Advanced Synthesis of N-Alkynylthio Phthalimides for High-Purity Pharmaceutical Intermediates
Introduction to Breakthrough Alkynylthio Transfer Technology
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more versatile building blocks that can access complex molecular architectures with greater efficiency. A pivotal advancement in this domain is detailed in Chinese Patent CN110066237B, which discloses a novel preparation method for N-alkynylthio phthalimide compounds. These compounds serve as powerful transfer reagents, capable of introducing the valuable alkynylthio functional group into a wide array of organic molecules. The significance of the alkynylthio moiety cannot be overstated; beyond its utility as a precursor to alkynyl sulfones or sulfoxides, the conjugation effect of the sulfur atom significantly enhances the reactivity of the adjacent carbon-carbon triple bond. This unique electronic profile allows for diverse downstream transformations, including various couplings, reductions, and oxidative conversions, making it an indispensable tool for constructing heterocyclic skeletons and modifying peptides.
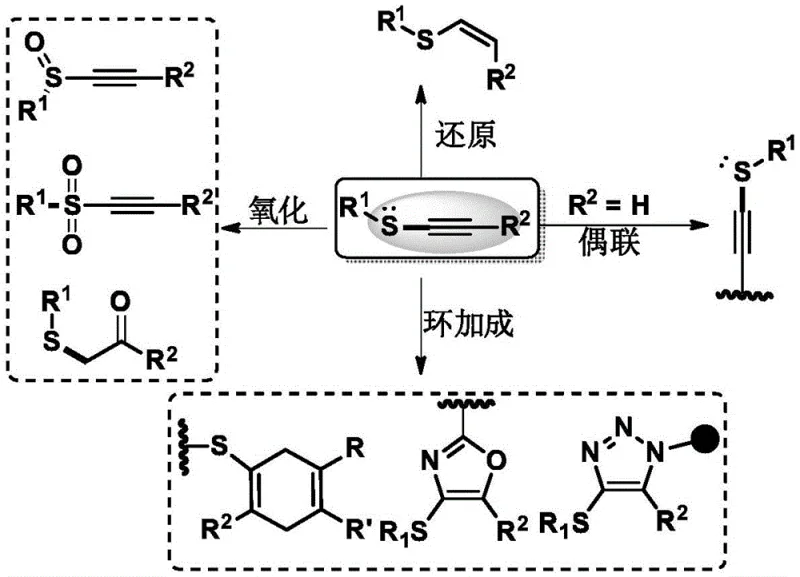
For research and development directors overseeing the synthesis of active pharmaceutical ingredients (APIs) or advanced agrochemical intermediates, the ability to install this functionality without pre-existing thiols or alkynes in the substrate is a game-changer. Traditionally, accessing such structures required specific starting materials that limited the scope of accessible chemical space. This patent introduces a robust methodology that bypasses these constraints, offering a reliable pathway for the commercial scale-up of complex pharmaceutical intermediates. By leveraging this technology, manufacturers can unlock new avenues for drug discovery and process optimization, ensuring a steady supply of high-purity intermediates that meet the rigorous standards of the global pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the introduction of alkynylthio groups into organic molecules was fraught with significant synthetic challenges that often hindered process scalability and substrate scope. Conventional strategies primarily relied on two distinct pathways, both of which imposed strict structural requirements on the starting materials. The first approach involved utilizing substrates that already contained a sulfhydryl (-SH) group, reacting them with alkynyl metal reagents or alkynyl cation intermediates. This necessitated the prior installation of the thiol functionality, which could be synthetically tedious and incompatible with other sensitive functional groups present in complex drug candidates.
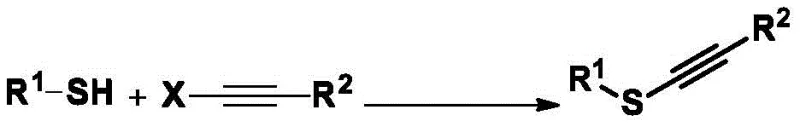
The second conventional method utilized terminal alkynes as the starting material, reacting them with activated thiol cation intermediates. Similarly, this approach mandated the presence of a terminal alkyne in the substrate, limiting the diversity of molecules that could be modified. Consequently, for organic molecules that lacked both mercapto and alkynyl groups—a common scenario in late-stage functionalization of bioactive scaffolds—existing technology offered no viable solution. These limitations created a bottleneck in the supply chain for specialized intermediates, forcing procurement managers to seek costly custom synthesis routes or accept lower yields and purity profiles associated with multi-step workaround strategies.
The Novel Approach
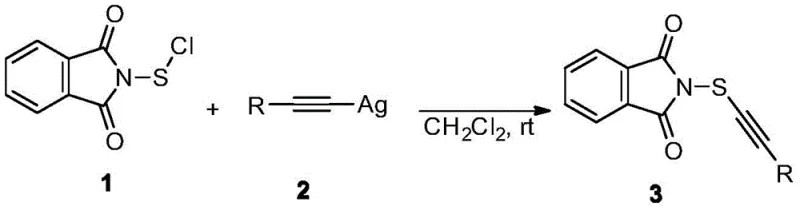
The methodology described in patent CN110066237B represents a paradigm shift by decoupling the source of the alkynylthio group from the structural constraints of the target substrate. Instead of relying on the substrate to provide the reactive handle, this novel approach utilizes a dedicated transfer reagent: the N-alkynylthio phthalimide. This reagent acts as a stable yet reactive reservoir of the alkynylthio moiety, capable of transferring the group to a broad spectrum of organic molecules regardless of their initial functionalization. The synthesis of this key reagent itself is remarkably straightforward, involving the reaction of phthalimide sulfur chloride with an alkynyl silver reagent. This process avoids the harsh conditions often associated with generating reactive sulfur species, operating instead under mild, neutral conditions that preserve the integrity of sensitive functional groups.
By employing this transfer reagent strategy, chemists can now access alkynylthio-substituted architectures that were previously inaccessible or economically unviable. This flexibility is crucial for cost reduction in fine chemical manufacturing, as it reduces the number of synthetic steps required to reach the target molecule. Furthermore, the simplicity of the reagent preparation—dissolving, mixing, filtering, and recrystallizing—translates directly into operational efficiency. For supply chain heads, this means a more robust and predictable production schedule, as the reliance on exotic or difficult-to-handle starting materials is minimized. The ability to generate this versatile reagent from cheap and readily available raw materials ensures a continuous supply, mitigating the risks associated with raw material shortages that often plague the specialty chemical sector.
Mechanistic Insights into Silver-Mediated Alkynylthio Formation
The core of this technological breakthrough lies in the efficient formation of the sulfur-alkyne bond through a silver-mediated substitution mechanism. The reaction initiates with the dissolution of phthalimide sulfur chloride in dichloromethane (DCM) under a nitrogen atmosphere. The choice of DCM as the solvent is critical; it provides excellent solubility for both the sulfur chloride electrophile and the resulting phthalimide product while remaining inert under the reaction conditions. The nitrogen protection is equally vital, preventing the oxidation of the sensitive sulfur species and ensuring the stability of the alkynyl silver reagent, which can be susceptible to decomposition in the presence of moisture or oxygen. Upon the addition of the alkynyl silver reagent, a nucleophilic attack occurs where the alkynyl group effectively displaces the chloride on the sulfur atom.
A distinctive feature of this mechanism is the immediate formation of a white precipitate, identified as silver chloride (AgCl). This precipitation serves as a thermodynamic driving force for the reaction, pulling the equilibrium towards the formation of the desired N-alkynylthio phthalimide product. From a process chemistry perspective, this is highly advantageous because the byproduct is insoluble and can be easily removed via simple suction filtration. This eliminates the need for complex aqueous workups or chromatographic purification at the reagent synthesis stage, significantly streamlining the workflow. The filtrate, containing the crude product, is then evaporated and concentrated. The final purification step involves recrystallization using chloroform, which yields the target compound with high purity, suitable for subsequent sensitive coupling reactions in API synthesis.
The structural versatility of this method is underpinned by the tolerance of the alkynyl silver reagent towards various substituents. As demonstrated in the patent examples, the 'R' group on the alkyne can range from simple phenyl rings to more complex naphthalene systems, thiophene heterocycles, and even aliphatic chains containing azide groups. This broad scope indicates that the electronic nature of the alkyne—whether electron-withdrawing or electron-donating—does not significantly impede the reaction efficiency. For R&D teams, this implies that a single standardized protocol can be applied to synthesize a library of diverse alkynylthio transfer reagents. This modularity accelerates the screening process for new drug candidates, allowing for rapid iteration and optimization of lead compounds without the need to develop bespoke synthetic routes for each new analog.
How to Synthesize N-Alkynylthio Phthalimide Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure maximum yield and safety. The process is designed to be user-friendly, minimizing the need for specialized equipment beyond standard glassware and filtration setups. The key to success lies in maintaining anhydrous conditions and controlling the addition rate of reagents to manage the exotherm, although the reaction is generally mild. The following guide outlines the standardized procedure derived from the patent data, serving as a foundational protocol for producing this high-value intermediate.
- Dissolve phthalimide sulfur chloride in dichloromethane under a nitrogen atmosphere to ensure anhydrous and oxygen-free conditions.
- Add the alkynyl silver reagent to the solution in one portion while stirring uniformly, observing the formation of a white precipitate.
- Continue stirring for 1 to 4 hours at room temperature, then filter to remove the precipitate, concentrate the filtrate, and recrystallize with chloroform.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this synthesis method offers tangible benefits that extend beyond mere chemical novelty. The primary value proposition is the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures. Unlike traditional methods that might require cryogenic temperatures, expensive transition metal catalysts, or hazardous reagents, this process operates at ambient temperature (optimally 25°C) using common solvents like dichloromethane. The elimination of energy-intensive heating or cooling cycles results in significant cost reduction in fine chemical manufacturing. Furthermore, the workup procedure is exceptionally clean; the removal of the silver chloride byproduct via filtration avoids the generation of large volumes of aqueous waste, simplifying effluent treatment and enhancing environmental compliance.
- Cost Reduction in Manufacturing: The economic efficiency of this route is driven by the use of cheap and readily available raw materials, specifically phthalimide sulfur chloride and alkynyl silver reagents. The stoichiometric nature of the reaction, combined with the ease of product isolation through recrystallization, minimizes material loss and maximizes throughput. There is no need for expensive chromatographic purification on a commercial scale, as the recrystallization step provides sufficient purity for most downstream applications. This streamlined workflow reduces labor costs and equipment occupancy time, allowing facilities to produce higher volumes of high-purity pharmaceutical intermediates with the same infrastructure.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on niche or unstable starting materials. This method mitigates such risks by utilizing robust reagents that are stable under standard storage conditions. The broad substrate scope means that a single manufacturing line can be adapted to produce various derivatives simply by changing the alkynyl silver component, providing flexibility to respond to shifting market demands. Additionally, the mild reaction conditions reduce the risk of batch failures due to thermal runaways or sensitivity issues, ensuring consistent delivery schedules for clients relying on these critical building blocks for their own production timelines.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the ton-scale often introduces unforeseen challenges, but this methodology is inherently scalable. The reaction does not involve gaseous reagents or high-pressure vessels, making it suitable for standard stirred-tank reactors. The solid byproduct (AgCl) can potentially be recovered and recycled for silver value, further improving the economic and environmental footprint. The use of chloroform for recrystallization, while requiring careful handling, is a well-established industrial solvent with established recovery protocols. This alignment with green chemistry principles—specifically waste minimization and energy efficiency—positions manufacturers favorably against increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its integration into their supply chain. The following questions address common inquiries regarding the practical application, scope, and benefits of this technology, based on the detailed experimental data provided in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this novel route for producing alkynylthio-functionalized intermediates.
Q: What is the primary advantage of this N-alkynylthio phthalimide synthesis method?
A: The primary advantage is the ability to introduce alkynylthio groups into complex organic molecules that lack both mercapto and alkynyl functional groups, overcoming significant limitations of conventional synthetic routes.
Q: What are the optimal reaction conditions for this transformation?
A: The reaction proceeds optimally at room temperature (25°C) in dichloromethane with a molar ratio of 1:1 between phthalimide sulfur chloride and the alkynyl silver reagent, typically completing within 2 hours.
Q: What types of substituents are tolerated in the alkynyl silver reagent?
A: The method exhibits broad substrate scope, tolerating aromatic rings with electron-withdrawing or donating groups, naphthalene rings, thiophene heterocycles, cyclic aliphatic alkanes, and even azide-containing aliphatic chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkynylthio Phthalimide Supplier
As the demand for specialized fine chemical intermediates continues to grow, partnering with a manufacturer that possesses deep technical expertise and scalable capacity is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN110066237B to deliver superior value to our global clientele. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-alkynylthio phthalimide meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to optimize your supply chain and accelerate your product development cycles. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, identifying opportunities to reduce expenses through process optimization and bulk sourcing. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and reliability can support your long-term strategic goals in the competitive landscape of fine chemical manufacturing.
