Scalable Production of High-Purity L-Tert-Leucine Using Novel LaLeuDH Mutants for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable solutions for the synthesis of optically pure non-protein amino acids, which serve as critical chiral building blocks for numerous high-value drugs. Patent CN113106078B introduces a groundbreaking advancement in this domain by disclosing a novel leucine dehydrogenase (LaLeuDH) mutant and its application in the preparation of L-tert-leucine, a key intermediate for antiviral agents such as telaprevir and atazanavir. This technology addresses the longstanding bottlenecks of substrate inhibition and excessive coenzyme dependence that have historically plagued the industrial biocatalytic production of this valuable compound. By leveraging structure-oriented gene mining and rational protein engineering, the inventors have developed a mutant strain capable of tolerating high substrate concentrations up to 1.5M while significantly reducing the requirement for expensive NADH cofactors. For global procurement and R&D teams, this represents a pivotal shift towards more economically viable and environmentally sustainable manufacturing processes for complex chiral amino acids.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis and early-generation enzymatic methods for producing L-tert-leucine face severe economic and technical constraints that hinder large-scale adoption. Conventional chemical routes often involve harsh reaction conditions, the use of toxic heavy metal catalysts, and complex resolution steps that inherently limit the maximum theoretical yield to 50%, resulting in significant waste generation and increased purification costs. In the realm of biocatalysis, while wild-type leucine dehydrogenases offer better stereoselectivity, they typically suffer from strong substrate inhibition when trimethylpyruvate concentrations exceed 0.75M, necessitating fed-batch strategies that complicate process control and extend production cycles. Furthermore, these native enzymes exhibit a high dependency on exogenous coenzymes, requiring the continuous addition of NADH or expensive regeneration systems to maintain catalytic activity, which substantially inflates the raw material costs and complicates the downstream purification of the final pharmaceutical intermediate.
The Novel Approach
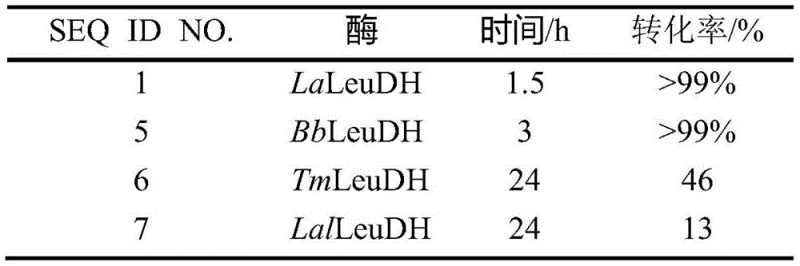
The innovative approach detailed in the patent overcomes these hurdles through the deployment of specifically engineered LaLeuDH mutants, particularly the double mutant D153N/H191N, which demonstrates exceptional catalytic performance under industrially relevant conditions. This novel biocatalyst enables the asymmetric reductive amination of trimethylpyruvate at a remarkable substrate concentration of 1.5M in a single batch, effectively doubling the space-time yield compared to previous enzymatic methods without the risk of enzyme deactivation. The enhanced stability and activity allow for a drastic reduction in coenzyme usage, with the capability to achieve complete conversion even without the addition of exogenous NADH when coupled with an efficient internal regeneration system. This leap in performance not only simplifies the operational workflow but also aligns perfectly with the industry's demand for cost reduction in pharmaceutical intermediate manufacturing by minimizing reagent consumption and maximizing reactor throughput.
Mechanistic Insights into LaLeuDH-D153N/H191N Catalytic Enhancement
The superior performance of the LaLeuDH-D153N/H191N mutant is rooted in precise structural modifications that optimize both the substrate-binding pocket and the coenzyme-binding domain. Through rational design based on homology modeling and molecular dynamics simulations, the inventors identified that mutating the aspartic acid at position 153 and the histidine at position 191 to asparagine residues significantly enhances the rigidity of the enzyme's loop structures. This structural reinforcement prevents the collapse of the active site under high concentrations of organic substrates like trimethylpyruvate, thereby mitigating substrate inhibition. Kinetic analysis reveals that the double mutant exhibits a Michaelis constant (Km) for NADH of merely 0.017 mM, a dramatic decrease from the 0.43 mM observed in the wild-type enzyme, indicating a vastly improved affinity for the cofactor. Consequently, the catalytic efficiency (kcat/Km) towards NADH is increased by approximately 50-fold, ensuring that the enzyme can recycle the limited available cofactor molecules with extreme rapidity to sustain high reaction rates throughout the conversion process.
In addition to coenzyme affinity, the mutant maintains high specificity for the substrate, ensuring the production of high-purity L-tert-leucine with an enantiomeric excess (ee) value reaching 99.9%. The reaction mechanism involves the transfer of a hydride ion from NADH to the carbonyl group of trimethylpyruvate in the presence of ammonium ions, followed by the regeneration of NAD+ which is subsequently reduced back to NADH by a coupled glucose dehydrogenase system. This closed-loop cofactor recycling is crucial for economic feasibility, as it eliminates the need for stoichiometric amounts of expensive reducing agents. The stability of the mutant enzyme in aqueous buffers at neutral pH further facilitates easy handling and scalability, making it an ideal candidate for the commercial scale-up of complex chiral amino acids where consistent quality and minimal impurity profiles are paramount for regulatory compliance.
How to Synthesize L-Tert-Leucine Efficiently
The implementation of this advanced biocatalytic route requires a streamlined workflow that integrates genetic engineering with optimized fermentation and conversion protocols. The process begins with the construction of a recombinant Escherichia coli host strain that co-expresses the engineered LaLeuDH-D153N/H191N mutant alongside a glucose dehydrogenase for in situ cofactor regeneration. Following the cultivation of these engineered cells to high density, the biomass is harvested and utilized directly as a whole-cell catalyst or as freeze-dried powder, eliminating the need for costly enzyme purification steps. The detailed standardized synthesis steps see the guide below, which outlines the precise conditions for achieving maximum conversion efficiency while maintaining the structural integrity of the biocatalyst throughout the reaction duration.
- Construct recombinant E. coli BL21(DE3) strains co-expressing the LaLeuDH-D153N/H191N mutant and glucose dehydrogenase (BmGDH) for cofactor regeneration.
- Cultivate the engineered bacteria in LB medium with kanamycin, induce protein expression with IPTG at 25°C, and harvest cells via centrifugation.
- Perform the biocatalytic reaction using 1.5M trimethylpyruvate and ammonium buffer at pH 8.0, achieving complete conversion without exogenous NADH addition.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented LaLeuDH mutant technology translates into tangible strategic benefits that extend beyond simple yield improvements. The ability to operate at high substrate concentrations fundamentally alters the economics of production by reducing the volume of water and buffer solutions required per kilogram of product, which in turn lowers energy consumption for heating, cooling, and wastewater treatment. The elimination of exogenous coenzyme addition removes a significant variable cost component, providing a buffer against market fluctuations in the price of biochemical reagents. Furthermore, the robust nature of the mutant enzyme reduces the risk of batch failures due to substrate inhibition, ensuring more predictable production schedules and enhancing overall supply chain reliability for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization lies in the mutant enzyme's extraordinary affinity for NADH, which allows the process to function with minimal to zero external cofactor supplementation. By removing the necessity for purchasing large quantities of expensive NADH and simplifying the cofactor regeneration system, manufacturers can achieve substantial cost savings in raw materials. Additionally, the high tolerance for substrate concentration means that smaller reactors can produce the same output as larger conventional setups, effectively reducing capital expenditure requirements and operational overheads associated with facility footprint and maintenance.
- Enhanced Supply Chain Reliability: The robustness of the LaLeuDH-D153N/H191N mutant ensures consistent performance even under varying process conditions, which minimizes the risk of production delays caused by enzyme instability or inhibition. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers who depend on just-in-time delivery of high-quality intermediates. The use of a whole-cell catalytic system further simplifies the logistics of enzyme storage and transport, as freeze-dried bacterial cells are generally more stable and easier to handle than purified enzyme preparations, thereby reducing lead time for high-purity L-tert-leucine deliveries.
- Scalability and Environmental Compliance: From an environmental perspective, this enzymatic process operates under mild aqueous conditions, avoiding the use of hazardous organic solvents and heavy metal catalysts typical of chemical synthesis. This green chemistry approach significantly reduces the burden of waste disposal and helps manufacturers meet increasingly stringent environmental regulations without compromising on productivity. The high atom economy of the reductive amination reaction, combined with the efficient cofactor recycling, ensures that resource utilization is maximized, making the process highly scalable from pilot plant to multi-ton commercial production while maintaining a low environmental impact profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel leucine dehydrogenase technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of how this innovation resolves historical challenges in chiral amino acid synthesis. Understanding these specifics is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into their existing manufacturing infrastructure.
Q: How does the LaLeuDH-D153N/H191N mutant reduce production costs compared to wild-type enzymes?
A: The mutant exhibits a 50-fold increase in catalytic efficiency towards NADH, allowing the reaction to proceed efficiently with significantly reduced or even zero exogenous coenzyme addition, drastically lowering material costs.
Q: What is the maximum substrate concentration tolerated by this new enzymatic process?
A: Unlike conventional enzymes that suffer inhibition above 0.75M, the LaLeuDH-D153N/H191N mutant maintains high activity at substrate concentrations up to 1.5M trimethylpyruvate, enabling higher space-time yields.
Q: Is the optical purity of the synthesized L-tert-leucine suitable for API manufacturing?
A: Yes, the enzymatic asymmetric reductive amination achieves an enantiomeric excess (ee) value of 99.9%, meeting the stringent purity specifications required for active pharmaceutical ingredient intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Tert-Leucine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the LaLeuDH-D153N/H191N mutant technology in reshaping the landscape of chiral intermediate production. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this patent are fully realized in practical, large-scale operations. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities designed to meet stringent purity specifications, guaranteeing that every batch of L-tert-leucine delivered meets the highest standards required by global regulatory bodies for pharmaceutical applications.
We invite forward-thinking pharmaceutical companies and chemical manufacturers to collaborate with us to leverage this cutting-edge biocatalytic technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive efficiency and profitability in your manufacturing of high-value chiral building blocks.
