Advanced Synthesis of Aminoalkoxy Curcumin Derivatives for Oncology Applications
Introduction to Novel Curcumin Analogues
The pharmaceutical industry continuously seeks to optimize natural product scaffolds to overcome inherent pharmacokinetic limitations, and the research detailed in patent CN102503842A represents a significant advancement in this domain. Curcumin, a well-known polyphenol derived from turmeric, possesses remarkable biological activities including anti-inflammatory and anticancer properties, yet its clinical application has been severely hindered by poor water solubility and low bioavailability. This patent introduces a strategic structural modification by incorporating aminoalkoxy side chains into the aromatic rings of the curcumin parent nucleus. By addressing the solubility bottleneck through chemical design rather than formulation alone, these derivatives offer a pathway to more effective oncology therapeutics. The innovation lies not just in the final molecule, but in the robust synthetic methodology that allows for the systematic variation of amine groups to tune physicochemical properties.
For R&D directors and procurement specialists, understanding the structural nuances of these compounds is critical for evaluating their potential as high-purity pharmaceutical intermediates. The general formula (I) described in the patent encompasses a broad range of derivatives where the central linker can be either a 1,6-heptadiene-3,5-dione or a 1,4-pentadiene-3-one system. This structural flexibility allows for the fine-tuning of metabolic stability and receptor binding affinity. The presence of the amino group, capable of forming pharmaceutically acceptable salts, is the key differentiator that transforms a poorly soluble natural product into a drug-like candidate suitable for systemic administration. This shift from a nutraceutical grade material to a defined chemical entity opens new avenues for intellectual property and commercial development in the anticancer sector.
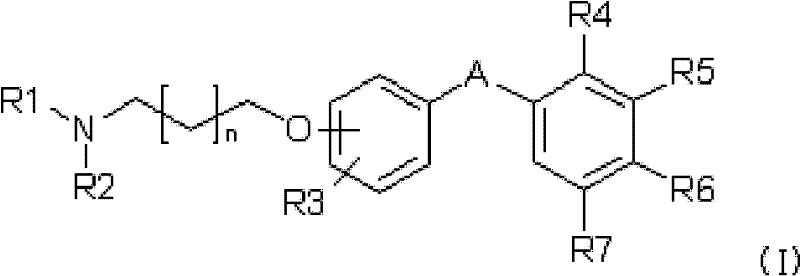
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to utilizing curcumin have largely relied on direct extraction or simple esterification, methods that fail to fundamentally alter the molecule's hydrophobic nature. Native curcumin exhibits extremely low aqueous solubility, which results in rapid metabolism and excretion, preventing it from reaching therapeutic concentrations in target tissues. Previous attempts to modify the structure often involved complex multi-step syntheses that yielded unstable products or required expensive, hard-to-source starting materials. Furthermore, many prior art derivatives focused on modifying the beta-diketone moiety, which is crucial for the molecule's metal-chelating ability and biological activity, thereby risking the loss of the very pharmacophore responsible for its efficacy. These conventional routes often suffer from low atom economy and generate significant waste, posing challenges for environmental compliance and cost-effective manufacturing at scale.
The Novel Approach
The methodology presented in this patent circumvents these issues by targeting the phenolic hydroxyl groups for modification, preserving the critical beta-diketone and conjugated double bond systems. The novel approach utilizes a convergent synthesis strategy where the aminoalkoxy side chain is introduced early in the process via nucleophilic substitution on a benzaldehyde precursor. This ensures that the solubility-enhancing group is securely attached before the final assembly of the curcuminoid skeleton. By employing standard aldol condensation reactions for the final coupling steps, the process leverages well-understood chemistry that is easily scalable. This strategy not only improves the water solubility dramatically but also enhances the antitumor activity, with specific derivatives showing significantly higher potency against breast cancer cell lines compared to the parent compound. The ability to synthesize these compounds using readily available reagents makes this a highly attractive route for commercial production.
Mechanistic Insights into Aldol Condensation Synthesis
The core of this synthetic innovation relies on a series of base-catalyzed aldol condensation reactions, a fundamental transformation in organic chemistry that allows for the construction of carbon-carbon bonds with high precision. The mechanism begins with the preparation of a specialized benzaldehyde intermediate containing the desired aminoalkoxy side chain. This is achieved by reacting a hydroxybenzaldehyde, such as vanillin or isovanillin, with a dihaloalkane under alkaline conditions to form a haloalkoxy intermediate, which is subsequently displaced by a secondary amine. This nucleophilic substitution is highly efficient and allows for the introduction of diverse amine functionalities, including piperidine, morpholine, and diethylamino groups, providing a versatile library of potential drug candidates. The retention of the aldehyde functionality is crucial for the subsequent chain extension steps.
Following the preparation of the functionalized aldehyde, the synthesis proceeds through the formation of a ketone intermediate. Depending on the target structure (Formula IV or V), either acetylacetone or acetone is condensed with a substituted aromatic aldehyde. In the case of the heptadiene-dione backbone, acetylacetone is complexed with boron reagents to protect the central methylene group, directing the condensation to the terminal methyl groups. This protection-deprotection strategy ensures regioselectivity, preventing polymerization and ensuring the formation of the symmetric or asymmetric curcuminoid structure. The final step involves the condensation of the aminoalkoxy-benzaldehyde with the ketone intermediate. Under basic conditions, enolate formation followed by nucleophilic attack on the aldehyde carbonyl generates the extended conjugated system characteristic of curcumin. The dehydration of the resulting beta-hydroxy ketone yields the stable alpha,beta-unsaturated ketone linkage, completing the synthesis of the target derivative.
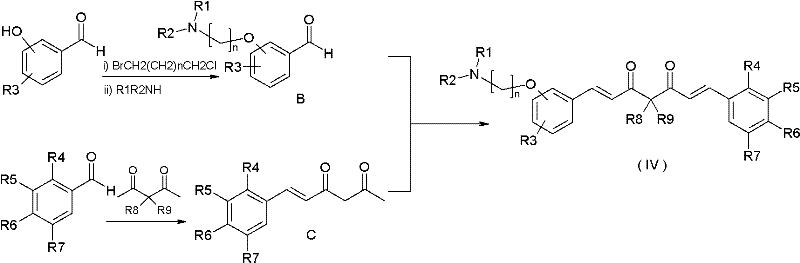
How to Synthesize Aminoalkoxy Curcumin Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction conditions to maximize yield and purity. The process is divided into three distinct stages: the functionalization of the aromatic ring, the preparation of the ketone linker, and the final condensation. Each step utilizes common laboratory solvents such as ethanol, ethyl acetate, and dichloromethane, facilitating easy workup and solvent recovery. The reaction temperatures are moderate, typically ranging from room temperature to reflux conditions, which minimizes energy consumption and thermal degradation of sensitive intermediates. Purification is primarily achieved through silica gel column chromatography or recrystallization, techniques that are standard in pharmaceutical manufacturing and ensure the removal of unreacted starting materials and byproducts.
- Preparation of aminoalkoxy-substituted benzaldehyde intermediates via nucleophilic substitution of haloalkanes with amines.
- Synthesis of styryl 1,3-dicarbonyl or styrene acetone intermediates through base-catalyzed condensation.
- Final coupling of aldehyde and ketone intermediates via aldol condensation to form the target curcumin derivative structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial benefits regarding raw material security and process scalability. The starting materials, including vanillin, acetone, and acetylacetone, are commodity chemicals produced in massive volumes globally, ensuring a stable and cost-effective supply chain that is not subject to the volatility often associated with exotic reagents. The reliance on base-catalyzed reactions eliminates the need for expensive transition metal catalysts, which not only reduces the direct cost of goods but also simplifies the downstream purification process by removing the requirement for rigorous heavy metal scavenging steps. This reduction in processing complexity translates directly into shorter production cycles and lower operational expenditures for manufacturing partners.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of abundant, low-cost starting materials significantly lower the overall production cost. The synthetic pathway avoids complex protection group strategies for the final coupling, streamlining the process and reducing the number of unit operations required. This efficiency gain allows for competitive pricing of the final pharmaceutical intermediate, making it an economically viable option for large-scale drug development programs seeking to optimize their cost of goods sold without compromising on quality.
- Enhanced Supply Chain Reliability: Because the synthesis relies on widely available chemical building blocks, the risk of supply disruption is minimized. The robustness of the aldol condensation chemistry means that the process is less sensitive to minor variations in reaction conditions, leading to consistent batch-to-batch quality. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the stringent delivery timelines required by global pharmaceutical clients. The ability to source reagents from multiple suppliers further mitigates supply chain risks.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in multi-gram laboratory preparations that can be directly translated to pilot and commercial plant scales. The use of standard organic solvents allows for established recovery and recycling protocols, reducing waste generation and environmental impact. Furthermore, the avoidance of toxic heavy metals aligns with increasingly strict regulatory guidelines regarding residual impurities in drug substances, facilitating smoother regulatory filings and approvals for downstream drug products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these curcumin derivatives. The answers are derived from the specific experimental data and structural claims found within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating these intermediates into their existing development pipelines.
Q: How do aminoalkoxy side chains improve curcumin efficacy?
A: The introduction of aminoalkoxy side chains significantly enhances water solubility compared to native curcumin, which directly improves bioavailability and allows for higher effective concentrations at the tumor site.
Q: What is the anticancer activity profile of these derivatives?
A: Experimental data indicates that these derivatives exhibit 2 to 9 times higher inhibitory activity against human breast cancer MCF-7 cells compared to standard curcumin.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the synthesis relies on commodity chemicals such as vanillin, acetone, acetylacetone, and common aliphatic amines, ensuring a robust and cost-effective supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Curcumin Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the aldol condensation processes described in patent CN102503842A to meet stringent purity specifications required for oncology applications. We understand that the transition from laboratory scale to commercial manufacturing requires meticulous attention to detail, particularly in controlling impurities and ensuring consistent polymorphic forms. Our rigorous QC labs are equipped to analyze these complex molecules, ensuring that every batch meets the highest standards of quality and safety.
We invite you to contact our technical procurement team to discuss your specific requirements for aminoalkoxy curcumin derivatives. Whether you need a Customized Cost-Saving Analysis for your project or require specific COA data and route feasibility assessments, we are ready to provide the support you need. Partnering with us ensures access to a reliable supply of high-quality pharmaceutical intermediates, enabling you to accelerate your drug development timelines with confidence.
