Scalable Manufacturing of 7,8-Dihydroxyflavone: A Technical Breakthrough for Neuroprotective Drug Development
Scalable Manufacturing of 7,8-Dihydroxyflavone: A Technical Breakthrough for Neuroprotective Drug Development
The pharmaceutical industry is witnessing a surge in demand for neuroprotective agents, with 7,8-dihydroxyflavone emerging as a potent TrkB receptor agonist capable of mimicking brain-derived neurotrophic factor. Addressing the critical need for reliable supply chains, the methodology detailed in patent CN114276321A presents a robust, scalable route for synthesizing this high-value intermediate. Unlike traditional approaches that often struggle with regioselectivity and purification bottlenecks, this novel process leverages a strategic combination of Friedel-Crafts acylation and selective benzyl protection to ensure high purity and yield. For R&D directors and procurement specialists alike, understanding the nuances of this synthetic pathway is essential for securing a stable supply of this complex flavonoid derivative.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyhydroxylated flavones has been plagued by significant challenges regarding regioselectivity and solvent compatibility. Conventional methods often rely on solvents such as dichloromethane or carbon disulfide for the initial acylation of pyrogallol; however, the poor solubility of pyrogallol in these media leads to heterogeneous reaction conditions. This heterogeneity not only complicates heat transfer and reaction control but also results in inconsistent conversion rates and difficult downstream processing. Furthermore, traditional protection strategies frequently lack the precision required to differentiate between adjacent hydroxyl groups on the aromatic ring, leading to complex mixtures of isomers that are costly and time-consuming to separate. These inefficiencies translate directly into higher production costs and extended lead times, creating substantial friction for commercial scale-up efforts.
The Novel Approach
The methodology disclosed in the referenced patent introduces a paradigm shift by optimizing solvent systems and catalytic conditions at every stage of the synthesis. By selecting acetic acid as the solvent for the initial Friedel-Crafts reaction, the process ensures a completely homogeneous reaction mixture, thereby maximizing the interaction between the substrate and the Lewis acid catalyst. The innovation extends to the protection phase, where steric hindrance is ingeniously exploited to selectively shield the 3 and 4-position hydroxyl groups while leaving the 2-position available for subsequent functionalization. This precise control eliminates the formation of unwanted isomers, streamlining the purification workflow. Ultimately, this approach transforms a historically problematic synthesis into a streamlined, commercially viable operation suitable for large-scale manufacturing.
Mechanistic Insights into Lewis Acid Catalyzed Acylation and Rearrangement
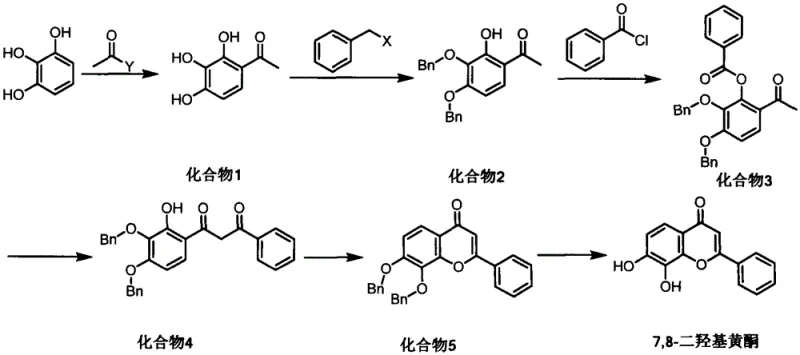
The core of this synthetic strategy lies in the meticulous control of reaction mechanisms, particularly during the initial acylation and the pivotal Baker-Venkataraman rearrangement. In the first step, the use of Lewis acids such as anhydrous stannic chloride or aluminum trichloride facilitates the electrophilic aromatic substitution of pyrogallol with acetyl chloride. Unlike protonic acids, which can lead to difficult-to-separate byproducts, Lewis acids promote the precipitation of the product directly from the reaction system, significantly simplifying isolation. The subsequent Baker-Venkataraman rearrangement is driven by a strong base system, typically potassium hydroxide in anhydrous pyridine. This step involves the migration of the acyl group to form a beta-diketone intermediate, which is the precursor to the flavone core. The strict exclusion of water in this phase is paramount, as moisture can hydrolyze the ester or deactivate the base, severely compromising the yield.
Impurity control is inherently built into the mechanistic design of this route. The selective protection step acts as a gatekeeper, ensuring that only the desired regioisomer proceeds to the esterification stage. By utilizing benzyl halides in acetone with potassium carbonate, the process capitalizes on the differential reactivity of the phenolic hydroxyls. Any unreacted starting material or over-protected species can be effectively removed during the aqueous workup or crystallization steps described in the examples. Furthermore, the final cyclization step, catalyzed by acids like sulfuric acid or trifluoroacetic acid in acetic acid, proceeds with high efficiency, minimizing the formation of open-chain byproducts. This rigorous attention to mechanistic detail ensures that the final 7,8-dihydroxyflavone meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize 7,8-Dihydroxyflavone Efficiently
Executing this synthesis requires precise adherence to the optimized conditions outlined in the patent to achieve maximum efficiency and yield. The process begins with the dissolution of pyrogallol in glacial acetic acid, followed by the controlled addition of acetyl chloride and a Lewis acid catalyst at temperatures below 35°C. Following the isolation of the trihydroxyacetophenone intermediate, the selective protection is carried out under reflux in acetone, utilizing benzyl bromide or chloride. The resulting protected intermediate is then subjected to esterification with benzoyl chloride in pyridine before undergoing the critical rearrangement and cyclization sequence. For a comprehensive breakdown of the specific molar ratios, temperature profiles, and workup procedures, please refer to the standardized protocol below.
- Perform Friedel-Crafts acylation of pyrogallol with acetyl chloride using acetic acid as solvent and Lewis acid catalyst.
- Selectively protect the 3 and 4-position hydroxyl groups using benzyl halides in acetone with potassium carbonate.
- Execute Baker-Venkataraman rearrangement followed by acid-catalyzed cyclization and final palladium-catalyzed debenzylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this optimized synthetic route offers tangible benefits that extend far beyond simple chemical yield. The elimination of heterogeneous reaction conditions in the initial step reduces the risk of batch-to-batch variability, a critical factor in maintaining supply continuity for long-term contracts. By avoiding solvents that require complex recovery systems or pose significant environmental hazards, the process aligns with modern green chemistry principles, potentially reducing waste disposal costs and regulatory burdens. The use of readily available raw materials, such as pyrogallol and acetyl chloride, ensures that the supply chain remains resilient against market fluctuations for exotic reagents. This stability is essential for securing reliable sourcing of high-purity pharmaceutical intermediates in a volatile global market.
- Cost Reduction in Manufacturing: The strategic selection of catalysts and solvents drives significant cost optimization throughout the production lifecycle. By employing Lewis acids that allow the product to precipitate directly from the reaction mixture, the need for extensive chromatographic purification is eliminated, drastically reducing solvent consumption and labor hours. Furthermore, the high selectivity of the protection step minimizes the loss of valuable starting materials to side reactions, improving the overall atom economy of the process. These cumulative efficiencies result in a substantially lower cost of goods sold, allowing for more competitive pricing structures without compromising on quality margins.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway directly translates to improved delivery performance and supply security. The reliance on commodity chemicals rather than specialized, single-source reagents mitigates the risk of supply disruptions caused by geopolitical issues or manufacturer shortages. Additionally, the simplified workup procedures reduce the turnaround time between batches, enabling manufacturers to respond more agilely to sudden increases in demand. This operational flexibility is a key differentiator for any organization seeking a dependable partner for complex fine chemical intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard unit operations and well-understood reaction engineering principles. The avoidance of highly toxic solvents like carbon disulfide simplifies the safety profile of the plant, reducing the capital expenditure required for specialized containment and ventilation systems. Moreover, the efficient use of acetic acid and ethanol in the final steps allows for easier solvent recovery and recycling, supporting sustainability goals. This alignment with environmental compliance standards ensures long-term operational viability and reduces the risk of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 7,8-dihydroxyflavone using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own drug development pipelines.
Q: Why is acetic acid preferred over dichloromethane for the initial acylation step?
A: Pyrogallol exhibits poor solubility in common organic solvents like dichloromethane or carbon disulfide. Acetic acid provides high solubility for pyrogallol without interfering with the reaction, ensuring a homogeneous reaction mixture which is critical for consistent kinetics and heat transfer.
Q: How does this process achieve regioselectivity during the protection step?
A: The process utilizes steric hindrance effects to selectively protect the 3-position and 4-position phenolic hydroxyl groups while leaving the 2-position hydroxyl group unprotected. This specific selectivity is crucial for the subsequent esterification and rearrangement steps to proceed correctly towards the flavone scaffold.
Q: What are the critical parameters for the Baker-Venkataraman rearrangement in this route?
A: The rearrangement requires a strong base system, specifically potassium hydroxide in anhydrous pyridine. Water must be strictly excluded to maintain high yield. Additionally, due to the high viscosity of the reaction mixture, mechanical stirring is essential to ensure proper mixing and heat dissipation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7,8-Dihydroxyflavone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of neuroprotective therapies depends on the availability of high-quality intermediates produced via robust and scalable routes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 7,8-dihydroxyflavone meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of flavonoid synthesis with precision and reliability.
We invite you to collaborate with us to leverage these technological advantages for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing capabilities can accelerate your time to market.
