Optimized Synthesis of Cardioactive Forskolin Derivatives for Commercial Scale-Up
Optimized Synthesis of Cardioactive Forskolin Derivatives for Commercial Scale-Up
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, driven by the demand for water-soluble derivatives of natural products with enhanced bioavailability. Patent CN1033583C discloses a robust and highly efficient methodology for the preparation of 6-beta-(3-substituted amino) propionyl forskolin derivatives, a class of compounds exemplified by the potent agent NKH-477. These molecules are critical intermediates in the development of drugs exhibiting positive inotropic, antihypertensive, and vasodilatory activities. The core innovation lies in the strategic manipulation of the forskolin skeleton, specifically utilizing a 1,9-O-isopropylidene protecting group strategy that fundamentally alters the reactivity profile of the molecule. This approach not only streamlines the synthetic pathway but also addresses long-standing challenges regarding regioselectivity and impurity control that have plagued earlier generations of synthesis protocols.
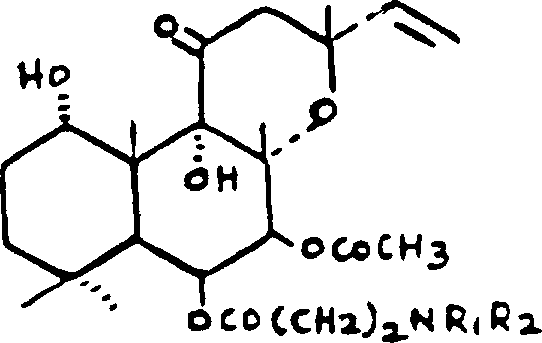
For R&D directors and process chemists, the significance of this patent extends beyond the mere creation of a new molecule; it represents a paradigm shift in how complex diterpenes can be functionalized. The ability to selectively modify the 6-position while maintaining the integrity of the sensitive 1,9-diol system is a testament to the sophistication of the protective group chemistry employed. By establishing a reliable sequence that moves from protection to selective deacetylation, followed by acylation and subsequent migration, the patent provides a blueprint for manufacturing high-purity pharmaceutical intermediates. This level of control is essential for meeting the stringent regulatory requirements of global health authorities, ensuring that the final active pharmaceutical ingredient (API) is free from structurally related impurities that could compromise patient safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in this patent, the synthesis of similar forskolin derivatives often relied on cumbersome and hazardous chemical environments. Historical methods, such as those referenced in Synthesis 711 (1989), utilized anhydrous aluminum chloride in ether and acetone to effect protection or transformation. These conditions are inherently problematic for large-scale manufacturing due to the exothermic nature of Lewis acid complexes and the difficulties associated with quenching and waste disposal. Furthermore, the lack of precise regiocontrol in these older pathways frequently resulted in complex mixtures of isomers, necessitating extensive chromatographic purification which drastically reduces overall throughput and increases production costs. The reliance on such aggressive reagents also poses significant supply chain risks, as the handling of moisture-sensitive Lewis acids requires specialized infrastructure and rigorous safety protocols that many contract manufacturing organizations prefer to avoid.
The Novel Approach
In stark contrast, the methodology presented in CN1033583C introduces a series of inventive steps that replace hazardous reagents with benign, scalable alternatives. The first major breakthrough is the formation of the 1,9-O-isopropylidene derivative using hydrochloric acid gas in anhydrous acetone at controlled low temperatures (0-5°C). This simple yet effective modification results in nearly quantitative formation of the protected intermediate, with yields reported between 96% and 97%. This high efficiency at the very beginning of the synthesis sets a positive tone for the entire campaign, minimizing material loss. Subsequent steps utilize common organic solvents like toluene and acetonitrile, along with standard bases such as triethylamine and sodium hydroxide, creating a process that is not only safer but also significantly more amenable to kilogram and tonne-scale production without the need for exotic catalysts or cryogenic conditions beyond simple ice baths.
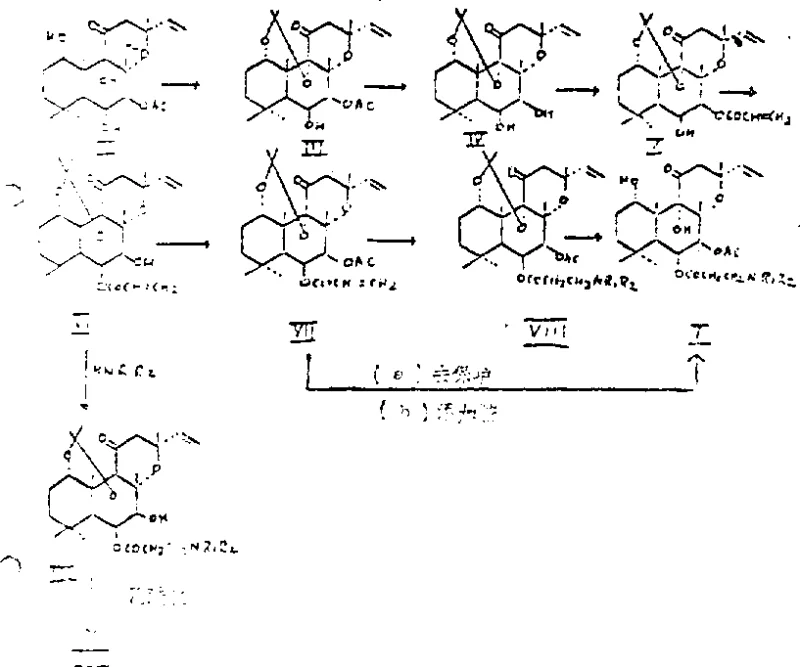
Mechanistic Insights into Regioselective Acylation and Migration
The heart of this synthetic strategy lies in the clever exploitation of steric and electronic effects facilitated by the 1,9-O-isopropylidene bridge. Once the forskolin core is protected, the 7-acetyl group is selectively removed using aqueous sodium hydroxide in methanol, exposing the 7-hydroxyl group while leaving the 6-hydroxyl shielded or less reactive due to the conformational constraints imposed by the protecting group. The subsequent acylation with beta-chloropropionyl chloride occurs exclusively at this newly freed 7-position. However, the true mechanistic elegance is revealed in the next step: the treatment of the 7-acylated intermediate with aqueous base in acetonitrile induces a migration of the acyl group from the 7-position to the 6-position. This intramolecular rearrangement is highly specific, yielding the 6-beta-acyloxy-7-deacetyl derivative in greater than 90% yield. This migration is critical because the biological activity of the target molecule is dependent on the substitution pattern at the 6-position, and achieving this directly without protecting group gymnastics would be nearly impossible.
From an impurity control perspective, this mechanism offers distinct advantages. By channeling the reaction through a specific migratory pathway, the formation of regioisomeric byproducts—where the acyl group might attach incorrectly at the 1, 9, or remaining 7 positions—is effectively suppressed. The crystalline nature of several intermediates, such as the 1,9-O-isopropylidene derivative (melting point 207-209°C) and the 7-deacetyl derivative (melting point 116-117°C), allows for purification via recrystallization rather than column chromatography. This solid-state purification capability is a hallmark of a robust industrial process, ensuring that the final API precursor meets stringent purity specifications. The final deprotection step, achieved by adjusting the pH to 1.0-3.5 with strong acid, cleanly removes the isopropylidene group to reveal the active diol system, completing the transformation with high fidelity.
How to Synthesize 6-(substituted aminopropionyl) Forskolin Derivatives Efficiently
The synthesis of these high-value cardiovascular intermediates requires strict adherence to the sequential logic defined in the patent to ensure optimal yield and purity. The process begins with the protection of the starting material, followed by a carefully orchestrated series of deprotection, acylation, migration, and amination steps. Each stage builds upon the structural modifications of the previous one, leveraging the unique reactivity of the forskolin scaffold. For process engineers and laboratory teams looking to replicate this success, understanding the critical parameters—such as temperature control during the HCl gas introduction and the specific solvent systems for the migration step—is paramount. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for execution.
- Protect forskolin at the 1,9-positions using acetone and hydrochloric acid gas at 0-5°C to form the 1,9-O-isopropylidene derivative.
- Perform selective deacetylation at the 7-position using aqueous sodium hydroxide in methanol to yield the 7-deacetyl intermediate.
- Acylation at the 7-hydroxyl group using beta-chloropropionyl chloride and triethylamine in toluene, followed by base-catalyzed migration to the 6-position.
- Re-acetylate the 7-position, substitute the chloro-group with the desired amine, and finally remove the 1,9-protecting group under acidic conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from academic-scale synthesis to commercial manufacturing is often fraught with hidden costs and logistical bottlenecks. The process described in CN1033583C addresses these pain points directly by prioritizing reagent availability and operational simplicity. By eliminating the need for expensive and hazardous Lewis acids like aluminum chloride, the process significantly reduces the cost of goods sold (COGS) associated with raw material procurement and hazardous waste disposal. The use of commodity chemicals such as acetone, toluene, and hydrochloric acid ensures that the supply chain remains resilient against market fluctuations, as these materials are universally available from multiple vendors globally. This diversification of supply sources mitigates the risk of production stoppages due to single-source dependency.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its high atom economy and exceptional yields at every critical juncture. With individual steps consistently achieving yields greater than 90% and an overall yield from forskolin to the key intermediate reaching approximately 70%, material wastage is minimized. This efficiency translates directly into lower production costs per kilogram of the final intermediate. Furthermore, the ability to purify intermediates through crystallization rather than expensive chromatographic techniques drastically reduces the consumption of silica gel and organic solvents, leading to substantial cost savings in both materials and waste treatment infrastructure.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is essential for maintaining uninterrupted supply to downstream API manufacturers. The process tolerances are well-defined, with reaction times ranging from 0.5 to 5 hours and temperatures easily maintained using standard industrial cooling systems (0-5°C). This predictability allows for accurate production scheduling and inventory management, reducing the lead time for high-purity pharmaceutical intermediates. The stability of the protected intermediates also allows for potential storage or transport between different manufacturing sites if a multi-site production strategy is employed.
- Scalability and Environmental Compliance: As regulatory pressure mounts on the pharmaceutical industry to reduce its environmental footprint, this process stands out for its green chemistry attributes. The replacement of ether and aluminum chloride with acetone and HCl gas eliminates the generation of aluminum-containing sludge, a difficult waste stream to manage. The solvents used, such as toluene and acetonitrile, are readily recoverable and recyclable through standard distillation processes. This alignment with environmental compliance standards facilitates easier permitting for new manufacturing facilities and supports the sustainability goals of modern pharmaceutical companies seeking eco-friendly supply chain partners.
Frequently Asked Questions (FAQ)
Navigating the complexities of custom synthesis for complex natural product derivatives often raises specific technical and commercial questions. The following answers are derived directly from the experimental data and claims within patent CN1033583C, addressing common concerns regarding yield, purity, and process scalability. These insights are intended to provide clarity for technical decision-makers evaluating the feasibility of integrating this technology into their existing supply chains.
Q: What is the primary advantage of using the 1,9-O-isopropylidene protecting group in this synthesis?
A: The use of the 1,9-O-isopropylidene protecting group allows for highly regioselective reactions at the 6 and 7 positions. It prevents unwanted side reactions at the 1 and 9 hydroxyls, enabling the specific migration of the acyl group from the 7-position to the 6-position, which is critical for the biological activity of the final cardiovascular agent.
Q: How does this process improve upon previous methods for synthesizing NKH-477 analogs?
A: Previous methods often relied on harsh Lewis acids like anhydrous aluminum chloride in ether, which posed safety risks and yielded lower purity. This patented process utilizes gaseous HCl in acetone for protection, achieving nearly quantitative yields (96-97%) and avoiding hazardous reagents, thereby simplifying downstream purification and waste treatment.
Q: What is the expected overall yield for the conversion of forskolin to the final protected intermediate?
A: The process demonstrates high efficiency across multiple steps. Specifically, the conversion from the starting forskolin material through to the 7-acetyl-6-acryloyl intermediate (Compound VII) achieves an overall yield of approximately 70%, with individual key steps such as the acylation and migration consistently exceeding 90% yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Forskolin Derivatives Supplier
The technical sophistication required to manufacture 6-beta-(3-substituted amino) propionyl forskolin derivatives demands a partner with deep expertise in complex organic synthesis and process optimization. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the safe handling of gaseous reagents and the precise temperature control necessary for the protection and migration steps. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and assay.
We understand that cost efficiency and speed to market are critical for your project's success. Our team is ready to collaborate with you to optimize this patented route for your specific volume requirements, ensuring a seamless transition from pilot scale to full commercialization. We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project needs. Reach out today to obtain specific COA data and comprehensive route feasibility assessments that will demonstrate how our manufacturing capabilities can drive value for your cardiovascular drug development program.
