Advanced Synthesis of Porphyrin Supramolecular Helical Polymers for High-Efficiency Photovoltaics
Advanced Synthesis of Porphyrin Supramolecular Helical Polymers for High-Efficiency Photovoltaics
The development of advanced organic photovoltaic materials continues to be a critical frontier in renewable energy research, with patent CN110527086B representing a significant technological leap in the design of porphyrin-based supramolecular architectures. This patent discloses a sophisticated synthesis method for a porphyrin-like supramolecular helical polymer that addresses the longstanding limitation of narrow light absorption spectra in traditional porphyrin dyes used for dye-sensitized solar cells. By engineering a specific 4_1 helical supramolecular structure, the invention leverages unique pi-pi stacking interactions to broaden the UV-Vis absorption profile, thereby enhancing light-harvesting efficiency and electron transport capabilities. For R&D directors and procurement specialists in the electronic chemical sector, understanding the mechanistic nuances and scalability of this Ni2+-catalyzed polymerization route is essential for evaluating its potential as a reliable electronic chemical supplier solution for next-generation photovoltaic devices.
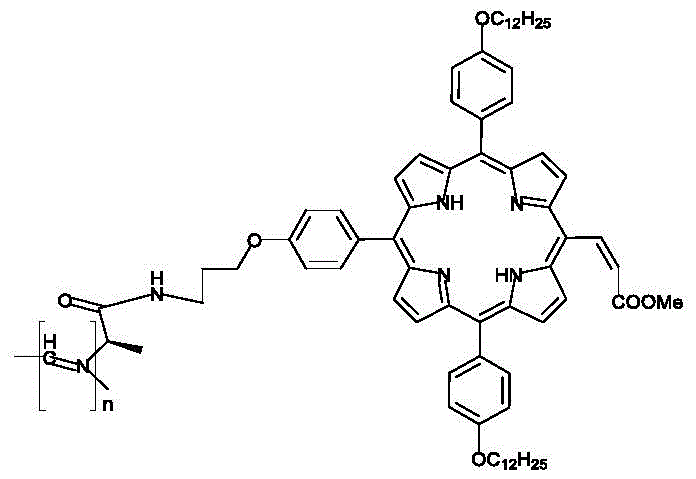
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional porphyrin compounds, while possessing excellent photothermal stability and distinct Soret and Q absorption bands, often suffer from insufficient light-harvesting ability when deployed as standalone dyes in solar cell applications. The core electronic structure of standard porphyrins, characterized by a sixteen-membered conjugated ring with basic four-fold symmetry, limits the breadth of the solar spectrum they can effectively utilize. Furthermore, unfunctionalized or randomly aggregated porphyrin molecules in solution states often exhibit unpredictable conformational changes, leading to inconsistent optical properties and reduced device performance. The lack of a defined supramolecular order in conventional processing methods means that the beneficial effects of intermolecular pi-pi stacking are often lost or result in detrimental aggregation-caused quenching, severely hampering the commercial viability of these materials for high-efficiency organic solar cells.
The Novel Approach
The methodology outlined in patent CN110527086B introduces a paradigm shift by constructing a rigid helical polymer framework that precisely organizes porphyrin units into a chiral 4_1 helical supramolecular structure. This novel approach utilizes a chiral amino acid-modified isocyanide monomer which, upon polymerization, is induced by the chiral center to form a perfect helical scaffold. This scaffold not only prevents random aggregation but also actively tunes the spatial orientation of the pendant porphyrin groups to optimize their electronic coupling. As illustrated in the comprehensive synthetic route below, the process involves a strategic sequence of acylation, etherification, and dehydration reactions to install the functional isocyanide group, followed by a controlled nickel-catalyzed polymerization. This structural precision results in a material with a significantly wider absorption spectrum and superior electron transport efficiency compared to its monomeric counterparts.
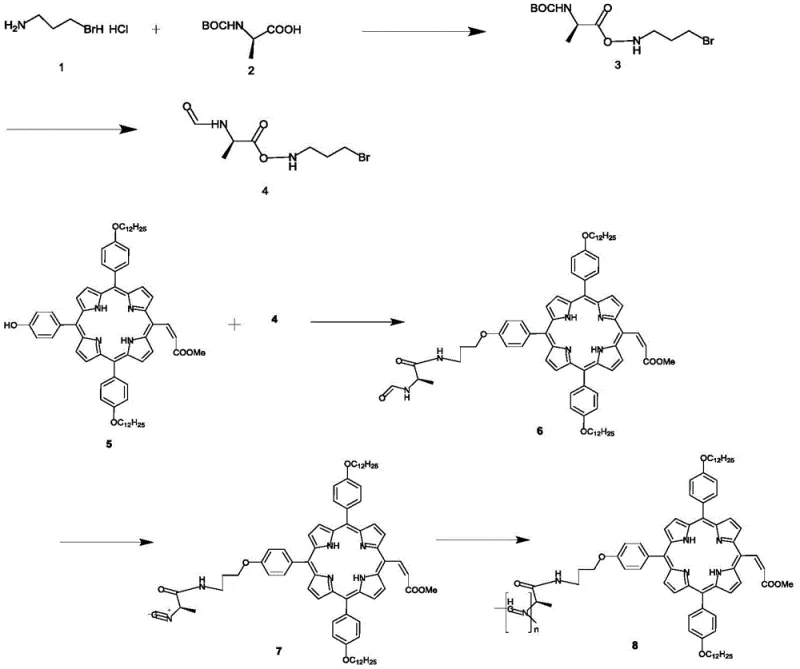
Mechanistic Insights into Ni2+-Catalyzed Isocyanide Polymerization
The core of this synthesis lies in the nickel-catalyzed polymerization of the isocyanide monomer, a reaction that proceeds through a coordination-insertion mechanism critical for maintaining stereochemical integrity. In this process, the nickel(II) center, typically supplied as nickel perchlorate hexahydrate, coordinates with the isocyanide carbon, facilitating the insertion of subsequent monomer units into the growing metal-carbon bond. The presence of the chiral L-alanine derivative on the monomer exerts a profound steric influence, biasing the rotation of the incoming monomers to favor a single-handed helical twist, specifically the 4_1 helix. This helical induction is vital because it ensures that the porphyrin side chains are displayed in a regular, periodic array along the polymer backbone, enabling the specific parallel and horizontal pi-pi stacking interactions that are responsible for the observed splitting of the Soret absorption band from a single peak into dual peaks at 409 nm and 434 nm.
Impurity control in this complex multistep synthesis is managed through rigorous purification protocols at each intermediate stage, particularly following the formation of the sensitive isocyanide functionality. The conversion of the formamide precursor to the isocyanide monomer using diphosgene requires precise temperature control, typically maintained between -50°C and -20°C, to prevent side reactions such as trimerization or hydrolysis which could terminate the polymer chain or introduce defects. Subsequent column chromatography purification using specific silica gel mesh sizes and solvent systems, such as petroleum ether and ethyl acetate gradients, ensures that only the high-purity monomer enters the polymerization reactor. This attention to detail in the upstream synthesis directly correlates to the molecular weight distribution and polydispersity of the final polymer, which are key quality attributes for consistent performance in electronic applications.
How to Synthesize Porphyrin Supramolecular Helical Polymer Efficiently
The synthesis of this high-value electronic material requires a disciplined approach to reaction conditions and stoichiometry to ensure reproducible yields and structural fidelity. The process begins with the preparation of the chiral aldehyde linker, followed by its conjugation to the porphyrin core and subsequent conversion to the polymerizable isocyanide species. Detailed operational parameters, including specific molar equivalents of coupling agents like EDC.HCl and HOBt, as well as precise thermal profiles for each transformation, are critical for success. The standardized synthesis steps provided below outline the proven workflow extracted from the patent examples, serving as a foundational guide for laboratory scale-up and process optimization.
- Perform acylation of Boc-L-alanine with bromoalanine hydrochloride followed by de-protection and N-formylation to generate the chiral aldehyde intermediate.
- Conduct etherification between the chiral aldehyde and hydroxy-porphyrin, followed by dehydration using diphosgene to form the isocyanide monomer.
- Polymerize the isocyanide monomer using nickel perchlorate hexahydrate catalyst in dichloromethane to obtain the final helical polymer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers distinct strategic advantages over sourcing traditional porphyrin dyes, primarily driven by the robustness of the chemical transformations and the scalability of the catalytic system. The reliance on commercially available starting materials such as Boc-protected amino acids and standard porphyrin derivatives mitigates supply risk, while the use of nickel catalysis avoids the cost volatility associated with precious metal catalysts like palladium or platinum. Furthermore, the modular nature of the synthesis allows for the independent optimization of the monomer production and the polymerization steps, facilitating a more flexible manufacturing schedule that can adapt to fluctuating demand in the photovoltaic sector without compromising product quality.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in favor of nickel salts significantly lowers the raw material cost per kilogram of the final polymer. Additionally, the high yields reported in the patent examples for key steps, such as the etherification and polymerization, reduce the burden on downstream purification processes, leading to substantial cost savings in solvent consumption and waste treatment. By streamlining the synthetic sequence and avoiding complex protecting group strategies that require harsh removal conditions, the overall process mass intensity is improved, driving down the cost reduction in organic photovoltaic material manufacturing.
- Enhanced Supply Chain Reliability: The synthetic pathway utilizes reagents and solvents that are commodity chemicals with stable global supply chains, reducing the risk of production delays due to raw material shortages. The robustness of the nickel-catalyzed polymerization, which tolerates a range of reaction conditions without significant loss of stereocontrol, ensures consistent batch-to-batch quality, a critical factor for long-term supply agreements. This reliability allows manufacturers to maintain steady inventory levels of high-purity porphyrin polymers, ensuring reducing lead time for high-purity electronic chemicals needed for urgent R&D or production cycles.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as liquid-liquid extraction and column chromatography that are easily translated from laboratory to pilot and commercial scales. The avoidance of highly toxic reagents in the final polymerization step, combined with the ability to recover and recycle solvents like dichloromethane and chloroform, aligns with stringent environmental compliance standards. This green chemistry profile simplifies the permitting process for new manufacturing facilities and supports the commercial scale-up of complex supramolecular structures with a lower environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this porphyrin supramolecular helical polymer. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the material's performance characteristics and manufacturing feasibility. Understanding these details is crucial for stakeholders evaluating the integration of this advanced material into their existing product pipelines or research frameworks.
Q: What is the primary advantage of the helical structure in this porphyrin polymer?
A: The 4_1 helical supramolecular structure induces specific pi-pi stacking interactions that split the Soret absorption band, significantly broadening the light absorption spectrum compared to monomeric porphyrins.
Q: What catalyst is used for the polymerization step?
A: The polymerization utilizes nickel perchlorate hexahydrate (Ni(ClO4)2·6H2O) as a Lewis acid catalyst to facilitate the coordination-insertion mechanism of the isocyanide monomer.
Q: How is the chirality controlled during synthesis?
A: Chirality is induced by the L-alanine moiety incorporated into the isocyanide monomer, which directs the formation of the single-handed helical backbone during nickel-catalyzed polymerization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Porphyrin Supramolecular Helical Polymer Supplier
As the demand for high-efficiency organic photovoltaic materials accelerates, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides the technical assurance needed to navigate the complexities of supramolecular polymer synthesis. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze the subtle structural nuances of helical polymers, guaranteeing that every batch meets the exacting standards required for high-performance electronic applications.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be adapted to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into the economic benefits of switching to this nickel-catalyzed process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a reliable electronic chemical supplier dedicated to advancing the frontiers of solar energy technology.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
