Advanced Biocatalytic Production of Chiral Acetoin and 2,3-Butanediol for Industrial Scale-Up
Advanced Biocatalytic Production of Chiral Acetoin and 2,3-Butanediol for Industrial Scale-Up
The landscape of chiral chemical synthesis is undergoing a profound transformation, driven by the urgent need for sustainable, high-purity intermediates in the flavor, fragrance, and pharmaceutical sectors. A pivotal advancement in this domain is documented in patent CN101565685A, which discloses a groundbreaking gene recombinant bacterium, specifically Escherichia coli BL21(pETDuet-ydjLnox). This engineered strain represents a significant leap forward in biocatalytic efficiency, capable of co-expressing 2R,3R-butanediol dehydrogenase (2R,3R-BDH) and NADH oxidase (NOX) to facilitate the precise production of chiral pure acetoin (AC) and 2,3-butanediol (BD). For R&D directors and procurement specialists seeking a reliable flavor & fragrance intermediate supplier, this technology offers a robust alternative to traditional chemical synthesis, promising superior stereocontrol and operational simplicity. The preservation of this strain under number CCTCC NO: M 208259 underscores its reproducibility and readiness for industrial application, marking a critical milestone in the commercialization of enzymatic resolution processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active acetoin and butanediol has been fraught with significant technical and economic challenges that hinder large-scale adoption. Traditional chemical approaches, such as the asymmetric hydrogenation of diacetyl using platinum catalysts modified with cinchona alkaloids, often suffer from moderate enantiomeric excess values ranging typically between 70% and 90%, which is insufficient for high-end pharmaceutical applications without further purification. Furthermore, these heterogeneous reactions require high-pressure equipment and expensive heavy metal catalysts, introducing severe safety risks and environmental burdens related to metal contamination and disposal. On the biological front, wild-type fermentation using strains like Klebsiella pneumoniae or Enterobacter aerogenes typically yields a complex mixture of stereoisomers, including meso-BD, 2R,3R-BD, and 2S,3S-BD, making downstream separation costly and inefficient. Additionally, previous attempts at enzymatic synthesis faced critical bottlenecks, such as the toxicity of substrates like diacetyl to microbial cells and the inability to economically regenerate essential cofactors like NADPH, leading to low product concentrations and poor overall yields that fail to meet commercial viability standards.
The Novel Approach
The methodology presented in CN101565685A fundamentally addresses these legacy issues through a sophisticated metabolic engineering strategy that decouples substrate toxicity from product formation while ensuring efficient cofactor cycling. By constructing a recombinant E. coli host that co-expresses the ydjL gene from Bacillus subtilis and the nox gene from Lactobacillus brevis, the inventors have created a self-sustaining biocatalytic system. This novel approach allows for the direct transformation of inexpensive 2,3-butanediol isomers into high-value chiral acetoin or the kinetic resolution of racemic mixtures to isolate pure 2S,3S-BD. Unlike previous methods that struggled with cofactor depletion, this system integrates an NADH oxidase that continuously regenerates NAD+ from NADH using molecular oxygen, effectively driving the reaction equilibrium towards the desired product without the need for expensive external cofactor supplementation. This results in a process that is not only chemically elegant but also economically superior, enabling product concentrations exceeding 6 grams per liter with exceptional optical purity, thereby establishing a new benchmark for cost reduction in chiral chemical manufacturing.
Mechanistic Insights into Dual-Enzyme Cofactor Regeneration
The core innovation of this technology lies in the synergistic coupling of two distinct enzymatic activities within a single whole-cell catalyst, creating a closed-loop redox system that maximizes atomic economy and reaction efficiency. The 2R,3R-butanediol dehydrogenase (2R,3R-BDH) acts as the primary stereoselective driver, catalyzing the reversible oxidation of specific 2,3-butanediol stereoisomers into their corresponding acetoin forms while reducing NAD+ to NADH. However, in isolated enzyme systems, the accumulation of NADH would rapidly inhibit the dehydrogenase activity, stalling the reaction. This limitation is elegantly overcome by the concurrent expression of NADH oxidase (NOX), which utilizes dissolved oxygen to oxidize the accumulated NADH back to NAD+, producing water as the only byproduct. This mechanism ensures a constant supply of the oxidized cofactor required by the dehydrogenase, allowing the reaction to proceed to high conversion rates without the stoichiometric consumption of expensive nicotinamide cofactors.
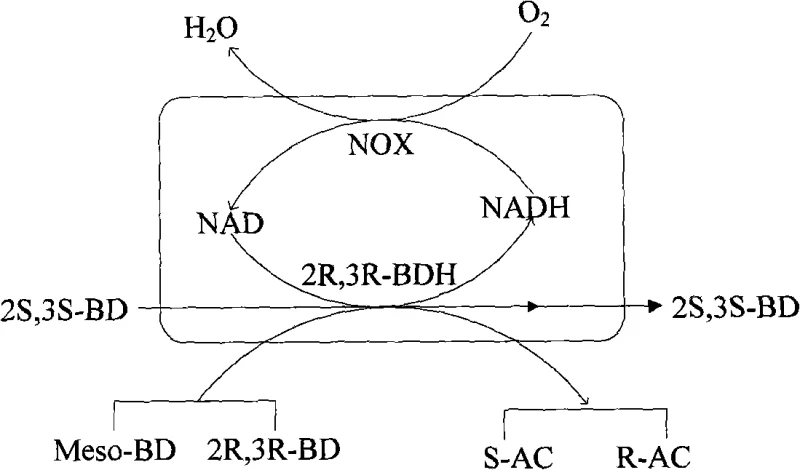
From a stereochemical perspective, the 2R,3R-BDH enzyme exhibits remarkable substrate specificity, distinguishing between the subtle structural differences of the butanediol isomers. It efficiently catalyzes the oxidation of meso-2,3-BD to S-acetoin and 2R,3R-BD to R-acetoin, while notably leaving the 2S,3S-BD isomer untouched. This differential reactivity is the mechanistic basis for the kinetic resolution of 2,3-BD mixtures, allowing for the enrichment of 2S,3S-BD in the reaction vessel as the other isomers are consumed. The impurity profile is inherently controlled by this enzymatic specificity; since the biocatalyst does not recognize the 2S,3S-BD substrate, side reactions leading to unwanted stereoisomers are minimized, resulting in product streams with ee values consistently above 96% for acetoin and 98% for 2S,3S-BD. This high level of intrinsic selectivity reduces the burden on downstream purification units, simplifying the overall process flow and enhancing the final quality of the high-purity OLED material or flavor intermediate produced.
How to Synthesize Chiral Acetoin Efficiently
The implementation of this biocatalytic route involves a streamlined series of fermentation and transformation steps designed for scalability and ease of operation in an industrial setting. The process begins with the aerobic cultivation of the recombinant E. coli BL21(pETDuet-ydjLnox) strain in a defined medium supplemented with ampicillin to maintain plasmid stability, followed by induction with IPTG to trigger the overexpression of the target enzymes. Once the cells reach the desired biomass and enzymatic activity, they are harvested and utilized as resting whole-cell catalysts, eliminating the need for complex enzyme purification procedures. The actual bioconversion is conducted in a buffered aqueous system where the substrate concentration, pH, and temperature are tightly controlled to optimize the reaction kinetics and stability of the biocatalyst.
- Cultivate the recombinant E. coli BL21(pETDuet-ydjLnox) strain in LB medium containing ampicillin at 37°C until the optical density indicates sufficient growth, followed by induction with IPTG at lower temperatures (10-30°C) to express 2R,3R-BDH and NOX enzymes.
- Harvest the whole cells via centrifugation, wash with PBS buffer, and resuspend in physiological saline to create the resting cell biocatalyst suspension ready for transformation reactions.
- Mix the biocatalyst with the specific substrate (meso-BD, 2R,3R-BD, or 2,3-BD mixture) in a buffered solution at controlled pH (6.0-9.0) and temperature (20-40°C), allowing the enzymatic conversion to proceed with continuous shaking.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this recombinant biocatalytic process offers substantial strategic benefits that extend far beyond simple yield improvements. The elimination of precious metal catalysts, such as platinum, removes a major source of raw material cost volatility and supply risk, while also obviating the need for expensive metal scavenging steps that are mandatory in traditional chemical synthesis to meet regulatory limits for heavy metals in consumer products. Furthermore, the use of a robust E. coli host organism ensures that the biocatalyst can be produced reliably at scale using standard fermentation infrastructure, significantly reducing lead time for high-purity intermediates compared to sourcing complex organometallic catalysts. The simplicity of the downstream processing, which involves merely separating the whole cells via centrifugation or filtration followed by distillation, translates directly into lower capital expenditure (CAPEX) and operational expenditure (OPEX) for manufacturing facilities.
- Cost Reduction in Manufacturing: The integrated cofactor regeneration system driven by NADH oxidase eliminates the recurring cost of purchasing stoichiometric amounts of NAD+ or NADP+, which are among the most expensive reagents in biocatalysis. By recycling the cofactor thousands of times within the reaction cycle, the effective cost per mole of product is drastically reduced, making the process economically competitive with petrochemical routes even at moderate scales. Additionally, the ability to use crude or mixed isomer feedstocks of 2,3-butanediol as substrates allows manufacturers to leverage lower-grade, cheaper raw materials that would otherwise be unsuitable for high-precision chemical synthesis, further driving down the total cost of goods sold.
- Enhanced Supply Chain Reliability: Relying on a genetically defined strain preserved in a public culture collection mitigates the risk of supply disruption associated with proprietary catalyst vendors or geographically concentrated mining operations for rare metals. The fermentation-based production of the biocatalyst itself is highly scalable and can be ramped up quickly to meet surges in demand, ensuring a consistent and secure supply of the critical enzymatic machinery needed for production. This biological manufacturing platform is also less susceptible to the geopolitical and logistical fluctuations that often impact the availability of specialized chemical reagents, providing a more resilient foundation for long-term supply agreements.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic reaction and the absence of toxic organic solvents or heavy metals align perfectly with increasingly stringent global environmental regulations, such as REACH and TSCA. The waste stream from this process is primarily biological biomass and water, which are significantly easier and cheaper to treat than the hazardous waste generated by traditional chemical hydrogenation processes. This green chemistry profile not only reduces compliance costs but also enhances the brand value of the final product for end-users in the food and pharmaceutical industries who are actively seeking sustainable and eco-friendly supply chains for their ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recombinant biocatalytic technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational parameters and performance capabilities of the E. coli BL21(pETDuet-ydjLnox) system for potential licensees and manufacturing partners. Understanding these specifics is crucial for assessing the feasibility of integrating this route into existing production lines or developing new facilities dedicated to chiral synthesis.
Q: What is the optical purity achievable with this biocatalytic method?
A: According to patent CN101565685A, the recombinant E. coli system can produce chiral acetoin with an enantiomeric excess (ee) value of greater than or equal to 96%, and chiral 2S,3S-butanediol with an ee value of greater than or equal to 98%.
Q: How does the cofactor regeneration system work in this process?
A: The process utilizes a dual-enzyme system where NADH oxidase (NOX) regenerates the essential NAD+ cofactor from NADH using molecular oxygen, eliminating the need for expensive external cofactor addition and driving the reaction equilibrium forward.
Q: Can this method resolve racemic mixtures of 2,3-butanediol?
A: Yes, the engineered strain is capable of kinetically resolving 2,3-butanediol mixtures. It selectively converts meso-BD and 2R,3R-BD into their corresponding acetoin forms, leaving behind highly enriched 2S,3S-butanediol in the reaction mixture.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Acetoin Supplier
The technological potential demonstrated in patent CN101565685A represents exactly the kind of innovative capability that NINGBO INNO PHARMCHEM brings to the global market as a premier CDMO partner. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs like this dual-enzyme system are successfully translated into robust, GMP-compliant manufacturing processes. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, including the precise determination of enantiomeric excess values required for high-value flavor and pharmaceutical intermediates. We understand that consistency is key, and our quality management systems are designed to guarantee batch-to-batch reproducibility that meets the exacting standards of multinational corporations.
We invite you to engage with our technical procurement team to discuss how this biocatalytic route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this enzymatic process compared to your current supply chain. We encourage potential partners to contact us directly to obtain specific COA data from our pilot runs and to receive detailed route feasibility assessments that demonstrate our commitment to delivering high-quality, cost-effective solutions for the complex challenges of modern chemical synthesis.
