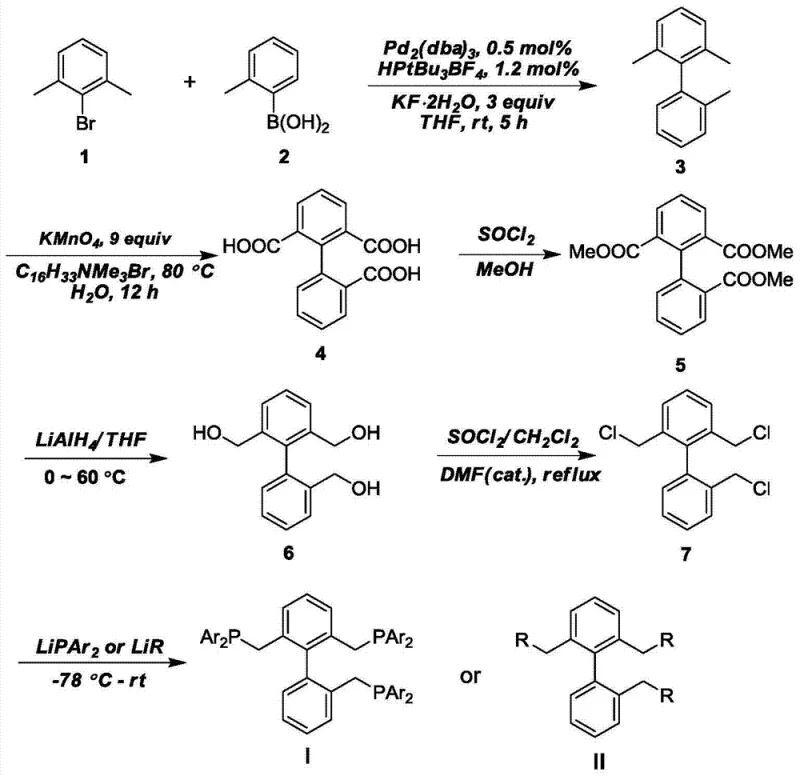
From a synthetic chemistry perspective, the accessibility of these ligands is equally impressive, relying on robust and scalable transformations. The core biphenyl scaffold is constructed via a palladium-catalyzed Suzuki-Miyaura coupling, a reaction known for its tolerance to functional groups and high yields. Subsequent functionalization involves oxidation of methyl groups to carboxylic acids, followed by reduction to alcohols and conversion to chlorides, providing versatile handles for the final phosphination step. This modular synthesis allows for the easy introduction of diverse aryl groups on the phosphorus atoms, enabling the fine-tuning of solubility and electronic properties. Such synthetic flexibility ensures that the ligand library can be adapted to specific solvent systems or substrate requirements, making it a versatile tool for reducing lead time for high-purity agrochemical intermediates. The high yields reported in each step of the synthesis confirm the practical viability of producing these ligands on a multi-kilogram scale.
How to Synthesize Biphenyl Triphosphine Ligands Efficiently
The synthesis of these advanced ligands follows a logical sequence of organic transformations that are well-established in industrial chemistry, ensuring reproducibility and safety. The process begins with the construction of the sterically hindered biphenyl core, followed by the systematic installation of the phosphine arms. Each step has been optimized in the patent to minimize side reactions and maximize isolation yields, which is critical for maintaining cost-effectiveness. The use of standard reagents like lithium aluminum hydride and thionyl chloride means that specialized equipment is not required, facilitating easy technology transfer. For detailed operational parameters, stoichiometry, and purification techniques, please refer to the standardized guide below which outlines the critical control points for each stage of the synthesis.
- Perform Suzuki coupling of 2,6-dimethylbromobenzene and 2-methylphenylboronic acid using Pd2(dba)3 catalyst to form the biphenyl backbone.
- Oxidize the methyl groups to carboxylic acids using KMnO4, followed by esterification with methanol and SOCl2 to protect the functionality.
- Reduce the esters to alcohols using LiAlH4, convert to chlorides with SOCl2, and finally react with lithium diarylphosphides to install the phosphine groups.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers tangible strategic benefits beyond mere technical performance. The primary advantage lies in the potential for significant cost optimization through the gradual replacement strategy. Instead of incurring the prohibitive expense of replacing an entire inventory of PPh3 catalyst overnight, facilities can incrementally introduce the biphenyl triphosphine ligand. This phased approach allows for budget smoothing and risk mitigation while still capturing the efficiency gains of the new technology. Furthermore, the higher selectivity directly translates to reduced waste generation and lower energy consumption during downstream purification, contributing to substantial operational expenditure (OpEx) savings. The robustness of the ligand synthesis also implies a stable supply chain, as the raw materials are commodity chemicals rather than exotic precursors.
- Cost Reduction in Manufacturing: The implementation of these ligands drives down manufacturing costs primarily through improved atom economy and reduced separation burdens. By achieving higher linear selectivity, the need for energy-intensive distillation columns to separate isomers is diminished, leading to direct savings in utility costs. Additionally, the enhanced stability of the tridentate catalyst reduces the frequency of catalyst replenishment, lowering the consumption of expensive rhodium metal. The synthesis route itself avoids the use of precious metal catalysts in the ligand preparation (except for the initial Suzuki step which uses recoverable Pd), keeping the ligand production costs competitive. These factors combine to create a more economically resilient production model for high-volume chemical commodities.
- Enhanced Supply Chain Reliability: The synthetic pathway relies on widely available starting materials such as bromobenzenes and boronic acids, which are produced by multiple global suppliers. This diversity in sourcing mitigates the risk of supply disruptions that often plague specialty chemical markets. The scalability of the process, demonstrated from gram to multi-gram scales in the patent examples, indicates that scaling to metric ton quantities is feasible with standard reactor setups. This ensures that reliable agrochemical intermediate supplier networks can maintain consistent delivery schedules even during periods of high demand. The chemical stability of the final ligand product also simplifies storage and logistics, reducing the risk of degradation during transit.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry principles by minimizing waste and improving energy efficiency. The high conversion rates and selectivity mean less unreacted feedstock ends up in waste streams, simplifying effluent treatment. The ability to operate at moderate temperatures and pressures compared to some older cobalt-based systems further reduces the carbon footprint of the facility. As regulatory pressures on industrial emissions tighten, adopting a catalyst system that inherently produces cleaner product streams provides a proactive compliance advantage. This scalability ensures that the technology remains viable as production volumes increase to meet global market demands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of biphenyl triphosphine ligands in hydroformylation processes. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines. The responses cover aspects ranging from catalyst compatibility to the specific benefits observed in pilot-scale reactions.
Q: Why replace Triphenylphosphine (PPh3) in hydroformylation?
A: While PPh3 is common, it offers poor selectivity for linear aldehydes and is difficult to separate from the product. Novel tridentate ligands provide superior regioselectivity (high l/b ratio) and facilitate easier catalyst recovery.
Q: Is the gradual replacement method cost-effective?
A: Yes. The patent demonstrates that gradually adding the novel ligand to an existing PPh3/Rh system significantly boosts selectivity without requiring a complete and expensive immediate overhaul of the catalyst inventory.
Q: What is the scalability of this ligand synthesis?
A: The synthesis utilizes robust reactions like Suzuki coupling and standard functional group transformations (oxidation, reduction), which are well-suited for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Triphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced ligand technologies in driving the next generation of chemical manufacturing. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering these complex ligands with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation. Whether you require custom modifications to the aryl substituents or large-volume supply for continuous processing, our infrastructure is designed to meet the exacting standards of the global pharmaceutical and fine chemical industries.
We invite you to collaborate with us to unlock the full value of this patented technology for your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current hydroformylation process. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our biphenyl triphosphine ligands can enhance your operational efficiency and product quality. Let us be your partner in achieving superior catalytic performance and sustainable growth.
