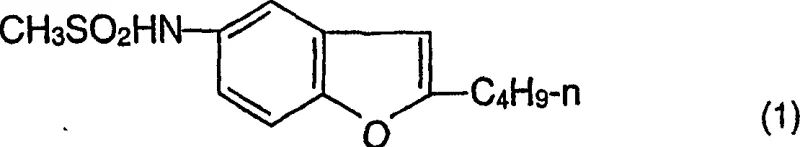
Advanced Synthesis of 2-Butyl-5-Methylsulfonamido-Benzofuran for Scalable Dronedarone Manufacturing
Advanced Synthesis of 2-Butyl-5-Methylsulfonamido-Benzofuran for Scalable Dronedarone Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes for complex cardiovascular agents, and patent CN1229366C provides a groundbreaking methodology for producing 2-butyl-5-methylsulfonamido-benzofuran. This specific compound serves as a critical synthetic intermediate in the preparation of dronedarone, a potent antiarrhythmic agent widely used in clinical settings. The innovation lies in replacing traditional, waste-intensive nitro-based pathways with a cleaner, sulfonamide-first approach that leverages advanced catalytic hydrogenation and optimized Friedel-Crafts acylation. By shifting the functional group installation sequence, manufacturers can achieve superior control over impurity profiles while significantly reducing the environmental burden associated with heavy metal waste disposal. This technical insight report analyzes the mechanistic advantages and commercial viability of this patented process for global supply chain optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dronedarone precursors relied heavily on a five-step sequence starting from 2-butyl-5-nitro-benzofuran, which presented severe industrial drawbacks regarding safety and efficiency. A primary concern was the extensive use of aluminum chloride, often requiring up to nine molar equivalents, which generated massive quantities of aluminum hydroxide sludge upon hydrolysis, creating expensive waste treatment challenges. Furthermore, the intermediate 2-butyl-3-(4-methoxy-benzoyl)-5-nitrobenzofuran possessed mutagenic properties, necessitating stringent containment protocols that increased operational complexity and cost. The cumulative yield of this legacy pathway rarely exceeded 60%, largely due to losses during multiple protection and deprotection steps required to manage the hydroxyl functionality. Additionally, the final hydrogenation step on the fully assembled nitro-compound was prone to side reactions, complicating purification and lowering the overall throughput of the manufacturing line.
The Novel Approach
The patented methodology introduces a paradigm shift by utilizing 2-butyl-5-methylsulfonamido-benzofuran as a stable, crystalline building block that streamlines the synthesis into just three highly efficient steps. By installing the methylsulfonamido group early via the hydrogenation of the nitro precursor followed by sulfonamidation, the process avoids the handling of mutagenic nitro-intermediates in later stages. This route employs a controlled amount of Lewis acid, preferably ferric chloride or limited aluminum chloride, which drastically reduces inorganic waste generation compared to the conventional excess usage. The resulting intermediate exhibits excellent crystallinity, allowing for straightforward isolation and washing procedures that remove impurities without the need for complex chromatographic separations. Consequently, the overall yield improves significantly, often surpassing 65% and reaching up to 90% in the final acylation step, demonstrating a clear advantage in both material efficiency and process safety.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Sulfonamidation
The core of this synthetic strategy relies on the precise manipulation of electronic effects within the benzofuran ring system to facilitate regioselective acylation. In the critical Friedel-Crafts step, the methylsulfonamido group acts as a moderate deactivator but directs the incoming acyl group to the desired position through steric and electronic guidance, minimizing the formation of regioisomers. The use of ferric chloride as a Lewis acid catalyst is particularly advantageous because it offers sufficient activation energy for the acylation of the electron-rich benzofuran ring without promoting excessive decomposition or polymerization side reactions. Unlike aluminum chloride, which forms tight complexes that require harsh hydrolysis conditions, ferric chloride complexes are more manageable, allowing for cleaner workup procedures where the product remains in the organic phase. This phase behavior is crucial for industrial scalability, as it eliminates the need for multiple aqueous extractions that typically lead to product loss and increased solvent consumption.
Impurity control is further enhanced by the physical properties of the 2-butyl-5-methylsulfonamido-benzofuran intermediate itself, which differs markedly from its nitro-analogue. While the nitro-precursor is an oil that is difficult to purify, the sulfonamide derivative readily forms stable crystals upon cooling, enabling a powerful purification mechanism via recrystallization. This crystallization step effectively rejects unreacted starting materials and minor by-products, ensuring that the subsequent acylation reaction proceeds with high-purity inputs. The hydrogenation step converting the nitro group to the amine is also highly selective, achieving near-quantitative conversion without affecting the butyl side chain or the furan ring integrity. Such high fidelity in each transformation step ensures that the final impurity profile of the dronedarone intermediate meets the rigorous standards required for pharmaceutical grade materials, reducing the burden on downstream quality control laboratories.
How to Synthesize 2-Butyl-5-Methylsulfonamido-Benzofuran Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to commercial production, emphasizing reagent availability and operational simplicity. The process begins with the catalytic hydrogenation of 2-butyl-5-nitro-benzofuran, a readily available starting material, using either platinum oxide or palladium on carbon under mild pressure conditions to generate the corresponding amine. This amine is then immediately subjected to sulfonamidation using methanesulfonyl chloride in a biphasic or homogeneous system with an acid scavenger, yielding the key crystalline intermediate in high purity. The final assembly involves reacting this intermediate with 4-[3-(dibutylamino)propoxy]benzoyl chloride in the presence of a Lewis acid, followed by a straightforward hydrolysis and basification sequence to isolate the final product. Detailed standardized operating procedures for these transformations are essential for maintaining batch-to-batch consistency and ensuring regulatory compliance.
- Hydrogenate 2-butyl-5-nitro-benzofuran using platinum oxide or palladium on carbon to obtain 5-amino-2-butyl-benzofuran with yields up to 100%.
- React the resulting amine with methanesulfonyl chloride in the presence of an acid acceptor like triethylamine or ammonia to form the sulfonamide derivative.
- Perform Friedel-Crafts acylation using 4-[3-(dibutylamino)propoxy]benzoyl chloride and a Lewis acid catalyst such as ferric chloride to finalize the structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates directly into tangible operational efficiencies and risk mitigation strategies. The reduction in synthetic steps from five to three inherently lowers the consumption of solvents, reagents, and energy, leading to a substantial decrease in the cost of goods sold without compromising quality. By eliminating the reliance on excessive amounts of aluminum chloride, manufacturers can avoid the significant costs associated with hazardous waste disposal and environmental remediation, which are increasingly stringent in global chemical markets. The use of readily available starting materials like 2-butyl-5-nitro-benzofuran ensures a stable supply base, reducing the risk of raw material shortages that could disrupt production schedules. Furthermore, the improved yield and crystallinity of the intermediate mean that less capacity is tied up in reprocessing off-spec material, thereby increasing the effective throughput of existing manufacturing assets.
- Cost Reduction in Manufacturing: The streamlined three-step process eliminates the need for expensive protection and deprotection sequences, significantly lowering reagent costs and labor hours per kilogram of output. By avoiding the generation of large volumes of aluminum hydroxide sludge, facilities can realize substantial savings in waste treatment fees and reduce the environmental compliance burden. The higher overall yield means that less raw material is required to produce the same amount of final active pharmaceutical ingredient, directly improving the margin structure for the finished drug product. Additionally, the ability to recover the product primarily from the organic phase reduces solvent loss and recovery costs, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Utilizing robust, crystalline intermediates minimizes the risk of batch failures due to purification difficulties, ensuring a consistent and reliable flow of materials to downstream formulation sites. The process relies on commodity chemicals and standard catalysts that are widely available from multiple global suppliers, mitigating the risk of single-source dependency for critical reagents. The simplified workflow reduces the total cycle time for production, allowing for faster response to market demand fluctuations and shorter lead times for order fulfillment. This reliability is crucial for maintaining continuous supply of life-saving cardiovascular medications, where interruptions can have significant clinical and reputational consequences.
- Scalability and Environmental Compliance: The chemistry is designed for scalability, with pilot data demonstrating successful execution at the 10-kilogram scale and clear pathways to multi-ton production without exothermic runaway risks. The reduction in hazardous waste generation aligns with green chemistry principles, making the process more attractive for facilities operating under strict environmental regulations. The use of ferric chloride as a preferred catalyst offers a safer alternative to more aggressive Lewis acids, simplifying equipment maintenance and extending the lifespan of reactor vessels. This environmental and operational safety profile facilitates easier regulatory approval and site audits, accelerating the time to market for generic or new formulations of dronedarone.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the comparative data and experimental examples provided in the patent literature. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for procurement specialists negotiating supply agreements. The answers reflect the specific advantages of the sulfonamide-first strategy over traditional nitro-based methods, highlighting improvements in yield, safety, and waste management. These insights serve as a foundation for deeper discussions regarding process validation and quality assurance protocols.
Q: Why is the new synthesis route superior to conventional methods for Dronedarone intermediates?
A: The novel route eliminates the need for excessive aluminum chloride, reducing hazardous aluminum hydroxide waste, and avoids mutagenic nitro-intermediates, ensuring a safer and more environmentally compliant manufacturing process.
Q: What are the yield advantages of using 2-butyl-5-methylsulfonamido-benzofuran?
A: This specific intermediate allows for a direct three-step synthesis with overall yields exceeding 65%, significantly higher than the maximum 60% yield associated with traditional five-step nitro-based pathways.
Q: How does the crystallinity of this intermediate impact purification?
A: Unlike the difficult-to-crystallize nitro-precursors, the methylsulfonamido derivative forms stable crystals easily, facilitating efficient isolation and washing steps that drastically improve final product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Butyl-5-Methylsulfonamido-Benzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of complex cardiovascular therapeutics like dronedarone. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-butyl-5-methylsulfonamido-benzofuran meets the highest industry standards for impurity profiles and physical properties. Our commitment to process excellence allows us to deliver materials that facilitate smooth downstream processing for our global partners.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. By leveraging our expertise in catalytic hydrogenation and Friedel-Crafts chemistry, we can provide a Customized Cost-Saving Analysis that demonstrates the economic benefits of switching to this optimized synthetic route. Let us collaborate to enhance your supply chain resilience and drive down manufacturing costs while maintaining the highest levels of quality and safety.
