Advanced Synthesis of Cefetamet Acid: A Technical Breakthrough for Industrial Scale Production
The pharmaceutical industry constantly seeks robust synthetic routes for critical antibiotic intermediates, and the methodology detailed in patent CN101712687A represents a significant leap forward in the production of cefetamet acid. This compound serves as the pivotal precursor for cefetamet pivoxil hydrochloride, a third-generation oral cephalosporin known for its broad-spectrum antibacterial activity. The disclosed innovation addresses long-standing inefficiencies in the synthesis of Ro 15-8075 intermediates by optimizing raw material ratios and, most critically, refining the crystallization process. By shifting away from harsh reaction conditions and complex protecting group strategies, this technology enables manufacturers to achieve exceptional purity levels exceeding 99% while maintaining high molar yields. For global stakeholders, this translates to a more reliable pharmaceutical intermediate supplier capable of delivering consistent quality without the volatility associated with older, less stable chemical processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefetamet acid has been plagued by operational complexities that hinder industrial scalability and economic efficiency. Traditional routes often necessitate the introduction and subsequent removal of amino protecting groups, a multi-step sequence that inherently lowers overall throughput and increases waste generation. Literature references, such as those by Li Kun, highlight methods requiring stringent low-temperature crystallization between 0°C and 5°C, which demands significant energy expenditure for cooling infrastructure. Furthermore, these legacy processes often struggle with product isolation, where wet products require extensive washing with alcohol and water, leading to substantial material loss. Reported molar yields in these conventional setups frequently hover around 79.4%, indicating a significant portion of valuable starting materials like 7-ADCA is lost to side reactions or inefficient recovery. Additionally, methods described by other researchers have resulted in products with high impurity profiles, complicating downstream purification and jeopardizing the quality of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the novel approach outlined in the patent data utilizes a direct condensation strategy that bypasses the need for cumbersome protecting group chemistry. By reacting 3-methyl-7-amino-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-formic acid (7-ADCA) directly with 2-methoxyimino-2-(2-amino-4-thiazolyl)-(z)-thioacetic acid phenylhydrazine thiazole ester (AE active ester), the process achieves remarkable efficiency. The reaction is conducted under mild conditions, specifically within a temperature range of 18°C to 22°C, eliminating the need for energy-intensive heating or deep cooling. A key differentiator is the precise control over the crystallization environment, where the pH is meticulously adjusted to initiate crystal growth at 4.50. This optimization allows for the production of cefetamet acid with a molar yield reaching 98% and purity levels of 99%.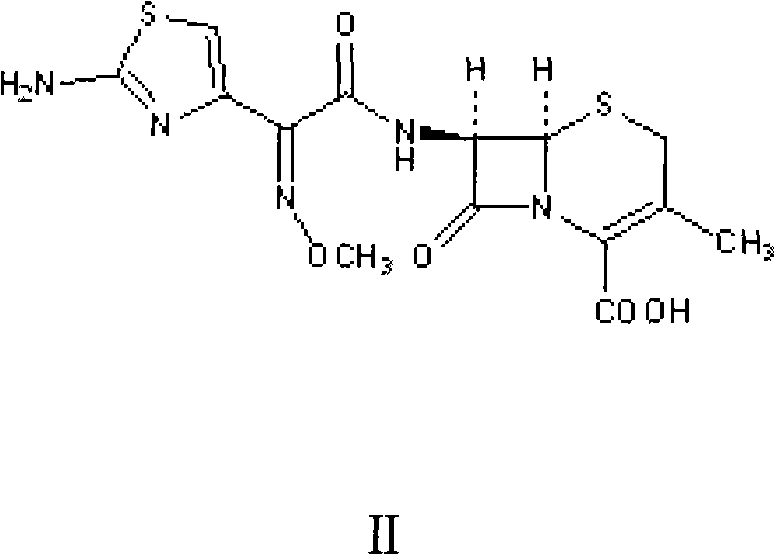
Mechanistic Insights into DMF-Catalyzed Condensation and pH-Controlled Crystallization
The core of this synthetic breakthrough lies in the catalytic condensation mechanism facilitated by N,N-dimethylformamide (DMF) within a mixed solvent system of acetone and water. The presence of triethylamine acts as a base to deprotonate the amine group on the 7-ADCA nucleus, enhancing its nucleophilicity for the attack on the activated ester carbonyl of the AE side chain. The choice of solvent ratio, optimally 4:1 acetone to water, creates a homogeneous reaction medium that supports high conversion rates while maintaining the solubility of reactants. Crucially, the reaction kinetics are managed at ambient temperatures (20°C), which minimizes thermal degradation of the sensitive beta-lactam ring, a common failure point in cephalosporin synthesis. The use of a specific catalyst loading, approximately 1% of the total gross weight of reactants, ensures rapid reaction completion within 3.5 hours, demonstrating a highly efficient catalytic cycle that maximizes atom economy.
Following the condensation, the purification mechanism relies heavily on a sophisticated pH-gradient crystallization technique rather than simple precipitation. After removing organic solvents like dichloromethane and acetone under reduced pressure, the aqueous solution is treated with activated carbon to adsorb colored impurities and trace organics. The subsequent addition of dilute hydrochloric acid is performed in a controlled stream to lower the pH. The data indicates that initiating crystal growth at a pH of 4.50 is the thermodynamic sweet spot; deviating to 4.40 or 4.60 results in measurable drops in both yield and purity. This suggests that at pH 4.50, the zwitterionic form of the cefetamet acid achieves optimal supersaturation for nucleation without co-precipitating acidic or basic impurities. Final adjustment to pH 3.00 ensures complete recovery of the product from the mother liquor, securing the high yields observed in the experimental embodiments.
How to Synthesize Cefetamet Acid Efficiently
The synthesis of this critical antibiotic intermediate requires strict adherence to the optimized parameters regarding stoichiometry and pH control to replicate the high success rates seen in the patent examples. The process begins with the preparation of a reaction solution containing 7-ADCA dissolved in a specific acetone-water mixture with triethylamine, followed by the addition of the AE active ester and DMF catalyst. Maintaining the insulation reaction at 20°C for 3.5 hours is vital to ensure complete conversion before proceeding to the workup phase. The subsequent steps involve solvent exchange, decolorization, and the critical pH-adjusted crystallization. For a comprehensive understanding of the exact operational parameters and safety protocols required for this synthesis, please refer to the standardized guide below.
- Dissolve 7-ADCA in an acetone-water-triethylamine mixture and react with AE active ester using a DMF catalyst at 20°C.
- Perform post-reaction treatment by extracting with dichloromethane, removing solvents under vacuum, and decolorizing with activated carbon.
- Adjust pH to 4.50 for crystal growth, then lower to 3.00 to complete crystallization, followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers profound strategic advantages beyond mere chemical yield. The elimination of protecting group steps fundamentally simplifies the supply chain by reducing the number of distinct raw materials required, thereby mitigating the risk of shortages for specialized reagents. Furthermore, the ability to recover and reuse solvents such as acetone and dichloromethane through condensation significantly lowers the variable costs associated with raw material consumption. This closed-loop solvent management not only drives down operational expenditures but also aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. The mild reaction conditions further contribute to cost reduction in API manufacturing by reducing the energy load on facility HVAC and chilling systems, allowing for production in standard stainless steel reactors without the need for specialized cryogenic equipment.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive protecting group reagents and the associated waste disposal costs, leading to substantial cost savings. By operating at near-room temperature, the method drastically reduces energy consumption compared to cryogenic processes, while the high yield ensures that the cost per kilogram of the final intermediate is minimized through better raw material utilization.
- Enhanced Supply Chain Reliability: The robustness of this method, demonstrated by its successful scale-up from grams to hundreds of kilograms, ensures a stable supply of high-purity intermediates. The use of commodity chemicals like acetone, water, and triethylamine reduces dependency on niche suppliers, thereby shortening lead times for high-purity pharmaceutical intermediates and insulating production schedules from market volatility.
- Scalability and Environmental Compliance: The process generates minimal three wastes due to efficient solvent recovery and high atom economy, simplifying wastewater treatment and reducing environmental compliance burdens. The stability of the reaction at ambient temperatures makes it highly scalable, allowing manufacturers to increase batch sizes from pilot plants to full commercial production without encountering the heat transfer or mixing limitations often seen in exothermic or low-temperature reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cefetamet acid synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process capabilities and quality outcomes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing lines.
Q: What is the optimal pH for crystal growth in cefetamet acid synthesis?
A: According to patent data, maintaining the pH at 4.50 during the crystal growth phase yields the highest product purity (99%) and molar yield (98%).
Q: How does this method improve upon traditional synthesis routes?
A: Unlike traditional methods requiring harsh protecting group manipulations and low-temperature crystallization (0-5°C), this novel approach operates at mild temperatures (18-22°C) and eliminates complex protection steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scale-up from laboratory gram scales to 1000x larger batches (hundreds of kilograms), proving stability and suitability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefetamet Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial reality is seamless. We are committed to delivering cefetamet acid that meets stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest international standards. Our facility is equipped to handle the specific solvent recovery and pH-controlled crystallization requirements of this advanced process, guaranteeing a consistent supply of material that supports your downstream synthesis of cefetamet pivoxil.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your operational efficiency and reduce your overall cost of goods sold.
