Advanced Manufacturing of Benzothiophene Carboxylic Acid Amide Derivatives for PGD2 Antagonists
Introduction to Advanced PGD2 Antagonist Synthesis
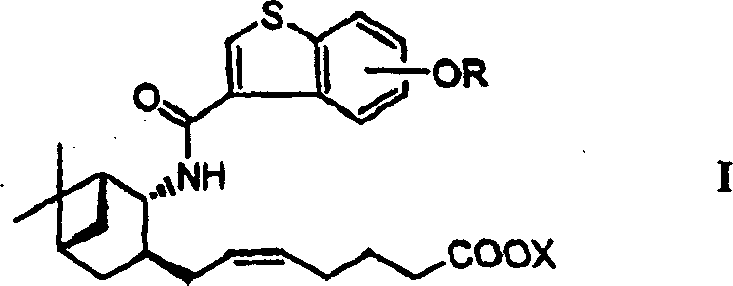
The pharmaceutical industry continuously seeks robust and scalable pathways for synthesizing complex therapeutic agents, particularly those targeting inflammatory and allergic responses. Patent CN1138772C discloses a highly efficient preparation method for benzothiophene carboxylic acid amide derivatives, which function as potent Prostaglandin D2 (PGD2) receptor antagonists. These compounds are critical intermediates in the development of treatments for systemic mastocytosis, asthma, allergic rhinitis, and atopic dermatitis. The disclosed technology represents a significant leap forward in process chemistry, addressing long-standing issues related to toxicity, yield, and operational complexity found in earlier synthetic routes. By integrating novel oxidation and reduction strategies, this methodology ensures the production of high-purity active pharmaceutical ingredients (APIs) suitable for clinical application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar benzothiophene derivatives relied heavily on oxidation protocols that posed severe environmental and safety hazards. Traditional methods frequently employed chromium-based oxidants, such as Jones reagent or pyridinium chlorochromate, which generate toxic heavy metal waste requiring expensive and complex detoxification procedures. Furthermore, alternative oxidation techniques like Swern oxidation necessitated the use of dimethyl sulfoxide and oxalyl chloride at cryogenic temperatures ranging from -50°C to -78°C. These extreme conditions not only increased energy consumption but also generated harmful byproducts like carbon monoxide and sulfur-containing odors, creating unsafe working environments. Additionally, previous reduction methods for preparing the chiral amino alcohol precursors suffered from poor stereocontrol and low yields, often reported around 39.6%, making them economically unviable for large-scale manufacturing.
The Novel Approach
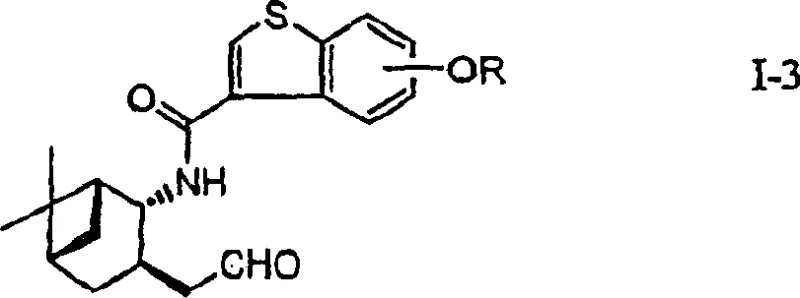
The innovative process outlined in the patent data fundamentally transforms the synthesis landscape by introducing a TEMPO-catalyzed oxidation system. This method utilizes 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) in the presence of an oxyhaloacid, such as sodium hypochlorite, to convert primary alcohols to aldehydes quantitatively. This shift eliminates the need for toxic chromium reagents and cryogenic conditions, allowing reactions to proceed safely from ice-cooling to room temperature. Moreover, the patent introduces a superior reduction strategy for the amino alcohol intermediate, employing a borohydride reducing agent combined with a Lewis acid. This combination achieves high stereoselectivity exceeding 99% and yields around 89%, effectively resolving the inefficiencies of prior art. The overall route avoids silica gel chromatography, streamlining purification and enhancing suitability for industrial scale-up.
Mechanistic Insights into TEMPO-Catalyzed Oxidation and Stereoselective Reduction
The core chemical innovation lies in the mechanistic efficiency of the TEMPO-mediated oxidation cycle. In this catalytic system, the nitroxyl radical of TEMPO acts as the active oxidant, converting the primary alcohol substrate into the corresponding aldehyde while being reduced to the hydroxylamine. The oxyhaloacid, typically sodium hypochlorite buffered to a pH of 8.5-9.5, serves as the terminal oxidant to regenerate the active nitroxyl radical from the hydroxylamine species. This catalytic turnover allows for the use of minimal catalyst loading, typically between 0.2 to 1 mol%, while driving the reaction to completion with high atom economy. The reaction proceeds rapidly in biphasic solvent systems involving ethyl acetate and water, facilitating easy separation of the organic product without the need for complex extraction protocols associated with heavy metal residues.
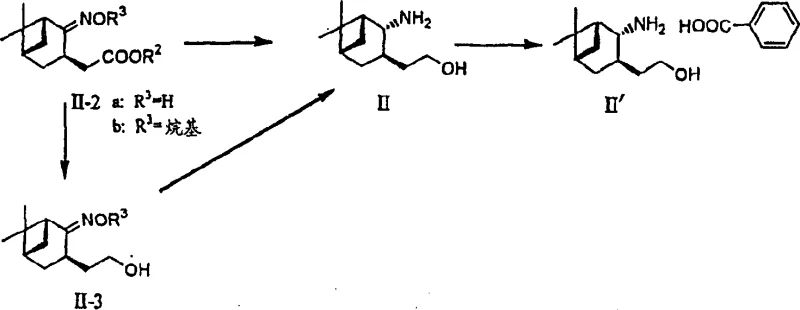
Complementing the oxidation step is the highly stereoselective reduction of the oxime ester intermediate to the chiral amino alcohol. The mechanism involves the activation of the oxime and ester functionalities by a Lewis acid, such as aluminum chloride or titanium tetrachloride, which coordinates with the oxygen atoms to increase electrophilicity. Subsequent hydride delivery from a borohydride source, such as sodium borohydride, occurs with precise facial selectivity dictated by the rigid bicyclic framework of the norpinane skeleton. This synergistic interaction between the Lewis acid and the hydride donor ensures that both the oxime and ester groups are reduced simultaneously to the amine and alcohol, respectively, without compromising the stereochemical integrity of the molecule. This results in the formation of the desired (1R, 2R, 3R, 5S) configuration with exceptional purity, as confirmed by HPLC analysis showing purity levels above 99.2%.
How to Synthesize Benzothiophene Carboxylic Acid Amide Efficiently
The synthesis of these high-value pharmaceutical intermediates requires precise control over reaction parameters to maintain stereochemical fidelity and maximize throughput. The process begins with the acylation of the chiral amino alcohol with a protected benzothiophene carboxylic acid derivative, followed by the critical TEMPO oxidation step. Detailed operational procedures involve maintaining specific pH levels during oxidation and careful temperature control during the Lewis acid-mediated reduction to prevent side reactions. The subsequent Wittig olefination installs the requisite heptenoic acid side chain, completing the carbon skeleton of the target molecule. For a comprehensive understanding of the specific reagent quantities, solvent choices, and workup procedures required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide provided below.
- Couple the chiral amino alcohol intermediate with the protected benzothiophene carboxylic acid chloride to form the amide linkage.
- Oxidize the resulting primary alcohol to an aldehyde using a TEMPO catalyst and sodium hypochlorite under mild conditions.
- Perform a Wittig reaction with a phosphonium ylide to install the heptenoic acid side chain, followed by deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers substantial benefits by fundamentally altering the cost structure and risk profile of the supply chain. The elimination of hazardous chromium reagents and cryogenic cooling requirements translates directly into reduced operational expenditures and lower regulatory compliance burdens. By shifting to ambient temperature reactions and benign solvent systems, manufacturers can significantly lower energy costs and minimize the need for specialized containment equipment. Furthermore, the high yields and stereoselectivity reduce the consumption of raw materials per kilogram of final product, optimizing the overall material balance and reducing waste disposal fees. These factors collectively contribute to a more resilient and cost-effective supply chain for critical asthma and allergy medication intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous oxidants with commodity chemicals like sodium hypochlorite drastically reduces raw material costs. Additionally, the high yield of the reduction step minimizes the loss of valuable chiral starting materials, ensuring that every gram of input contributes effectively to the final output. The avoidance of silica gel chromatography further reduces consumable costs and processing time, allowing for faster batch turnover and improved capital efficiency in production facilities.
- Enhanced Supply Chain Reliability: The use of stable and readily available reagents ensures consistent supply availability, mitigating the risks associated with sourcing specialized or controlled chemicals. The robustness of the reaction conditions means that production is less susceptible to interruptions caused by equipment failures related to extreme temperature maintenance. This reliability supports continuous manufacturing schedules and ensures timely delivery of intermediates to downstream API producers, strengthening the overall security of the pharmaceutical supply network.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing simple extraction and crystallization techniques rather than complex chromatographic separations. This simplicity facilitates the transition from pilot plant to commercial scale production without significant re-engineering. Moreover, the generation of benign byproducts like table salt instead of heavy metal waste simplifies effluent treatment and ensures compliance with increasingly stringent environmental regulations, safeguarding the manufacturer's social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement officers assessing vendor capabilities. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-making purposes. These insights highlight the practical advantages of adopting this advanced manufacturing technology over legacy methods.
Q: What are the advantages of the TEMPO oxidation method over traditional chromium reagents?
A: The TEMPO-catalyzed oxidation using oxyhaloacids eliminates the use of toxic chromium(VI) reagents, significantly reducing hazardous waste disposal costs and improving operator safety while maintaining quantitative yields.
Q: How does the new reduction method improve stereoselectivity?
A: By utilizing a borohydride reducing agent in combination with a Lewis acid, the process achieves over 99% stereoselectivity and approximately 89% yield, vastly outperforming previous methods which suffered from low yields around 39.6%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability by avoiding silica gel chromatography, using inexpensive reagents like sodium hypochlorite, and employing safe solvent systems like ethyl acetate and water.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiophene Carboxylic Acid Amide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging deep expertise in complex organic synthesis to deliver high-quality pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN1138772C can be executed with precision and consistency. We adhere to stringent purity specifications and utilize rigorous QC labs to verify the stereochemical integrity and chemical purity of every batch, guaranteeing that our products meet the exacting standards required for global pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless integration of these high-performance intermediates into your drug development pipeline.
