Advanced Enzymatic Synthesis Of Chiral Beta-Amino Acids Using Engineered Dehydrogenase Mutants
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce chiral building blocks, and the recent advancements detailed in patent CN113755462B represent a significant leap forward in enzymatic catalysis. This intellectual property introduces a novel beta-amino acid dehydrogenase mutant derived from Candidatus Cloacamonas acidaminovorans, specifically engineered to overcome the limitations of natural enzymes in synthesizing chiral beta-amino acid compounds. The core innovation lies in the strategic mutation of key amino acid residues, such as the E310G substitution, which dramatically enhances catalytic activity and substrate tolerance. For R&D directors and procurement specialists, this technology offers a robust alternative to traditional chemical synthesis, promising higher purity and reduced environmental impact. The ability to achieve high enantiomeric excess values through biocatalysis is critical for meeting the stringent regulatory requirements of modern drug development, positioning this mutant enzyme as a valuable asset for reliable pharmaceutical intermediate supplier networks aiming to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino acids has been plagued by significant technical hurdles that impact both cost and scalability in industrial settings. Conventional chemical methods often require harsh reaction conditions, including extreme temperatures and pressures, which not only increase energy consumption but also pose safety risks in large-scale manufacturing facilities. Furthermore, traditional enzymatic approaches using wild-type beta-amino acid dehydrogenases have suffered from narrow substrate specificity and low catalytic efficiency, with reported yields often stagnating between 12% and 22%. This inefficiency necessitates extensive downstream processing to isolate the desired product, leading to substantial material loss and increased waste generation. For supply chain heads, these limitations translate into unpredictable lead times and higher operational costs, as the low conversion rates require larger reactor volumes and more raw materials to achieve the same output. The reliance on expensive transition metal catalysts in chemical routes also introduces the risk of heavy metal contamination, requiring additional purification steps that further erode profit margins and complicate compliance with environmental regulations.
The Novel Approach
In stark contrast, the novel approach utilizing the engineered beta-amino acid dehydrogenase mutants described in the patent data offers a transformative solution to these longstanding industry challenges. By introducing specific mutations at core amino acid positions, the new enzyme variants demonstrate a remarkably broadened substrate spectrum and significantly improved catalytic activity, capable of converting beta-keto acids into chiral beta-amino acids with yields reaching up to 95%. This dramatic improvement in efficiency means that manufacturers can achieve higher throughput with smaller reactor footprints, directly addressing the need for cost reduction in chiral amino acid manufacturing. The process operates under mild physiological conditions, typically around 30°C, which preserves the integrity of sensitive functional groups and reduces the thermal load on production equipment. Additionally, the integration of a cofactor regeneration system using glucose dehydrogenase ensures sustainable consumption of NAD(P)H, eliminating the need for stoichiometric amounts of expensive cofactors. 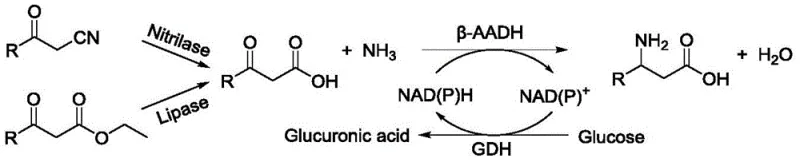
Mechanistic Insights into Beta-Amino Acid Dehydrogenase Catalysis
The mechanistic superiority of this technology stems from the precise structural modifications made to the enzyme's active site, which facilitate more efficient binding and transformation of non-natural substrates. The mutant protein, characterized by at least 90% identity to the sequence in SEQ ID NO.: 2, features alterations at positions such as 159, 179, 180, 323, 324, and 328, which collectively optimize the geometry of the substrate binding pocket. This structural refinement allows the enzyme to accommodate a wider variety of beta-keto acid derivatives, enabling the synthesis of diverse chiral intermediates like (R)-beta-homomethionine and (S)-beta-aminobutyric acid with high stereoselectivity. The catalytic cycle relies on the reversible oxidative deamination and reductive amination reactions dependent on NAD(H) or NADP(H) cofactors, but the engineered mutants exhibit a much higher turnover number compared to their wild-type counterparts. For technical teams, understanding this mechanism is crucial for process optimization, as it highlights the importance of maintaining optimal pH levels between 6.0 and 11.0 to maximize enzyme stability and activity throughout the reaction duration.
Furthermore, the impurity control mechanism inherent in this biocatalytic route provides a distinct advantage over chemical synthesis, particularly regarding the enantiomeric purity of the final product. The high stereoselectivity of the mutant dehydrogenase ensures that the resulting beta-amino acids possess ee values greater than 99%, effectively eliminating the need for complex chiral resolution steps that are often required in racemic chemical syntheses. This level of purity is essential for pharmaceutical applications where even trace amounts of the wrong enantiomer can lead to toxicity or reduced efficacy in the final drug product. The use of whole-cell biocatalysts, where the enzyme is expressed in Escherichia coli, also simplifies the recovery process, as the cells can be easily separated from the reaction mixture via centrifugation. 
How to Synthesize Chiral Beta-Amino Acids Efficiently
Implementing this advanced biocatalytic route requires a systematic approach to ensure maximum yield and consistency across batches, leveraging the specific parameters outlined in the patent examples. The process begins with the preparation of the biocatalyst, where genetically engineered E. coli strains expressing the mutant dehydrogenase are cultured and harvested as wet cells, providing a stable and reusable source of enzymatic activity. Detailed standardized synthesis steps see the guide below, which outlines the precise concentrations of substrates, buffers, and cofactors needed to replicate the high-performance results observed in the laboratory data. By adhering to these protocols, manufacturers can minimize variability and ensure that the commercial scale-up of complex enzyme catalysts proceeds smoothly without unexpected deviations in product quality. The flexibility of the system allows for the use of various beta-keto esters or nitriles as starting materials, which are first hydrolyzed to the corresponding acids before undergoing the key reductive amination step.
- Prepare the reaction system using wet bacterial cells containing the beta-amino acid dehydrogenase mutant as the biocatalyst in a buffered solution.
- Introduce the beta-keto acid substrate along with a cofactor regeneration system comprising glucose and glucose dehydrogenase to sustain NAD(P)H levels.
- Maintain the reaction at mild temperatures between 25°C and 50°C with controlled pH, followed by separation and purification using cationic resin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this mutant dehydrogenase technology presents compelling advantages for procurement managers and supply chain leaders focused on long-term sustainability and cost efficiency. The shift from low-yield wild-type enzymes to high-activity mutants fundamentally alters the economics of production by drastically reducing the amount of raw material required per unit of output, leading to substantial cost savings in manufacturing operations. Since the process achieves conversion rates exceeding 90% and often reaching 95%, there is significantly less unreacted starting material to recover or dispose of, which streamlines the downstream purification workflow and reduces solvent usage. This efficiency gain translates directly into a more competitive pricing structure for the final intermediates, allowing companies to offer high-purity beta-amino acids at a more attractive market price point while maintaining healthy profit margins. Moreover, the mild reaction conditions reduce the wear and tear on production equipment, extending the lifespan of capital assets and lowering maintenance costs over time.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent consumption due to higher conversion rates contribute to a leaner manufacturing cost structure. By avoiding the need for heavy metal removal steps, companies can save on specialized filtration media and waste treatment expenses, resulting in a more environmentally friendly and economically viable process. The high catalytic efficiency also means that less enzyme is needed to achieve the same output, reducing the overall cost of goods sold and improving the bottom line for large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of the engineered enzyme mutants ensures consistent performance across different batches, minimizing the risk of production delays caused by catalyst failure or low yields. This reliability is crucial for maintaining steady supply lines to downstream pharmaceutical customers who depend on just-in-time delivery of critical intermediates. The ability to produce a wide range of substrates with a single enzyme platform also simplifies inventory management, as fewer distinct biocatalysts need to be stocked and validated, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic reaction and the absence of hazardous organic solvents make this process inherently safer and easier to scale from pilot to commercial production volumes. Regulatory compliance is simplified as the process generates less hazardous waste and avoids the use of toxic reagents, aligning with global trends towards green chemistry and sustainable manufacturing practices. This environmental advantage not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturer, appealing to eco-conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzyme technology, based on the specific data and embodiments provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this biocatalytic route into their existing production frameworks. The answers reflect the proven capabilities of the mutant strains in terms of activity, stability, and substrate scope, providing a clear picture of what can be expected during technology transfer. This transparency is vital for building trust between technology providers and manufacturing partners, ensuring that all parties have aligned expectations regarding performance metrics and project timelines.
Q: How does the mutant dehydrogenase improve upon wild-type enzymes?
A: The engineered mutants exhibit significantly broadened substrate spectra and catalytic activities compared to wild-type enzymes, achieving conversion rates exceeding 95% and ee values greater than 99%, whereas previous methods often struggled with yields below 25%.
Q: What are the typical reaction conditions for this biocatalytic process?
A: The process operates under mild conditions, typically requiring temperatures between 25°C and 50°C and a pH range of 6.0 to 11.0, which reduces energy consumption and equipment stress compared to harsh chemical synthesis methods.
Q: Is this technology suitable for large-scale pharmaceutical manufacturing?
A: Yes, the high stability of the mutant enzymes and the efficient cofactor regeneration system allow for scalable production, making it viable for commercial manufacturing of complex pharmaceutical intermediates with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Acid Dehydrogenase Mutant Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented enzyme technology and are fully equipped to support its transition from laboratory discovery to industrial reality. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in the patent can be replicated at a commercial scale. Our rigorous QC labs and state-of-the-art fermentation facilities allow us to maintain stringent purity specifications for all enzymatic products, guaranteeing that every batch meets the exacting standards required by the global pharmaceutical industry. We understand that adopting a new biocatalytic route involves careful validation, and our team is dedicated to providing the technical expertise needed to navigate this process efficiently and effectively.
We invite you to collaborate with us to explore how this advanced dehydrogenase mutant can optimize your supply chain and reduce your overall production costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific product portfolio. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions about integrating this high-efficiency biocatalysis into your manufacturing strategy. Let us help you engineer a bottleneck-free future for your chiral intermediate production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
