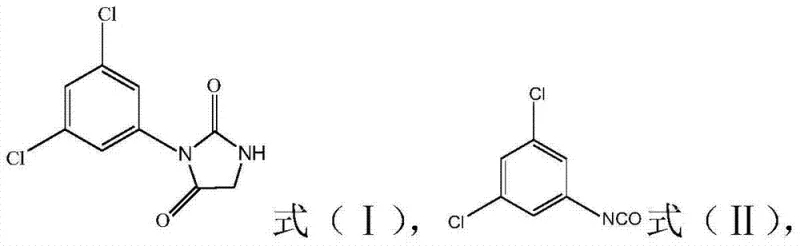
Scalable Production of 3-(3,5-Dichlorophenyl)-2,4-Imidazolidinedione via Optimized Condensation and Cyclization
The pharmaceutical and agrochemical industries constantly seek robust synthetic routes that balance high purity with economic efficiency, particularly for critical intermediates like 3-(3,5-dichlorophenyl)-2,4-imidazolidinedione. Patent CN103539744A discloses a breakthrough methodology for preparing this key structural motif, which serves as the essential precursor for the broad-spectrum fungicide RP-26019. This innovation addresses longstanding inefficiencies in heterocyclic synthesis by streamlining the transition from acyclic urea derivatives to the final imidazolidinedione ring system. By leveraging a telescoped two-step sequence that eliminates the need for intermediate isolation, the technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity agrochemical intermediates. The process demonstrates exceptional adaptability, utilizing readily available glycine esters and achieving yields that significantly surpass traditional alkaline cyclization methods, thereby establishing a new benchmark for reliability in fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-phenyl-2,4-imidazolidinedione derivatives has relied heavily on alkaline conditions, often employing bases such as triethylamine, potassium carbonate, or potassium hydroxide to facilitate ring closure. As documented in prior art like US2004/0009998A1 and literature by Kennedy et al., these conventional routes typically necessitate the rigorous separation and purification of the intermediate urea adduct before cyclization can occur. This isolation step is not only labor-intensive but also introduces significant opportunities for yield loss, with reported efficiencies often stagnating around 80% or lower. Furthermore, the requirement for multiple solvent exchanges and purification stages inflates the environmental footprint and operational costs, creating bottlenecks for producers seeking cost reduction in agrochemical manufacturing. The sensitivity of isocyanate starting materials to moisture in these prolonged multi-step processes further complicates quality control, often leading to inconsistent batch-to-batch performance.
The Novel Approach
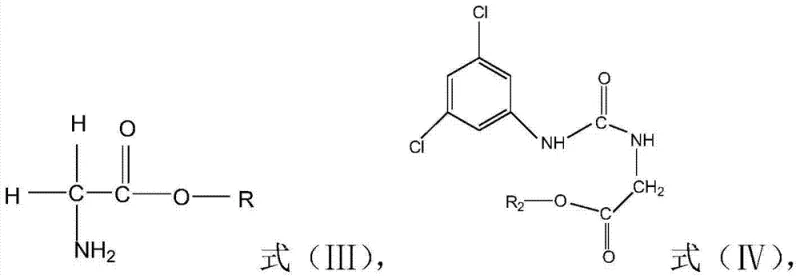
In stark contrast, the methodology outlined in CN103539744A introduces a streamlined acid-catalyzed cyclization strategy that fundamentally redefines the process flow. By reacting glycine ester derivatives (Formula III) directly with 3,5-dichlorophenyl isocyanate (Formula II) in a condensation step, the process generates the urea intermediate (Formula IV) in situ. Crucially, instead of isolating this intermediate, the reaction mixture is directly subjected to ring-closing conditions using a catalyst such as sulfuric acid. This telescoped approach allows the reaction system from the first step to be utilized directly in the second, eliminating the need for additional solvent additions or complex workup procedures between stages. The result is a drastic simplification of the operational workflow, where the final product can be obtained simply by cooling, filtering, and washing, achieving yields as high as 85.5% with purity exceeding 98%. This shift from isolation-dependent synthesis to a continuous one-pot operation represents a significant technological leap for reliable agrochemical intermediate suppliers.
Mechanistic Insights into Acid-Catalyzed Cyclization and Impurity Control
The core of this synthetic advancement lies in the precise manipulation of reaction equilibria and the strategic selection of catalytic conditions. In the initial condensation phase, the nucleophilic amino group of the glycine ester hydrochloride attacks the electrophilic carbon of the isocyanate group in Formula II. This step is facilitated by a base like triethylamine, which neutralizes the hydrochloric acid salt to liberate the free amine while scavenging the proton released during urea formation. The subsequent cyclization is driven by the addition of a strong acid catalyst, such as sulfuric acid, which activates the carbonyl oxygen of the urea moiety towards intramolecular nucleophilic attack by the adjacent ester carbonyl or amide nitrogen, depending on the specific tautomeric pathway. Simultaneously, the process employs azeotropic distillation to remove methanol, a byproduct of the cyclization, effectively shifting the equilibrium towards the formation of the imidazolidinedione ring according to Le Chatelier's principle. This mechanistic understanding is vital for R&D directors focusing on the commercial scale-up of complex polymer additives or agrochemical intermediates, as it highlights the importance of thermal management and byproduct removal.
Impurity control is another critical aspect where this patent demonstrates superior engineering. A major challenge in isocyanate chemistry is the susceptibility of the -NCO group to hydrolysis, which can generate unwanted urea byproducts or amines that degrade product quality. The patent explicitly mandates maintaining the water content in the reaction system below 0.5% by weight during the condensation step. This stringent control is achieved through the use of anhydrous solvents like toluene or chlorobenzene and potentially incorporating drying apparatuses such as oil-filled bubblers or desiccant tubes. By minimizing water presence, the process ensures that the 3,5-dichlorophenyl isocyanate reacts exclusively with the glycine ester rather than undergoing hydrolytic decomposition. This attention to detail in moisture management directly translates to a cleaner crude product profile, reducing the burden on downstream purification and ensuring that the final active pharmaceutical ingredient or agrochemical precursor meets rigorous specification standards without extensive recrystallization.
How to Synthesize 3-(3,5-Dichlorophenyl)-2,4-Imidazolidinedione Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and thermal profiles to maximize the benefits of the telescoped design. The process begins with the preparation of the isocyanate starting material, typically generated from 3,5-dichloroaniline and solid phosgene in a separate upstream unit, ensuring a steady supply of high-quality Formula II. Once the isocyanate solution is ready, it is charged into a reactor with a suitable organic solvent, and the glycine ester hydrochloride is introduced along with a tertiary amine base. The mixture is heated to approximately 80°C to drive the condensation to completion, monitored by the disappearance of the isocyanate peak. Without isolating the resulting urea intermediate, sulfuric acid is added directly to the same vessel, and the temperature is maintained to facilitate cyclization while stripping off the alcohol byproduct. The detailed standardized synthesis steps, including specific molar ratios, heating rates, and filtration protocols, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Condense glycine methyl ester hydrochloride with 3,5-dichlorophenyl isocyanate in toluene using triethylamine at 80°C.
- Directly add sulfuric acid to the reaction mixture to catalyze ring closure while removing methanol azeotropically.
- Cool the mixture, filter the solid product, wash to neutrality, and dry to obtain high-purity imidazolidinedione.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers transformative potential in terms of operational expenditure and logistical stability. The most immediate impact is seen in the reduction of processing time and resource consumption; by eliminating the isolation and purification of the intermediate urea derivative, manufacturers can significantly cut down on solvent usage, energy consumption for drying, and labor hours associated with filtration and transfer operations. This streamlining of the workflow directly contributes to substantial cost savings in production, allowing companies to remain competitive in the global market for fungicide intermediates. Furthermore, the reliance on commodity chemicals such as glycine methyl ester hydrochloride and toluene ensures that raw material sourcing remains stable and unaffected by the volatility often associated with specialized reagents. This accessibility enhances supply chain reliability, reducing the risk of production stoppages due to material shortages.
- Cost Reduction in Manufacturing: The elimination of the intermediate isolation step is the primary driver for cost optimization in this process. Traditional methods require distinct unit operations for separating the urea adduct, which involves additional solvent volumes, filtration equipment capacity, and drying time. By telescoping the condensation and cyclization into a single reaction vessel, the process reduces the overall cycle time and minimizes the physical footprint required for production. Additionally, the high yield of over 85% means that less raw material is wasted per kilogram of final product, further driving down the cost of goods sold. The use of sulfuric acid as a cyclization catalyst is also economically advantageous compared to more expensive or hazardous alternatives, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route is bolstered by the use of widely available and stable starting materials. Glycine esters and 3,5-dichloroaniline derivatives are produced on a large industrial scale, ensuring a consistent supply stream that is less prone to disruption. The process tolerance for slight variations in reaction conditions, combined with the simplicity of the workup (direct filtration), makes it highly suitable for multi-purpose plants that may need to switch between different campaigns. This flexibility allows supply chain heads to plan production schedules with greater confidence, knowing that the lead time for high-purity agrochemical intermediates can be minimized without compromising on quality or yield consistency.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns well with modern green chemistry principles. The reduction in solvent usage and the avoidance of complex extraction procedures lower the volume of waste streams generated, simplifying wastewater treatment and disposal. The ability to run the reaction in common solvents like toluene, which can be easily recovered and recycled via distillation, further enhances the sustainability profile. For facilities aiming to scale up from pilot batches to commercial tonnage, the straightforward nature of the reaction—requiring only standard heating and stirring equipment—lowers the barrier to entry and reduces the capital investment needed for specialized reactors, making it an ideal candidate for rapid capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing a clear understanding of the operational parameters and benefits. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing lines or for procurement specialists assessing the long-term viability of the supply source.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: The primary advantage is the elimination of the intermediate isolation step. Conventional methods require separating and purifying the urea derivative (Formula IV), which increases solvent usage and processing time. This patent allows for a direct one-pot cyclization, significantly simplifying the workflow and improving overall yield to over 85%.
Q: How is moisture control managed during the condensation step?
A: Moisture control is critical because 3,5-dichlorophenyl isocyanate is highly sensitive to hydrolysis. The process mandates that the water content in the reaction system be maintained below 0.5% by weight. This is achieved through the use of anhydrous solvents like toluene and potentially adding drying agents or using azeotropic distillation to ensure the integrity of the isocyanate reactant.
Q: Can this process be scaled for industrial production of fungicide intermediates?
A: Yes, the process is highly suitable for industrial scale-up. It utilizes common organic solvents like toluene and chlorobenzene, and the workup involves simple filtration rather than complex chromatography or extraction. The use of inexpensive raw materials like glycine methyl ester hydrochloride further supports cost-effective large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(3,5-Dichlorophenyl)-2,4-Imidazolidinedione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of final agrochemical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3-(3,5-dichlorophenyl)-2,4-imidazolidinedione meets the exacting standards required by global regulatory bodies. Our infrastructure is designed to support the complex demands of modern chemical synthesis, providing a secure and dependable foundation for your supply chain.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your specific application needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your bottom line. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing you to make informed decisions based on transparent and verified performance metrics. Let us be your partner in driving efficiency and quality in the production of essential agrochemical intermediates.
