Scalable Copper-Catalyzed Synthesis of Fused Tricyclic Sulfonamides for Commercial API Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic scaffolds, particularly those serving as critical intermediates for neurodegenerative disease treatments. Patent CN102439010A introduces a transformative methodology for the preparation of fused tricyclic sulfonamide analogs, specifically 5-(aryl-sulfonyl)-4,5-dihydro-1H-pyrazolo[4,3-c]quinolines, which are potent gamma-secretase inhibitors. This technology addresses the longstanding challenges associated with constructing the tricyclic core, shifting from inefficient stoichiometric metal usage to a refined catalytic system. By leveraging a copper-based catalyst augmented with specific organic ligands, the process achieves superior control over regioselectivity and impurity profiles. For R&D and manufacturing teams, this represents a significant leap forward in process chemistry, enabling the production of high-purity intermediates essential for the development of Alzheimer's disease therapeutics. The patent outlines not just the chemical transformation but a holistic approach to process optimization that considers yield, safety, and environmental impact.
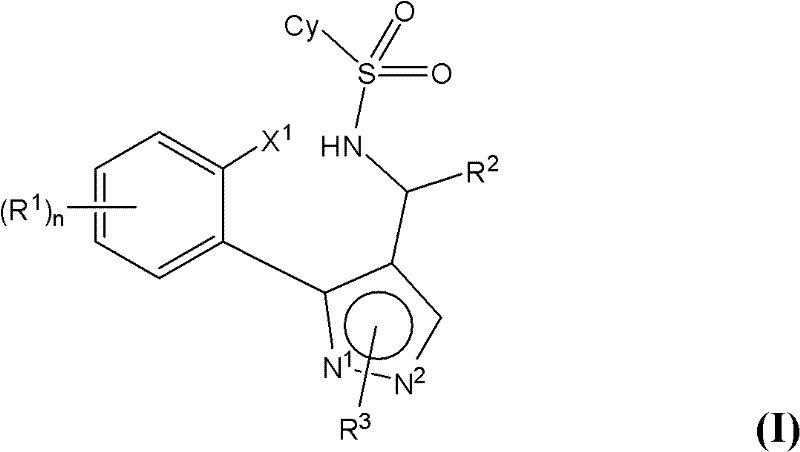
The transition from legacy synthetic strategies to this novel catalytic approach marks a pivotal improvement in manufacturing efficiency and product quality. Conventional methods for forming the carbon-nitrogen bond in these tricyclic systems often relied on harsh conditions or excessive amounts of copper reagents, which frequently led to significant levels of debrominated by-products and difficult-to-remove metal residues. These limitations necessitated extensive purification steps, such as column chromatography, which are impractical for commercial scale-up. In contrast, the novel approach described in the patent employs a ligand-accelerated copper catalysis system. This system facilitates the intramolecular cyclization under milder conditions while drastically suppressing side reactions. The result is a cleaner reaction profile that minimizes the formation of undesired debrominated species, thereby simplifying the downstream processing requirements. This shift eliminates the need for costly and time-consuming chromatographic separations, allowing for direct crystallization of the product.
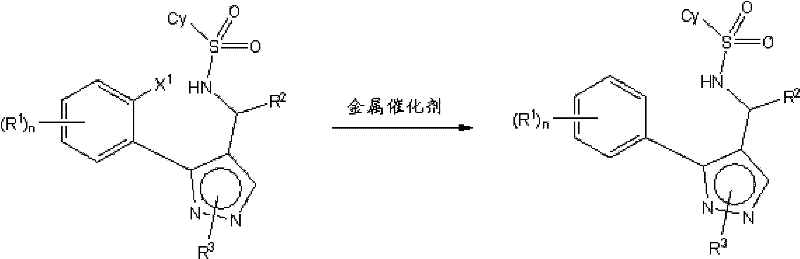
At the heart of this technological advancement lies a sophisticated understanding of organometallic catalysis and ligand effects on reaction kinetics. The mechanism involves the formation of an active copper-ligand complex, likely involving Cu(I) species coordinated by diamines such as DMEDA. This complex facilitates the oxidative addition into the carbon-halogen bond of the precursor, followed by intramolecular amidation to close the ring. The presence of the organic ligand is crucial; it stabilizes the copper center and modulates its electronic properties, preventing the premature reduction of the aryl halide which leads to debromination. Furthermore, the process allows for precise control over the stereochemistry when chiral precursors are used, maintaining high enantiomeric excess throughout the cyclization. The ability to tune the catalyst loading down to catalytic levels (e.g., less than 2 equivalents, often as low as 2 mol%) without sacrificing conversion rates demonstrates the high turnover frequency of this system. This mechanistic efficiency translates directly into reduced raw material costs and lower metal waste generation.
How to Synthesize Fused Tricyclic Sulfonamides Efficiently
The synthesis of these complex pharmaceutical intermediates requires precise control over reaction parameters to ensure consistent quality and yield. The patented process outlines a sequence that begins with the preparation of a sulfonamide precursor containing a leaving group, followed by the critical cyclization step. Detailed operational guidelines regarding temperature profiles, solvent choices, and work-up procedures are essential for replicating the high success rates reported in the patent examples. Implementing this route involves careful selection of the copper source and ligand ratio to maximize catalytic activity while minimizing impurity formation. The following guide summarizes the critical operational steps derived from the patent data to assist technical teams in evaluating the feasibility of this route for their specific manufacturing needs.
- Prepare the precursor molecule containing a sulfonamide group and a leaving group on the aromatic ring.
- React the precursor with a copper catalyst system comprising CuI and an organic ligand like DMEDA in a high-boiling solvent.
- Purify the resulting tricyclic product through crystallization to achieve high chemical and chiral purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain stakeholders, the adoption of this catalytic cyclization technology offers substantial strategic benefits regarding cost structure and supply reliability. Traditional synthesis routes often suffered from poor atom economy due to the use of stoichiometric metal reagents and generated significant waste streams that required expensive disposal. By transitioning to a catalytic regime, the consumption of copper is drastically reduced, leading to direct savings on raw material costs. Moreover, the suppression of debrominated impurities means that the crude product quality is significantly higher, reducing the burden on quality control laboratories and shortening the overall production cycle time. This efficiency gain enhances the reliability of supply, as the process is less prone to batch failures caused by difficult purifications. The use of readily available ligands and common solvents further secures the supply chain against volatility in specialized reagent markets.
- Cost Reduction in Manufacturing: The shift from stoichiometric to catalytic copper usage fundamentally alters the cost basis of the synthesis. By reducing the copper load to mere mole percentages, the expense associated with metal reagents is minimized, and the cost of removing heavy metal residues from the final product is significantly lowered. Additionally, the elimination of chromatographic purification steps in favor of crystallization reduces solvent consumption and labor hours, contributing to a leaner manufacturing process. These cumulative efficiencies result in a more cost-effective production model without compromising the stringent purity standards required for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more stable supply of critical intermediates. The process tolerates a wide range of substrates and uses common industrial solvents like toluene and alcohols, which are readily available globally. This reduces the risk of supply disruptions associated with exotic or highly regulated reagents. Furthermore, the high yield and reproducibility of the cyclization step mean that production schedules can be met with greater confidence, supporting just-in-time manufacturing strategies and reducing the need for excessive safety stock inventory.
- Scalability and Environmental Compliance: Designed with scale-up in mind, this method avoids hazardous conditions and excessive waste generation. The reduced metal content in the waste stream simplifies environmental compliance and wastewater treatment processes. The ability to perform the reaction at moderate temperatures and pressures enhances operational safety, making it suitable for large-scale reactors. The streamlined work-up, involving simple filtration and crystallization, facilitates rapid throughput, allowing manufacturers to respond quickly to market demand while adhering to green chemistry principles.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into existing production lines. The following questions address common concerns regarding the practical application of this patented technology, focusing on impurity control, scalability, and compatibility with downstream processes. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable foundation for technical decision-making.
Q: How does the new copper-ligand system improve upon traditional cyclization methods?
A: The patented method utilizes a catalytic amount of copper combined with specific organic ligands like DMEDA, which significantly reduces the formation of debrominated by-products compared to stoichiometric copper methods, leading to higher yields and easier purification.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the process is explicitly designed for variable-scale to large-scale preparation, utilizing robust reaction conditions and simplified work-up procedures that avoid complex chromatographic separations.
Q: What are the key impurities controlled in this process?
A: The method effectively suppresses the formation of debrominated side products and aromatized quinoline impurities, ensuring the final API intermediate meets stringent purity specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fused Tricyclic Sulfonamide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced catalytic technologies like the one described in CN102439010A to deliver high-value pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fused tricyclic sulfonamides meets the exacting standards of the global pharmaceutical industry. Our commitment to process innovation allows us to offer competitive solutions that balance cost, quality, and speed.
We invite you to collaborate with us to optimize your supply chain for these critical Alzheimer's disease therapeutic intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can enhance your project's success and accelerate your time to market.
