Advanced Manufacturing of High-Purity Calcium 5-Methyltetrahydrofolate for Global Pharma Supply Chains
Advanced Manufacturing of High-Purity Calcium 5-Methyltetrahydrofolate for Global Pharma Supply Chains
The global demand for bioactive folate derivatives continues to surge, driven by the nutritional supplement and pharmaceutical sectors' need for highly bioavailable forms of Vitamin B9. Patent CN114957257A presents a transformative approach to the synthesis of calcium 5-methyltetrahydrofolate, addressing the persistent challenge of impurity control that has long plagued conventional manufacturing routes. This technology specifically targets the removal of pterin-type degradation products that typically accumulate during the harsh alkaline conditions of borohydride reduction. By introducing a strategic acid-solubilization purification step prior to the final salt formation, the process ensures that the intermediate 5-methyltetrahydrofolic acid achieves a purity level exceeding 98 percent. For R&D directors and procurement specialists, this represents a significant leap forward in securing a reliable pharmaceutical intermediates supplier capable of delivering material that meets stringent regulatory specifications without the need for costly downstream reprocessing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for calcium 5-methyltetrahydrofolate rely heavily on the reduction of folic acid using sodium borohydride or potassium borohydride. While these reducing agents are effective for converting the pteridine ring, they inherently create a strongly alkaline reaction environment. In such high-pH conditions, the delicate folate structure is prone to degradation, leading to the formation of stubborn pterin impurities and p-aminobenzoylglutamic acid. These byproducts are structurally similar to the target molecule, making them exceptionally difficult to separate using standard crystallization techniques. Consequently, manufacturers often struggle to push final product purity above the critical 98 percent threshold required for high-grade active pharmaceutical ingredients. This limitation forces many producers to either accept lower quality batches or invest in expensive and time-consuming purification technologies like preparative HPLC, which severely impacts the cost reduction in pharmaceutical intermediates manufacturing and complicates the commercial scale-up of complex vitamin derivatives.
The Novel Approach
The methodology disclosed in the patent fundamentally alters the purification landscape by inserting a targeted acid-solubilization step immediately after the methylation reaction and before the final calcium salification. Instead of attempting to purify the final calcium salt, which is cumbersome, the process isolates the free acid form of 5-methyltetrahydrofolic acid. By dissolving the crude intermediate in concentrated hydrochloric acid and subsequently adding purified water, the process leverages differential solubility to separate the product from contaminants. The pterin impurities, which are basic in nature, react with the hydrochloric acid to form water-soluble hydrochloride salts that remain in the supernatant. Meanwhile, the desired 5-methyltetrahydrofolic acid precipitates as an off-white solid. This elegant chemical maneuver allows for the efficient removal of degradation products through simple solid-liquid separation, such as filtration, thereby streamlining the workflow for any reliable agrochemical intermediate supplier or pharma partner looking to optimize their production lines.
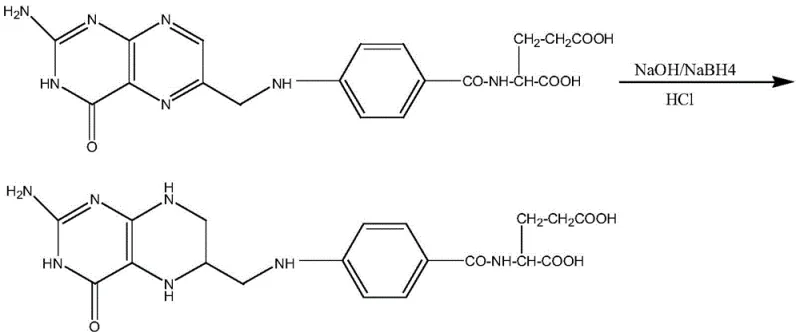
Mechanistic Insights into Acid-Solubilization Purification
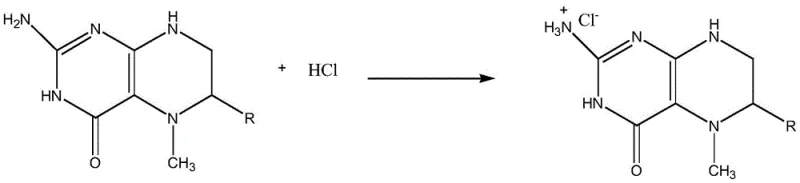
To fully appreciate the technical superiority of this method, one must understand the specific degradation pathways that occur during the initial reduction phases. As illustrated in the reaction mechanisms, the exposure of folate derivatives to strong alkaline systems triggers the cleavage of the C9-N10 bond, resulting in the generation of pterin moieties and glutamic acid derivatives. These pterin impurities are not merely inert contaminants; they possess chemical properties that can interfere with the stability and bioavailability of the final product. The innovation lies in the subsequent treatment with concentrated hydrochloric acid. Under acidic conditions, the nitrogen atoms within the pterin ring system become protonated. This protonation converts the neutral or weakly basic impurities into ionic hydrochloride salts. Unlike the target 5-methyltetrahydrofolic acid, which has limited solubility in the specific acid-water mixture used, these protonated impurities exhibit high solubility in the aqueous phase.
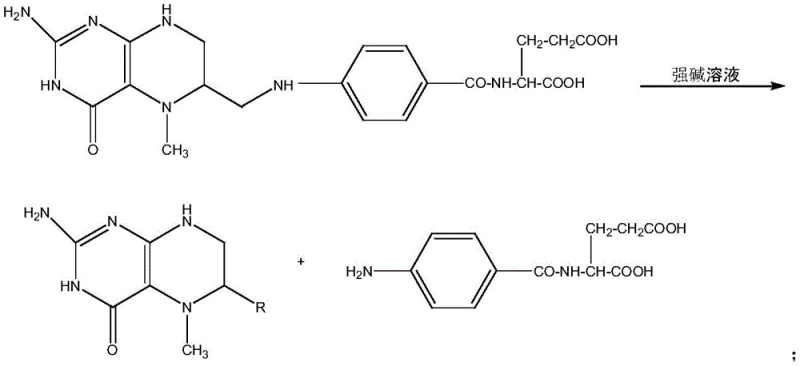
This differential solubility is the cornerstone of the purification strategy. When purified water is added to the concentrated hydrochloric acid solution containing the dissolved crude product, the dielectric constant of the medium changes, inducing the precipitation of the 5-methyltetrahydrofolic acid. Crucially, the pterin hydrochlorides remain dissolved in the mother liquor due to their ionic character and high hydration energy. This allows for a clean physical separation via filtration, effectively stripping the impurity profile of the intermediate. By removing these contaminants at the free acid stage, the subsequent salt formation reaction with calcium chloride proceeds with a much cleaner starting material. This prevents the co-crystallization of impurities into the final calcium salt lattice, ensuring that the final product consistently meets the rigorous purity specifications demanded by international pharmacopoeias. Such mechanistic control is vital for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for iterative recrystallization cycles.
How to Synthesize Calcium 5-Methyltetrahydrofolate Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield while maintaining the integrity of the folate backbone. The process begins with the careful reduction of folic acid, where temperature and pH must be tightly regulated to minimize initial degradation. Following methylation, the critical acid-solubilization step demands accurate stoichiometry regarding the ratio of concentrated hydrochloric acid to purified water to ensure optimal precipitation kinetics. The detailed operational procedures involve specific mass ratios and temperature ramps that have been optimized to balance purity gains against yield losses. For process engineers and technical teams looking to adopt this methodology, adhering to the standardized protocol is essential for reproducibility. The following guide outlines the critical operational milestones derived from the patent data, providing a roadmap for scaling this chemistry from laboratory benchtop to industrial reactor vessels.
- Reduce folic acid to tetrahydrofolic acid using sodium borohydride in alkaline solution at 60-90°C.
- Methylate tetrahydrofolic acid with formaldehyde and sodium borohydride to form crude 5-methyltetrahydrofolic acid.
- Purify the intermediate by dissolving in concentrated hydrochloric acid and precipitating with purified water to remove pterin impurities.
- React the purified intermediate with calcium chloride and L-ascorbic acid to form the final calcium salt product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented purification method offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. By replacing complex chromatographic separations or multiple recrystallization steps with a single, robust acid-base precipitation, the overall processing time is significantly compressed. This efficiency translates directly into enhanced throughput capacity, allowing manufacturers to respond more agilely to market fluctuations and urgent orders. Furthermore, the reliance on common, inexpensive reagents like hydrochloric acid and water, rather than exotic solvents or expensive resins, contributes to substantial cost savings in raw material procurement. The robustness of the method also implies a lower risk of batch failure, which is a critical metric for maintaining supply continuity in the highly regulated pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of expensive purification technologies and the reduction in solvent consumption directly lower the cost of goods sold. By achieving high purity through simple precipitation, manufacturers avoid the capital expenditure associated with advanced separation equipment and the operational costs of running energy-intensive processes. This economic efficiency allows for more competitive pricing structures without compromising on quality standards, making it an attractive option for cost-sensitive markets.
- Enhanced Supply Chain Reliability: Consistency is the currency of the supply chain. This method's ability to consistently produce intermediates with purity greater than 98 percent ensures that downstream API synthesis is not delayed by out-of-specification raw materials. The predictability of the yield and quality reduces the safety stock requirements and minimizes the risk of production stoppages, thereby strengthening the overall resilience of the supply network against disruptions.
- Scalability and Environmental Compliance: The process utilizes aqueous systems and avoids the generation of hazardous organic waste streams associated with solvent-heavy purification methods. This aligns well with modern green chemistry principles and environmental regulations, simplifying waste disposal and reducing the environmental footprint of the manufacturing facility. The simplicity of the unit operations—dissolution, precipitation, and filtration—makes the process inherently scalable, facilitating a smooth transition from pilot plant to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of calcium 5-methyltetrahydrofolate using this advanced purification technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical implications of the method. Understanding these nuances helps stakeholders make informed decisions regarding vendor selection and process validation.
Q: Why is acid-solubilization purification critical for 5-MTHF-Ca quality?
A: Standard borohydride reduction creates a highly alkaline environment that degrades folates into pterin impurities. Acid-solubilization converts these impurities into water-soluble hydrochlorides, allowing them to be washed away while the pure product precipitates.
Q: How does this method improve supply chain reliability for API manufacturers?
A: By ensuring intermediate purity exceeds 98% before the final salt formation step, this method drastically reduces the risk of batch rejection and rework, leading to more consistent lead times and reliable inventory availability.
Q: What are the cost advantages of this purification technique?
A: Eliminating complex chromatographic purification steps in favor of simple acid-base precipitation significantly lowers processing costs and solvent consumption, offering substantial economic benefits for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Calcium 5-Methyltetrahydrofolate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of pH and temperature required for this synthesis is maintained at every scale. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the absence of pterin impurities down to trace levels. We understand that for your applications, whether in nutraceuticals or prescription medicines, consistency is non-negotiable, and our quality management systems are designed to deliver exactly that level of assurance.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer picture of the economic advantages tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the superior quality of our calcium 5-methyltetrahydrofolate against your internal standards before committing to large-scale orders.
