Revolutionizing Pitavastatin Intermediate Production via Green NBS Bromination Technology
The pharmaceutical industry is constantly seeking robust synthetic pathways that balance high efficiency with stringent environmental standards. Patent CN112175007A introduces a significant advancement in the preparation of pitavastatin calcium intermediates, specifically targeting the synthesis of [[2-cyclopropyl-4-(4-fluorophenyl)-3-quinolyl]methyl]triphenylphosphonium bromide. This innovation addresses critical bottlenecks in traditional statin intermediate manufacturing by replacing hazardous phosphorus tribromide reagents with N-bromosuccinimide (NBS). For R&D directors and procurement specialists, this shift represents a move towards greener chemistry that does not compromise on yield or purity. The disclosed method achieves yields over 90% and purity levels exceeding 99%, establishing a new benchmark for reliable pharmaceutical intermediate supplier capabilities in the competitive lipid-lowering drug market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
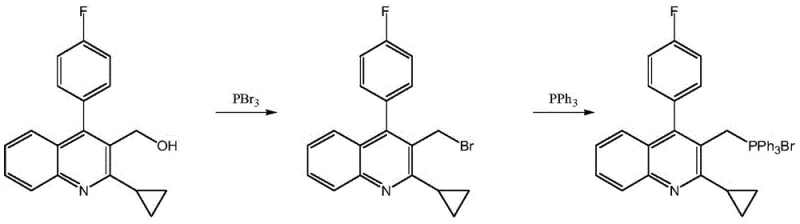
Historically, the commercial production of this key pitavastatin precursor relied heavily on phosphorus tribromide (PBr3) as the primary brominating agent. While effective in converting the hydroxymethyl group to a bromomethyl group, this conventional route suffers from severe operational and environmental drawbacks. The reaction necessitates a rigorous quenching step using alkaline water to neutralize excess phosphorus tribromide, which inevitably generates massive quantities of wastewater contaminated with both bromine and phosphorus species. Furthermore, the brominated intermediate produced in this manner is chemically sensitive; exposure to acidic or basic conditions during the quenching and washing phases often leads to partial decomposition. This instability not only lowers the overall yield but also complicates the purification process, introducing variability that is unacceptable for high-purity API manufacturing.
The Novel Approach
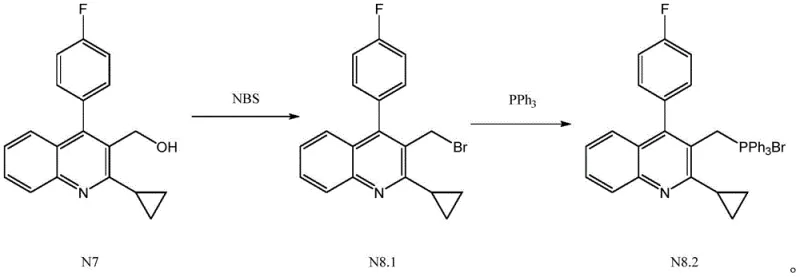
In stark contrast, the novel methodology outlined in the patent utilizes N-bromosuccinimide (NBS) to effect the bromination under much milder conditions. A defining feature of this approach is the seamless integration of the bromination and phosphonium salt formation steps into a single pot. By reacting compound N7 with NBS at controlled temperatures between 30-80°C, the bromo-intermediate N8.1 is generated in situ. Crucially, this intermediate is not isolated; instead, triphenylphosphine is added directly to the reaction mixture. This eliminates the need for the destructive alkaline quenching step entirely. The result is a streamlined process that preserves the integrity of the intermediate, prevents the formation of hazardous waste streams, and significantly simplifies the downstream workup, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into NBS-Mediated Bromination and Phosphonium Salt Formation
The core of this technological breakthrough lies in the selective reactivity of NBS towards the benzylic-like hydroxymethyl group on the quinoline ring of compound N7. Unlike PBr3, which proceeds via an SN2 mechanism often requiring strict anhydrous conditions and generating HBr gas, NBS offers a more controlled radical or ionic bromination pathway depending on the solvent and initiation conditions. In the preferred embodiment, the reaction is conducted in solvents such as toluene or dichloroethane at temperatures ranging from 50-60°C. This thermal window is critical; temperatures below 30°C result in incomplete conversion, while exceeding 80°C promotes the formation of undesired by-products. The mechanistic precision ensures that the hydroxyl group is replaced by bromine with high fidelity, setting the stage for the subsequent nucleophilic attack by triphenylphosphine.
Following the in situ generation of the bromo-intermediate, the addition of triphenylphosphine triggers a classic Menshutkin reaction, forming the quaternary phosphonium salt. Because the intermediate N8.1 is not subjected to aqueous workup, it remains stable and free from hydrolysis products that typically plague the PBr3 route. The absence of acid-base exposure during the process is a key factor in maintaining the structural integrity of the cyclopropyl and fluoro-phenyl moieties, which are sensitive to harsh conditions. This mechanistic advantage directly translates to the observed purity levels of over 99%, as the formation of degradation impurities is effectively suppressed. For technical teams, understanding this stability profile is essential for scaling up the process while maintaining strict quality control specifications.
How to Synthesize [[2-Cyclopropyl-4-(4-Fluorophenyl)-3-Quinolyl]Methyl]Triphenylphosphonium Bromide Efficiently
The synthesis protocol described in the patent offers a straightforward yet highly optimized pathway for producing this complex statin intermediate. The process begins with the suspension of compound N7 in a suitable organic solvent, followed by the controlled addition of NBS. Maintaining the reaction temperature within the 30-80°C range for a duration of 4 to 8 hours ensures complete conversion to the bromo-species. Once this first stage is complete, triphenylphosphine is introduced directly into the same vessel. The mixture is then heated to a higher temperature range of 60-120°C to drive the phosphonium salt formation to completion. The final product is recovered simply by cooling the reaction mass to 0-20°C, inducing crystallization, and filtering. This operational simplicity reduces the equipment footprint and labor intensity required for commercial scale-up of complex pharmaceutical intermediates.
- React compound N7 with NBS in a solvent like toluene at 30-80°C for 4-8 hours to form the bromo-intermediate N8.1.
- Without isolation, add triphenylphosphine directly to the reaction mixture and heat to 60-120°C for another 4-8 hours.
- Cool the solution to 0-20°C, stir to crystallize the product, and filter to obtain the high-purity phosphonium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from the PBr3 route to the NBS-based one-pot synthesis offers profound strategic benefits beyond mere chemical elegance. The most immediate impact is the drastic simplification of the waste management profile. By eliminating the generation of phosphorus and bromine-laden wastewater, manufacturers can significantly reduce the costs associated with effluent treatment and regulatory compliance. This green chemistry approach aligns perfectly with the increasing global pressure on pharmaceutical suppliers to adopt sustainable manufacturing practices. Furthermore, the removal of the isolation step for the unstable bromo-intermediate reduces the number of unit operations, thereby shortening the overall production cycle time and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of the alkaline quenching step and the subsequent aqueous washes removes the need for large volumes of water and the associated energy costs for heating and treating this water. Additionally, the high yield (>90%) and high purity (>99%) minimize the loss of valuable starting materials and reduce the burden on downstream purification processes like recrystallization or chromatography. The use of NBS, a stable and easily handled solid reagent, also simplifies inventory management compared to the corrosive and moisture-sensitive liquid PBr3, leading to substantial cost savings in logistics and storage.
- Enhanced Supply Chain Reliability: The robustness of the NBS route against variations in pH and moisture during the reaction workup enhances batch-to-batch consistency. In the traditional method, the sensitivity of the bromo-intermediate to acid-base conditions often resulted in variable yields and unpredictable quality, posing risks to supply continuity. The new method's tolerance and stability ensure that production schedules can be met with greater certainty. Moreover, the raw materials required, such as NBS and triphenylphosphine, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages that could disrupt the production of this critical pitavastatin precursor.
- Scalability and Environmental Compliance: Scaling a chemical process often amplifies safety and environmental risks, but this new methodology inherently mitigates them. The absence of HBr gas evolution and the reduction in hazardous waste volume make the process safer for operators and easier to permit in regions with strict environmental regulations. The one-pot nature of the synthesis reduces the need for multiple reactors and transfer lines, lowering the capital expenditure required for capacity expansion. This scalability ensures that suppliers can meet growing market demand for pitavastatin without compromising on their environmental commitments or facing regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NBS-based synthesis route. These insights are derived directly from the experimental data and comparative examples provided in patent CN112175007A, offering a transparent view of the technology's capabilities. Understanding these details is crucial for partners evaluating the feasibility of adopting this greener alternative for their supply chains.
Q: How does the NBS method improve environmental compliance compared to PBr3?
A: The NBS method eliminates the need for alkaline quenching required by phosphorus tribromide, thereby preventing the generation of large volumes of bromine and phosphorus-containing wastewater.
Q: What purity levels can be achieved with this new synthetic route?
A: The patent data indicates that the target intermediate can achieve a purity of over 99% with a yield exceeding 90%, significantly reducing downstream purification burdens.
Q: Why is the one-pot process advantageous for supply chain stability?
A: By avoiding the isolation of the unstable bromo-intermediate, the process reduces material loss and simplifies operations, leading to more consistent batch-to-batch quality and reliable delivery timelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable [[2-Cyclopropyl-4-(4-Fluorophenyl)-3-Quinolyl]Methyl]Triphenylphosphonium Bromide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the NBS-mediated process is key to staying competitive in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in industrial practice. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of pitavastatin intermediate supports the efficacy and safety of the final API. Our infrastructure is designed to handle complex heterocyclic chemistry with the precision required for modern drug development.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic and environmental benefits for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition to a more efficient and sustainable sourcing strategy for your statin portfolio.
