Advanced Synthesis of 1,3-Disubstituted Pyrazole Derivatives for Oncology Applications
Introduction to Novel Antitumor Scaffolds
The escalating global burden of prostate cancer necessitates the continuous development of novel therapeutic agents capable of overcoming resistance mechanisms associated with current androgen deprivation therapies. Patent CN107400088B discloses a series of 1,3-disubstituted pyrazole derivatives that exhibit potent androgen receptor antagonistic activity and significant inhibition of prostate cancer cell proliferation. These compounds represent a strategic advancement in medicinal chemistry, offering a new structural backbone that differs from first-generation antagonists like bicalutamide. The core structure, defined by General Formula (I), allows for diverse substitution patterns at the phenyl rings, enabling fine-tuning of pharmacokinetic properties and binding affinity. As a reliable pharmaceutical intermediates supplier, understanding the structural nuances of such scaffolds is critical for supporting downstream drug development pipelines.
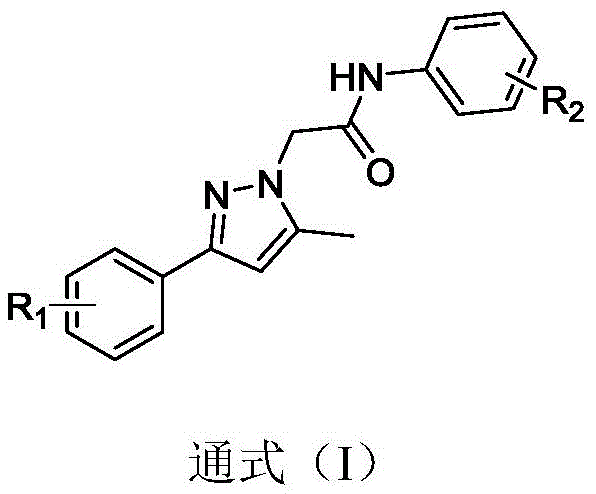
The significance of this chemical class lies in its dual mechanism of action, potentially blocking androgen receptor signaling pathways while simultaneously inhibiting tumor cell growth through alternative mechanisms. For R&D directors evaluating new leads, the versatility of the pyrazole core offers a robust platform for structure-activity relationship (SAR) studies. The patent details specific embodiments where R1 and R2 groups include fluorine, cyano, chlorine, and trifluoromethyl moieties, which are known to enhance metabolic stability and membrane permeability. This level of structural detail provides a clear roadmap for the commercial scale-up of complex pharmaceutical intermediates required for preclinical and clinical trials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for heterocyclic androgen receptor antagonists often rely on harsh reaction conditions, expensive transition metal catalysts, or multi-step sequences that suffer from poor atom economy. Conventional methods for constructing the pyrazole ring frequently involve hydrazine hydrate under reflux conditions, which can pose safety hazards and generate significant waste streams. Furthermore, older alkylation strategies to attach the acetamide side chain often require strong bases at elevated temperatures, leading to side reactions such as hydrolysis of the amide bond or over-alkylation. These inefficiencies result in lower overall yields and complicate the purification process, thereby increasing the cost of goods sold (COGS) for the final active pharmaceutical ingredient (API). Supply chain heads are particularly concerned with the consistency of supply when legacy processes are prone to batch-to-batch variability due to sensitive reaction parameters.
The Novel Approach
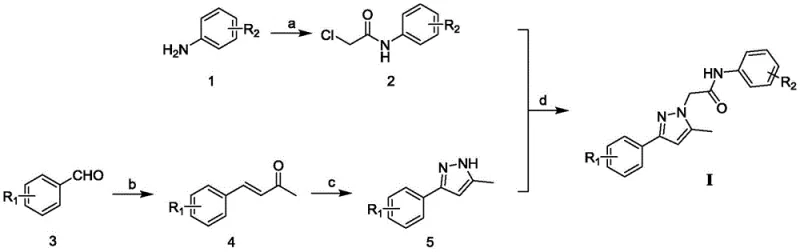
The methodology described in the patent introduces a streamlined, four-step synthetic route that mitigates many of these traditional drawbacks by utilizing milder conditions and organocatalysis. A key innovation is the use of morpholine trifluoroacetate as an organocatalyst for the aldol condensation step, which proceeds efficiently at 80-90°C without the need for corrosive inorganic acids or bases. This approach not only simplifies the workup procedure but also aligns with green chemistry principles by reducing the environmental footprint of the manufacturing process. The subsequent cyclization to form the pyrazole ring is conducted at a moderate temperature of 45-55°C, preserving the integrity of sensitive functional groups like nitriles and halogens. This novel approach ensures cost reduction in pharmaceutical intermediates manufacturing by minimizing energy consumption and reducing the need for extensive chromatographic purification.
Mechanistic Insights into Organocatalytic Condensation and Cyclization
The synthetic pathway begins with the acylation of substituted anilines using chloroacetyl chloride in the presence of triethylamine, a standard Schotten-Baumann type reaction that proceeds rapidly at 20-30°C. The mechanistic precision here is vital for R&D teams, as the stoichiometry of triethylamine (1:1 to 1:2 ratio relative to aniline) ensures complete scavenging of the generated HCl, preventing protonation of the aniline nucleophile which would halt the reaction. Following this, the formation of the chalcone-like intermediate via aldol condensation is catalyzed by morpholine trifluoroacetate. This organocatalyst activates the acetone enolate through hydrogen bonding interactions, facilitating nucleophilic attack on the substituted benzaldehyde. The reaction is driven to completion over 60-80 hours, yielding the alpha,beta-unsaturated ketone with high stereoselectivity for the trans-isomer, which is crucial for the subsequent cyclization step.
The construction of the pyrazole core involves the reaction of the enone intermediate with p-toluenesulfonyl hydrazide, followed by base-mediated cyclization using sodium hydroxide. This mechanism likely proceeds through the formation of a hydrazone intermediate, which then undergoes intramolecular nucleophilic attack to close the five-membered ring. The use of sodium hydroxide in acetonitrile at 45-55°C promotes the elimination of the tosyl group and aromatization of the pyrazole ring. Finally, the N-alkylation step utilizes sodium hydride in DMF to deprotonate the pyrazole nitrogen, generating a nucleophilic anion that displaces the chloride from the chloroacetamide intermediate. This sequence demonstrates excellent chemoselectivity, as the amide carbonyl remains intact despite the strongly basic conditions, ensuring the final product retains the necessary pharmacophore for androgen receptor binding.
How to Synthesize 1,3-Disubstituted Pyrazole Derivatives Efficiently
Executing this synthesis requires strict adherence to the specified reaction parameters to maximize yield and purity, particularly during the final alkylation step where yields can vary between 9% and 44% depending on the electronic nature of the substituents. The process is designed to be scalable, utilizing common solvents like dichloromethane, acetone, acetonitrile, and DMF which are readily available in bulk quantities for commercial production. Purification is primarily achieved through recrystallization from petroleum ether and ethyl acetate mixtures, a technique that is highly favorable for large-scale manufacturing compared to column chromatography. Detailed standardized synthesis steps are provided below to guide process chemists in replicating the results described in the patent documentation.
- Acylation of substituted aniline with chloroacetyl chloride in dichloromethane at 20-30°C to form the chloroacetamide intermediate.
- Organocatalytic aldol condensation of substituted benzaldehyde with acetone using morpholine trifluoroacetate at 80-90°C.
- Cyclization of the enone intermediate with p-toluenesulfonyl hydrazide and sodium hydroxide in acetonitrile to form the pyrazole core.
- Final N-alkylation of the pyrazole intermediate with the chloroacetamide using sodium hydride in DMF to yield the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route outlined in this patent offers substantial advantages regarding raw material sourcing and inventory management. The starting materials, including various substituted anilines and benzaldehydes, are commodity chemicals produced on a multi-ton scale globally, ensuring a stable and resilient supply chain. This abundance mitigates the risk of supply disruptions that often plague projects relying on exotic or custom-synthesized building blocks. Furthermore, the avoidance of precious metal catalysts such as palladium or platinum eliminates the need for costly metal scavenging resins and rigorous testing for residual metals in the final API, which significantly streamlines the quality control workflow. These factors collectively contribute to a more predictable and cost-effective manufacturing model for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of organocatalysis drastically simplify the downstream processing requirements. By removing the need for specialized metal removal steps, manufacturers can reduce both the direct costs of reagents and the indirect costs associated with waste disposal and equipment cleaning. The mild reaction temperatures (mostly below 100°C) also translate to lower energy consumption compared to high-pressure or high-temperature alternatives. Additionally, the ability to purify intermediates via simple recrystallization rather than preparative HPLC or flash chromatography allows for significant savings in solvent usage and labor time, enhancing the overall economic viability of the process.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity feedstocks ensures that production schedules are not held hostage by the lead times of niche suppliers. Substituted anilines and benzaldehydes are produced by numerous chemical manufacturers worldwide, fostering a competitive market that stabilizes pricing and availability. This diversity in sourcing options allows supply chain managers to establish multi-vendor strategies, reducing the risk of single-point failures. Moreover, the robustness of the synthetic steps, which tolerate a range of functional groups without requiring protecting group strategies, reduces the total number of unit operations, thereby shortening the overall production cycle time and improving responsiveness to market demand.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot plant operations without fundamental changes to the chemistry. The use of acetonitrile and DMF, while requiring proper handling, is well-established in the industry with mature recovery and recycling protocols. The absence of heavy metals simplifies environmental compliance and wastewater treatment, aligning with increasingly stringent global regulations on pharmaceutical manufacturing emissions. This environmental compatibility not only reduces regulatory risk but also enhances the corporate sustainability profile of the manufacturing partner, a key consideration for modern pharmaceutical companies seeking responsible supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 1,3-disubstituted pyrazole derivatives. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this chemistry into your existing drug development portfolio.
Q: What is the primary therapeutic application of these pyrazole derivatives?
A: These compounds are designed as androgen receptor antagonists for the treatment of prostate cancer, showing significant growth inhibitory activity against LNCaP and PC-3 cell lines.
Q: Does the synthesis process involve toxic heavy metal catalysts?
A: No, the synthesis utilizes organocatalysis (morpholine trifluoroacetate) for the condensation step, eliminating the need for expensive and toxic transition metal catalysts.
Q: What are the critical reaction conditions for the pyrazole ring formation?
A: The cyclization requires reacting the enone intermediate with p-toluenesulfonyl hydrazide at 45-55°C followed by the addition of sodium hydroxide for 10-15 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Disubstituted Pyrazole Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing complex heterocyclic syntheses, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest standards required for oncology drug development. Our commitment to quality and reliability makes us the preferred partner for global pharmaceutical companies seeking to advance novel antitumor agents.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of this specific pyrazole scaffold, we are ready to assist. Please contact us to request specific COA data and route feasibility assessments tailored to your development timeline. Let us collaborate to bring these promising therapeutic candidates from the laboratory to the patients who need them most.
