Advanced Synthesis of Benzo[e][1,3]oxazine-2,4-dione for High-Purity SNAC Manufacturing
The pharmaceutical industry's relentless pursuit of safer and more efficient delivery systems for peptide therapeutics has placed significant spotlight on Sodium 8-(2-hydroxybenzamido)octanoate, widely known as SNAC. As a critical absorption enhancer capable of facilitating the oral bioavailability of macromolecules like insulin and heparin without dosage form protection, the demand for its key precursor, benzo[e][1,3]oxazine-2,4-dione, is escalating rapidly. Patent CN114195730A introduces a transformative preparation method that fundamentally alters the synthetic landscape by replacing hazardous carbonylating agents with di-tert-butyl dicarbonate (Boc2O). This innovation not only addresses the severe safety concerns associated with traditional phosgene-based routes but also delivers a robust, scalable process yielding products with purity exceeding 98%. The strategic shift towards this benign reagent profile represents a pivotal advancement for manufacturers aiming to secure a sustainable supply chain for high-value peptide delivery intermediates.
![Downstream application of benzo[e][1,3]oxazine-2,4-dione in the synthesis of SNAC acid and its sodium salt](/insights/img/benzo-oxazine-dione-boc-synthesis-pharma-supplier-20260309052322-06.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzo[e][1,3]oxazine-2,4-dione has relied heavily on aggressive and highly regulated reagents such as phosgene, ethyl chloroformate, or N,N'-carbonyldiimidazole (CDI). These conventional pathways present substantial operational hurdles; phosgene is a notorious chemical warfare agent requiring extreme containment measures, while ethyl chloroformate poses significant toxicity risks and lachrymatory effects that complicate worker safety protocols. Furthermore, methods utilizing CDI, although less acutely toxic, generate imidazole as a byproduct, which creates environmental disposal challenges and necessitates complex purification steps to meet stringent pharmaceutical impurity profiles. The logistical burden of transporting and storing these controlled substances often leads to supply chain fragility and inflated raw material costs, rendering these traditional routes increasingly obsolete for modern, large-scale industrial production.
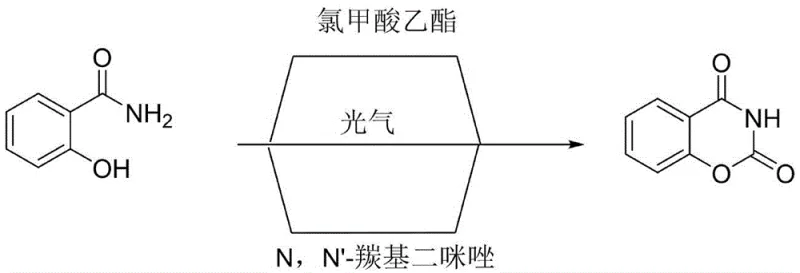
The Novel Approach
In stark contrast, the methodology disclosed in CN114195730A leverages di-tert-butyl dicarbonate (Boc2O) as a superior carbonylation cyclization reagent, effectively circumventing the hazards of its predecessors. This novel approach operates under mild alkaline conditions where 2-hydroxybenzamide reacts with Boc2O in the presence of a pyridine-based catalyst or potassium carbonate. The process is characterized by its operational simplicity and the absence of strong pungent odors or acute human toxicity, allowing for unrestricted transportation and usage without the heavy regulatory overhead associated with phosgene derivatives. By enabling a clean conversion through a stable tert-butyl ester intermediate, this route ensures high atom economy and simplifies the downstream workup, ultimately delivering the target heterocyclic scaffold with exceptional efficiency and minimal environmental footprint.
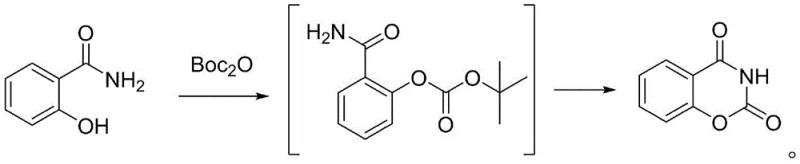
Mechanistic Insights into Boc2O-Mediated Carbonylation Cyclization
The chemical elegance of this synthesis lies in its two-stage mechanistic progression, which ensures high selectivity and minimizes side reactions. Initially, under basic catalysis at temperatures ranging from 20°C to 50°C, the phenolic hydroxyl group of 2-hydroxybenzamide undergoes nucleophilic attack on the carbonyl carbon of Boc2O, forming a reactive tert-butyl carbonate intermediate. This step is crucial as it activates the molecule for the subsequent ring-closing event without degrading the sensitive amide functionality. The reaction kinetics are carefully managed to ensure complete consumption of the starting material before thermal energy is applied, preventing the formation of polymeric byproducts or incomplete conversions that could plague less controlled exothermic reactions.
Upon heating the reaction mixture to between 70°C and 100°C, the second stage commences, driven by an intramolecular nucleophilic attack. The nitrogen atom of the amide group attacks the adjacent carbonyl carbon, forming a tetrahedral transition state that subsequently collapses with the elimination of tert-butanol. This proton-transfer-mediated cyclization results in the formation of the stable six-membered oxazine-2,4-dione ring system. The mechanism avoids the generation of harsh acidic byproducts like HCl, which are common in chloroformate routes, thereby preserving the integrity of the product and reducing corrosion risks in reactor vessels. This clean mechanistic pathway is the cornerstone of the method's ability to achieve purities greater than 98% with yields consistently around 80%.
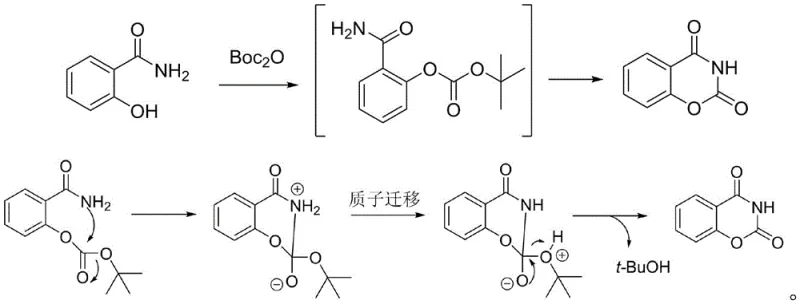
How to Synthesize Benzo[e][1,3]oxazine-2,4-dione Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the formation of the cyclic product while minimizing hydrolysis of the reagents. The process typically begins by dissolving 2-hydroxybenzamide in a polar aprotic solvent such as N,N-Dimethylformamide (DMF) or N,N-Dimethylacetamide (DMAc), followed by the addition of a catalytic amount of pyridine or DMAP. The careful addition of Boc2O at ambient temperature allows for the controlled formation of the intermediate, which can then be subjected to elevated temperatures to drive the cyclization to completion. For detailed standardized operating procedures including specific molar ratios, stirring rates, and filtration protocols, please refer to the technical guide below.
- React 2-hydroxybenzamide with di-tert-butyl dicarbonate (Boc2O) in an organic solvent like DMF using a pyridine-based catalyst at 20-50°C to form a tert-butyl ester intermediate.
- Heat the reaction mixture to 70-100°C to facilitate intramolecular nucleophilic cyclization, converting the intermediate into the target benzo[e][1,3]oxazine-2,4-dione.
- Cool the solution, adjust pH to 1-2 with acid, filter the precipitated white solid, wash with dilute acid, and dry to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the adoption of this Boc2O-based methodology offers profound advantages that extend far beyond simple chemical efficacy. The primary value proposition lies in the drastic simplification of logistics and hazard management; by eliminating the need for phosgene or ethyl chloroformate, companies can bypass the rigorous security clearances, specialized storage facilities, and expensive insurance premiums required for handling scheduled toxic substances. This shift not only de-risks the manufacturing site but also broadens the pool of potential contract manufacturing organizations (CMOs) capable of executing the synthesis, as fewer facilities possess the infrastructure to handle extreme toxins. Consequently, this leads to a more resilient supply chain with reduced vulnerability to regulatory shutdowns or transportation bottlenecks.
- Cost Reduction in Manufacturing: The economic implications of switching to Boc2O are significant, primarily driven by the reduction in safety infrastructure costs and waste treatment expenses. Unlike CDI, which is notoriously expensive and generates stoichiometric amounts of imidazole waste that requires costly removal, Boc2O is a commodity reagent with a stable global supply and lower unit cost. Furthermore, the ability to run the reaction in a one-pot sequence without isolating the intermediate reduces solvent consumption, labor hours, and equipment occupancy time, collectively driving down the cost of goods sold (COGS) for the final API intermediate without compromising quality.
- Enhanced Supply Chain Reliability: Supply continuity is markedly improved because Boc2O is not subject to the same stringent public security regulations as phosgene or ethyl chloroformate, meaning raw material lead times are shorter and more predictable. Manufacturers are no longer beholden to a limited number of specialized suppliers who hold licenses for toxic gas handling, allowing for diversified sourcing strategies that mitigate the risk of single-point failures. This accessibility ensures that production schedules for critical peptide absorption enhancers can be maintained consistently, supporting the reliable delivery of finished drug products to the market.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently greener, generating tert-butanol as the primary organic byproduct rather than corrosive acids or persistent nitrogenous wastes. This simplifies effluent treatment processes and reduces the environmental compliance burden, making it easier to scale production from pilot batches to multi-ton commercial campaigns. The robustness of the reaction conditions, which tolerate standard stainless steel reactors without requiring exotic lining materials resistant to phosgene corrosion, further facilitates rapid capacity expansion to meet growing market demand for SNAC-based formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in patent CN114195730A, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning technology transfers and for procurement officers negotiating long-term supply agreements.
Q: Why is Boc2O preferred over phosgene for synthesizing benzo[e][1,3]oxazine-2,4-dione?
A: Boc2O eliminates the severe toxicity and regulatory restrictions associated with phosgene and ethyl chloroformate, offering a safer, non-pungent alternative that simplifies transportation and industrial handling while maintaining high yields.
Q: What is the typical purity and yield achievable with this novel method?
A: According to patent CN114195730A, the process consistently achieves a product purity exceeding 98% (often reaching 99%) with isolated yields around 80%, making it highly suitable for pharmaceutical grade applications.
Q: Can this synthesis be performed as a one-pot reaction?
A: Yes, the patent describes a viable one-pot non-separation process where the intermediate tert-butyl ester is directly heated in the same solvent system to effect cyclization, significantly reducing processing time and solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo[e][1,3]oxazine-2,4-dione Supplier
As the global demand for oral peptide delivery systems surges, securing a dependable source of high-quality intermediates like benzo[e][1,3]oxazine-2,4-dione is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to ensure your supply needs are met with precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of SNAC intermediate we produce meets the exacting standards required for clinical and commercial API synthesis.
We invite you to collaborate with our technical team to explore how this advanced Boc2O-mediated synthesis can optimize your specific project requirements. By engaging with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of adopting this safer, more economical route. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments tailored to your volume and timeline expectations.
