Advanced Sm-Cu Catalyzed Synthesis of 4,4'-Biphenol for Commercial Scale-Up
Advanced Sm-Cu Catalyzed Synthesis of 4,4'-Biphenol for Commercial Scale-Up
The chemical industry is constantly seeking more efficient and environmentally benign pathways for producing high-value intermediates like 4,4'-biphenol, a critical monomer for liquid crystal polymers and engineering plastics. Patent CN108715574B introduces a groundbreaking methodology that utilizes a synergistic Samarium-Copper (Sm-Cu) catalytic system within an ionic liquid medium to achieve this transformation. This novel approach represents a significant departure from traditional harsh synthetic routes, offering a streamlined one-step reductive coupling of p-halophenols. By leveraging the unique redox properties of rare earth metals combined with the green solvent characteristics of imidazolium salts, this technology addresses key pain points regarding waste generation and energy consumption. For R&D directors and process chemists, this patent provides a robust framework for developing scalable, high-yield processes that align with modern green chemistry principles while maintaining exceptional product purity standards required for advanced material applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 4,4'-biphenol has relied on cumbersome and hazardous methodologies such as the benzidine rearrangement or biphenyl sulfonation followed by alkali fusion. These legacy processes are fraught with significant operational challenges, including the requirement for extreme temperatures, high pressures, and the use of corrosive reagents like concentrated sulfuric acid and molten alkalis. Furthermore, traditional routes often suffer from poor atom economy and generate substantial quantities of toxic wastewater and solid waste, creating severe environmental compliance burdens for manufacturers. The reliance on noble metal catalysts in some newer variations also introduces prohibitive costs and supply chain vulnerabilities associated with precious metal procurement. Consequently, finding a method that avoids these harsh conditions while delivering high purity has been a longstanding objective for chemical engineers aiming to optimize production efficiency and reduce the overall environmental footprint of fine chemical manufacturing facilities.
The Novel Approach
In stark contrast to these archaic methods, the technology disclosed in patent CN108715574B employs a mild, room-temperature reductive coupling strategy driven by inexpensive copper salts and samarium metal. This innovative route eliminates the need for extreme thermal inputs or high-pressure equipment, thereby drastically simplifying the reactor design and operational safety protocols. The use of p-chlorophenol as a starting material is particularly advantageous due to its low cost and commercial availability, yet it is typically inert under standard coupling conditions without specialized activation. By utilizing an ionic liquid solvent system, the reaction achieves excellent solubility for the catalyst precursors, facilitating a homogeneous reaction environment that promotes efficient conversion. This shift towards milder conditions not only enhances operator safety but also significantly reduces energy consumption, making it an attractive alternative for companies seeking to modernize their production lines and achieve sustainability goals without compromising on yield or product quality.
Mechanistic Insights into Sm-Cu Catalyzed Reductive Coupling
The core of this technological breakthrough lies in the synergistic interaction between the divalent copper salt and the metallic samarium promoter within the ionic liquid matrix. Mechanistically, the samarium metal acts as a potent single-electron reductant, initiating the formation of radical intermediates from the aryl halide substrate through a single electron transfer (SET) process. The copper species, soluble in the ionic liquid but insoluble in common organic solvents, likely serves to mediate the coupling of these radical species, stabilizing the transition state and directing the formation of the biaryl bond. This specific catalytic cycle avoids the formation of homocoupled byproducts often seen in uncontrolled radical reactions, ensuring high selectivity for the desired 4,4'-isomer. The ionic liquid itself plays a dual role as both solvent and stabilizer, potentially coordinating with the metal centers to maintain their active oxidation states throughout the reaction duration. Understanding this mechanistic nuance is crucial for process chemists aiming to further optimize reaction parameters such as stoichiometry and mixing rates to maximize throughput in a commercial setting.
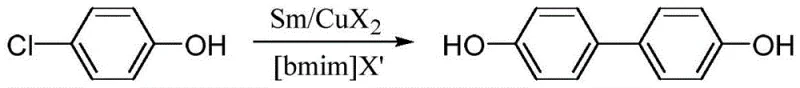
Controlling the impurity profile is paramount when synthesizing intermediates for high-performance polymers, and this method offers distinct advantages in that regard. The mild reaction conditions inherently suppress thermal degradation pathways that often lead to complex tar formation or oligomerization in high-temperature processes. Additionally, the specific solvation properties of the [bmim]Cl ionic liquid appear to inhibit side reactions such as dehalogenation or over-reduction, which are common pitfalls in metal-mediated couplings. The subsequent workup procedure, involving extraction and column chromatography, allows for the effective removal of metal residues and unreacted starting materials, yielding a product with the high purity necessary for downstream polymerization. For quality assurance teams, this implies a more predictable and manageable impurity spectrum, reducing the burden on analytical testing and purification steps. The ability to recycle the ionic liquid further ensures that cross-contamination risks are minimized between batches, supporting consistent quality output essential for regulatory compliance in pharmaceutical and electronic material supply chains.
How to Synthesize 4,4'-Biphenol Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating high-purity 4,4'-biphenol suitable for immediate scale-up evaluation. The process begins with the rigorous drying of the ionic liquid solvent to ensure anhydrous conditions, which is critical for preventing the quenching of the reactive samarium species. Following the dissolution of the p-chlorophenol substrate, the sequential addition of the metal reductant and copper catalyst initiates the exothermic coupling reaction, which proceeds smoothly at ambient temperature. Operators should monitor the reaction progress via TLC or HPLC to determine the optimal endpoint, typically achieved within several hours depending on the specific batch size and mixing efficiency. The detailed standardized synthesis steps below provide a comprehensive guide for implementing this technology in a pilot or production plant environment, ensuring that all critical parameters are controlled to achieve the reported yields of 70-95%.
- Dissolve p-chlorophenol in dried ionic liquid [bmim]Cl under stirring to form a homogeneous mixture.
- Sequentially add activated samarium metal powder and anhydrous copper chloride catalyst to the reaction vessel.
- Stir the mixture at room temperature under anhydrous conditions until completion, then extract and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this Sm-Cu catalyzed process offers compelling economic and logistical benefits that extend beyond simple yield improvements. The substitution of expensive noble metal catalysts like palladium with abundant and low-cost copper salts results in a direct reduction in raw material expenditure, significantly lowering the cost of goods sold (COGS). Furthermore, the elimination of volatile organic solvents in favor of recyclable ionic liquids reduces the costs associated with solvent purchase, storage, and hazardous waste disposal. This shift not only improves the bottom line but also mitigates regulatory risks related to environmental emissions, ensuring long-term operational continuity in regions with strict environmental laws. The simplified workup and purification requirements further contribute to cost efficiency by reducing labor hours and consumable usage during the isolation phase, making the overall process leaner and more competitive in the global market.
- Cost Reduction in Manufacturing: The economic impact of replacing precious metal catalysts with base metal alternatives cannot be overstated in large-scale production scenarios. Copper chloride is vastly cheaper and more readily available than palladium or platinum complexes, removing a major variable cost driver from the manufacturing equation. Additionally, the ability to recover and reuse the ionic liquid solvent multiple times without significant loss of performance creates a closed-loop system that minimizes material waste. This circular approach to solvent management drastically cuts down on recurring procurement costs for solvents and the fees associated with treating spent solvent waste. By streamlining the reaction to a single step at room temperature, the process also reduces energy consumption for heating and cooling, contributing to lower utility bills and a smaller carbon footprint for the manufacturing facility.
- Enhanced Supply Chain Reliability: Relying on widely available commodity chemicals like p-chlorophenol and copper salts insulates the supply chain from the volatility often seen in the market for specialty reagents and rare earth metals. Unlike processes dependent on imported catalysts with long lead times, the raw materials for this synthesis are sourced from robust domestic and international supply networks. The mild operating conditions also mean that the process is less susceptible to disruptions caused by equipment failure or utility fluctuations, as it does not require complex high-pressure or cryogenic systems. This inherent robustness ensures a more stable and predictable production schedule, allowing supply chain managers to commit to delivery timelines with greater confidence and reduce the need for excessive safety stock inventory.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this room-temperature reaction is inherently easier to manage in larger vessels. The absence of exothermic runaway risks associated with high-energy reagents simplifies the engineering controls required for scale-up, reducing capital expenditure on specialized reactor infrastructure. From an environmental perspective, the use of non-volatile ionic liquids aligns perfectly with increasingly stringent global regulations on VOC emissions and green chemistry initiatives. This compliance advantage future-proofs the manufacturing asset against tightening environmental legislation, avoiding potential fines or forced shutdowns. The simplified waste stream, primarily consisting of recoverable metals and reusable solvents, facilitates easier treatment and disposal, further enhancing the sustainability profile of the operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process feasibility and product quality. Understanding these details is essential for stakeholders evaluating the potential integration of this technology into existing production portfolios. The answers reflect the practical realities of operating this specific catalytic system and highlight the key differentiators that set it apart from conventional manufacturing methods.
Q: What are the advantages of using ionic liquids in 4,4'-biphenol synthesis?
A: Ionic liquids like [bmim]Cl serve as green solvents that can be recycled and reused multiple times without significant loss of efficiency. They provide a unique solvation environment that enhances the solubility of copper catalysts while minimizing volatile organic compound (VOC) emissions compared to traditional solvents.
Q: Why is Samarium preferred over Magnesium or Zinc for this coupling?
A: Samarium exhibits superior reactivity for the reductive coupling of inert aryl chlorides under mild conditions where magnesium or zinc often fail. Its specific redox potential allows for effective activation of the carbon-chlorine bond at room temperature, leading to higher yields and fewer side reactions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates at room temperature and atmospheric pressure, eliminating the need for expensive high-pressure reactors or extreme heating. The simplicity of the workup and the ability to recover the ionic liquid solvent make it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Biphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the Sm-Cu mediated coupling described in patent CN108715574B for producing high-performance chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle sensitive organometallic reactions and ionic liquid systems with the utmost precision, adhering to stringent purity specifications required by the pharmaceutical and advanced materials sectors. With our rigorous QC labs and dedicated process development teams, we guarantee that every batch of 4,4'-biphenol meets the highest standards of quality and consistency, providing our partners with a secure and reliable source for their critical supply chains.
We invite forward-thinking organizations to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for their specific applications. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can drive value and efficiency in your 4,4'-biphenol sourcing strategy.
