Advanced Manufacturing of 2-n-propyl-4-methyl-6-carboxybenzimidazole for Global Supply Chains
Advanced Manufacturing of 2-n-propyl-4-methyl-6-carboxybenzimidazole for Global Supply Chains
The global demand for Angiotensin II Receptor Blockers (ARBs) continues to drive the need for robust, scalable, and high-purity intermediate supply chains. A pivotal development in this sector is detailed in patent CN109320461B, which outlines a superior preparation method for 2-n-propyl-4-methyl-6-carboxybenzimidazole, a critical precursor for the antihypertensive drug Telmisartan. This technology addresses long-standing industry pain points regarding product consistency, color quality, and environmental safety. By shifting away from harsh nitration and heavy metal catalysis, this novel approach offers a streamlined pathway that aligns perfectly with modern Good Manufacturing Practice (GMP) standards. For procurement leaders and R&D directors, understanding the mechanistic advantages of this route is essential for securing a reliable pharmaceutical intermediate supplier partnership that guarantees both quality and continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key benzimidazole derivative has been plagued by complex multi-step sequences that introduce significant operational risks and quality variability. Traditional routes, such as those disclosed in earlier literature, often rely on the acylation of methyl 3-methyl-4-aminobenzoate followed by harsh mixed-acid nitration and subsequent hydrogenation. These processes frequently result in batch-to-batch variations and the accumulation of stubborn impurities that are difficult to purge. Furthermore, alternative pathways involving palladium-catalyzed carbonyl insertion, while chemically elegant, introduce expensive transition metals that require rigorous and costly removal protocols to meet residual solvent and heavy metal specifications. The reliance on strong acids like fuming sulfuric acid also generates substantial hazardous waste, complicating environmental compliance and increasing the total cost of ownership for manufacturers.
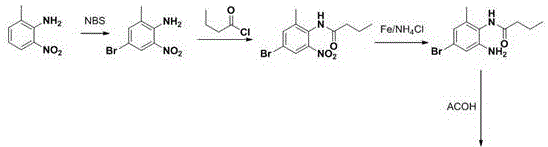
The Novel Approach
In stark contrast, the methodology protected under CN109320461B introduces a concise, three-step strategy that fundamentally simplifies the molecular construction. By utilizing butyronitrile as a starting material and activating it via a Pinner-like reaction with anhydrous hydrogen chloride, the process bypasses the need for pre-functionalized esters or dangerous nitration steps. The subsequent condensation with 3-methyl-4-aminobenzoic acid is conducted under mild acidic conditions using glacial acetic acid, which strategically prevents unwanted esterification of the carboxylic acid moiety. This clever manipulation of reaction conditions eliminates the need for a separate hydrolysis step, thereby reducing unit operations and solvent consumption. The final oxidative cyclization using sodium hypochlorite is not only cost-effective but also highly selective, ensuring that the final API intermediate meets stringent purity profiles without the dark coloration typical of older methods.
Mechanistic Insights into Oxidative Cyclization and pH Control
The first stage of this innovative synthesis involves the activation of butyronitrile in an ethanolic hydrogen chloride environment. As illustrated in the reaction scheme below, the nitrile group undergoes nucleophilic attack by ethanol in the presence of anhydrous HCl gas to form an imino ether salt intermediate. This step is critically dependent on maintaining anhydrous conditions and controlling the temperature between 0-35°C to maximize the formation of the reactive species while minimizing polymerization or degradation. The precise stoichiometry of hydrogen chloride to nitrile is optimized to ensure complete conversion, setting the stage for a high-yielding coupling reaction in the subsequent step. This activation strategy is far more atom-economical than generating acyl chlorides or using activated esters.
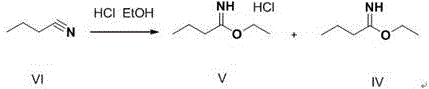
Following activation, the process moves to the coupling phase where pH control becomes the defining parameter for success. The reaction mixture is carefully adjusted to a pH range of 5.0-11.0, preferably 5.5-7.8, during the addition of the amine component. This specific window is vital; if the pH is too low, the amine remains protonated and unreactive, while a pH that is too high can lead to the hydrolysis of the imino ether before coupling occurs. Furthermore, the use of glacial acetic acid as the reaction medium, rather than concentrated sulfuric acid, prevents the esterification of the benzoic acid group. This ensures that the carboxylic acid functionality remains free and available, avoiding the need for a harsh base hydrolysis step later in the process which often degrades product quality.

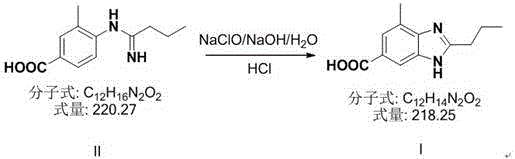
The final transformation involves an oxidative cyclization to close the benzimidazole ring. In this step, the linear amidine intermediate is treated with sodium hypochlorite under basic conditions. The hypochlorite acts as a mild oxidant, facilitating the intramolecular condensation that forms the heterocyclic core. A key innovation here is the post-reaction treatment with sodium dithionite and activated carbon. This dual treatment effectively reduces colored oxidation byproducts and adsorbs trace organic impurities, transforming the crude product from a deep yellow solid into a pharmaceutically acceptable off-white or white powder. This capability to control the physical appearance of the intermediate is a significant advantage for downstream API synthesis, where color bodies can persist through multiple steps.
How to Synthesize 2-n-propyl-4-methyl-6-carboxybenzimidazole Efficiently
Implementing this synthesis route requires precise adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility at scale. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates and reducing solvent exchange operations. Operators must pay close attention to the gas absorption rates during the HCl saturation step and the exotherm management during the hypochlorite addition. Detailed standard operating procedures (SOPs) regarding the specific order of addition and agitation speeds are critical for maintaining the narrow impurity profile. For a comprehensive breakdown of the exact reagent quantities, temperature ramps, and workup procedures, please refer to the standardized synthesis guide below.
- Activate butyronitrile using anhydrous hydrogen chloride in ethanol at 0-35°C to form the imino ether salt intermediate.
- React the activated nitrile with 3-methyl-4-aminobenzoic acid in acetic acid/methanol, maintaining pH 5.0-11.0 to prevent esterification.
- Perform oxidative cyclization using sodium hypochlorite under basic conditions, followed by decolorization with sodium dithionite.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing route offers compelling advantages that directly impact the bottom line and supply security. By eliminating the reliance on precious metal catalysts like palladium, the process removes a major variable cost driver and mitigates the risk associated with the supply volatility of rare earth metals. Furthermore, the avoidance of mixed-acid nitration significantly reduces the burden on waste treatment facilities, lowering the environmental compliance costs that are increasingly being passed down the supply chain. The simplified workflow, which removes the need for separate esterification and hydrolysis steps, translates to shorter cycle times and higher throughput capacity per reactor volume. These efficiencies allow for a more responsive supply chain capable of meeting fluctuating market demands for Telmisartan without the need for excessive safety stock.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in unit operations lead to substantial cost savings. By avoiding the esterification-hydrolysis sequence, the process saves on reagent costs, solvent usage, and energy consumption associated with heating and cooling cycles. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to byproduct formation, improving the overall mass balance and yield efficiency of the plant.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as butyronitrile and 3-methyl-4-aminobenzoic acid, are commodity chemicals with robust global supply networks. This reduces the risk of raw material shortages that can plague specialized reagent-dependent processes. The stability of the intermediates and the robustness of the reaction conditions mean that production schedules are less likely to be disrupted by failed batches or extended purification times, ensuring consistent on-time delivery to API manufacturers.
- Scalability and Environmental Compliance: The process is inherently safer as it avoids the use of highly flammable solvents in critical steps and eliminates the generation of nitro-containing waste streams. The use of aqueous workups and common solvents like xylene and methanol simplifies solvent recovery and recycling. This green chemistry profile facilitates easier regulatory approval and permits for capacity expansion, allowing suppliers to scale production from pilot plants to multi-ton commercial volumes with minimal environmental friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this high-value intermediate. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this material into their existing supply chains. The answers reflect the specific improvements in purity, color, and process safety that distinguish this method from legacy technologies.
Q: How does this new process improve product color compared to conventional methods?
A: Conventional methods often yield dark yellow products due to side reactions and difficult-to-remove impurities. This patented process utilizes a specific decolorization step with sodium dithionite and activated carbon, resulting in an off-white or white final product with purity exceeding 99.7%.
Q: Why is the pH control critical in the second step of the synthesis?
A: Maintaining the system pH between 5.0 and 11.0 during the reaction of the activated nitrile with 3-methyl-4-aminobenzoic acid is crucial. This specific range prevents the formation of excessive related substances and avoids the esterification of the carboxyl group, which simplifies downstream processing.
Q: Does this manufacturing route involve expensive transition metal catalysts?
A: No, unlike some prior art routes that require palladium-catalyzed carbonylation, this method relies on readily available reagents like sodium hypochlorite and anhydrous hydrogen chloride. This eliminates the need for costly heavy metal removal steps and reduces overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-n-propyl-4-methyl-6-carboxybenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final API is inextricably linked to the purity of its precursors. Our technical team has thoroughly analyzed the pathway described in CN109320461B and validated its potential for large-scale implementation. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch of 2-n-propyl-4-methyl-6-carboxybenzimidazole meets the exacting standards required for global pharmaceutical registration.
We invite you to discuss how this advanced synthesis route can optimize your Telmisartan supply chain. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation drives value for your organization.
