Advanced Catalytic Synthesis of 2-Hydroxy-5-Amidobenzophenones for Commercial Pharmaceutical Applications
Advanced Catalytic Synthesis of 2-Hydroxy-5-Amidobenzophenones for Commercial Pharmaceutical Applications
The chemical landscape for producing critical intermediates is undergoing a significant transformation driven by the need for greener, more efficient catalytic systems. Patent CN1618787A introduces a groundbreaking methodology for the synthesis of 2-hydroxy-5-amidobenzophenone compounds, which serve as vital building blocks in the pharmaceutical and agrochemical industries. This technology replaces traditional, waste-intensive stoichiometric Lewis acids with a catalytic amount of Ytterbium Trifluoromethanesulfonate [Yb(OTf)3]. By shifting from conventional aluminum-based catalysts to this lanthanide triflate system, manufacturers can achieve superior reaction control, enhanced product purity, and a drastically reduced environmental footprint. This report analyzes the technical merits and commercial viability of this process for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-hydroxy-5-amidobenzophenone derivatives has relied heavily on traditional Lewis acids such as Aluminum Trichloride (AlCl3). While effective in promoting Friedel-Crafts acylation, these conventional catalysts present severe operational drawbacks for modern manufacturing. The primary issue is the requirement for stoichiometric or even excess amounts of the catalyst, often exceeding a 1:1 molar ratio with the substrate, which generates massive quantities of aluminum-containing waste upon hydrolysis. Furthermore, AlCl3 is extremely moisture-sensitive, necessitating strictly anhydrous conditions that increase energy consumption and equipment complexity. The post-reaction workup is notoriously difficult, often involving acidic quenches that complicate product isolation and lead to lower overall yields and purity profiles due to side reactions and emulsion formation during extraction.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes Ytterbium Trifluoromethanesulfonate [Yb(OTf)3] as a highly efficient Lewis acid catalyst. This method allows for the reaction between 4-alkoxybenzamides and acyl halides to proceed with catalytic loading as low as 0.02 molar equivalents. The reaction operates under much milder conditions, typically between 20°C and 150°C, with preferred ranges of 50°C to 80°C, ensuring thermal stability of sensitive functional groups. As illustrated in the reaction scheme below, the process facilitates the direct acylation to form the target benzophenone structure with exceptional regioselectivity. This shift not only simplifies the reaction setup but also enables a straightforward workup procedure involving simple filtration and ice-water quenching, resulting in high-purity solids directly from the reactor.
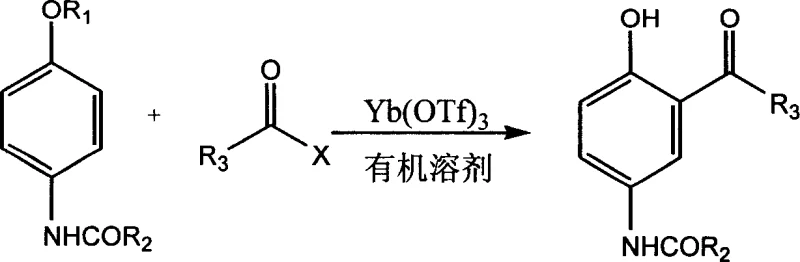
Mechanistic Insights into Yb(OTf)3-Catalyzed Acylation
The efficacy of Yb(OTf)3 lies in its unique electronic properties as a lanthanide triflate. Unlike traditional Lewis acids that form stable complexes with products and require stoichiometric quantities to drive the reaction forward, Yb(OTf)3 acts as a water-tolerant Lewis acid that activates the acyl halide electrophile without being permanently consumed. The Ytterbium ion coordinates with the carbonyl oxygen of the acyl halide, increasing the electrophilicity of the carbonyl carbon and facilitating nucleophilic attack by the electron-rich aromatic ring of the 4-alkoxybenzamide. This mechanism proceeds through a stabilized transition state that minimizes side reactions such as polyacylation or demethylation, which are common pitfalls in harsh acidic environments. The result is a clean conversion profile that maintains the integrity of the amide and ether functionalities present in the starting materials.
Furthermore, the impurity control mechanism inherent in this catalytic system is robust. The high selectivity of the Yb(OTf)3 catalyst ensures that the acylation occurs predominantly at the ortho-position relative to the alkoxy group, driven by the directing effects of the substituents and the specific coordination geometry of the catalyst. This regioselectivity is crucial for pharmaceutical applications where isomeric impurities can be difficult to separate and may pose toxicological risks. The patent data demonstrates that across a wide range of substrates, including those with varying alkyl chains and heterocyclic groups, the purity remains consistently above 99%. This level of chemical fidelity reduces the burden on downstream purification units, such as recrystallization or chromatography, thereby streamlining the entire production workflow.
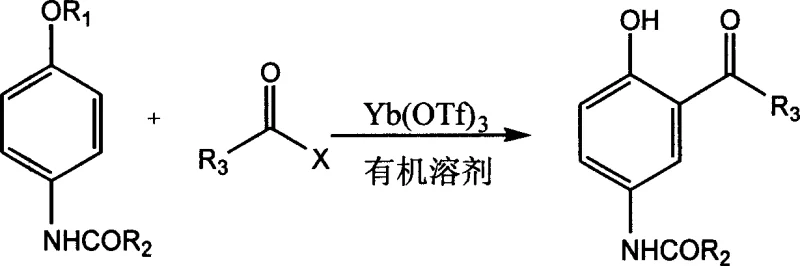
How to Synthesize 2-Hydroxy-5-Amidobenzophenone Efficiently
To implement this synthesis effectively, operators must adhere to precise molar ratios and temperature controls to maximize catalyst turnover and product recovery. The process begins by dissolving the 4-alkoxybenzamide substrate in a suitable organic solvent, such as dichloromethane or nitrobenzene, followed by the addition of the acyl halide and the Yb(OTf)3 catalyst. The reaction mixture is then heated to the optimal temperature range, typically 50°C to 80°C, and maintained for 2 to 10 hours while monitoring conversion via HPLC. Upon completion, the solvent is removed, and the residue is treated with ice water to precipitate the product, which is then filtered and dried. The detailed standardized synthesis steps are provided in the guide below.
- Dissolve 4-alkoxybenzamide and Yb(OTf)3 catalyst in an organic solvent such as dichloromethane at room temperature.
- Add the acyl halide reagent and heat the mixture to 50-80°C for 2-10 hours while monitoring reaction progress via HPLC.
- Quench the reaction with ice water, filter the precipitate to isolate the crude product, and recover the catalyst from the filtrate by dehydration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Yb(OTf)3 catalytic process offers substantial strategic advantages over legacy manufacturing routes. The most significant benefit is the drastic reduction in raw material consumption, specifically regarding the catalyst. Since the ytterbium catalyst is used in sub-stoichiometric amounts and is recoverable, the recurring cost of catalyst purchase is significantly lowered compared to the continuous consumption of aluminum chloride. Moreover, the simplified workup procedure eliminates the need for complex aqueous extractions and neutralization steps, which translates to reduced labor hours, lower utility consumption for wastewater treatment, and faster batch cycle times. These operational efficiencies collectively contribute to a more resilient and cost-effective supply chain for high-value intermediates.
- Cost Reduction in Manufacturing: The transition to a catalytic system fundamentally alters the cost structure of production. By utilizing Yb(OTf)3 at loadings as low as 0.02 equivalents, the process avoids the massive material costs associated with stoichiometric Lewis acids. Additionally, the patent highlights a catalyst recovery rate of approximately 95%, meaning the expensive lanthanide metal can be recycled repeatedly, further amortizing the initial investment. This reduction in consumable materials, combined with the elimination of extensive waste disposal fees associated with aluminum sludge, results in a leaner manufacturing model that protects margins against raw material price volatility.
- Enhanced Supply Chain Reliability: High purity is a critical metric for supply chain reliability, particularly in the pharmaceutical sector where impurity profiles must be tightly controlled. This method consistently delivers products with purity exceeding 99%, reducing the risk of batch rejection and ensuring a steady flow of qualified material to downstream customers. The robustness of the reaction across various solvents and substrates means that production is less susceptible to disruptions caused by specific reagent shortages. Manufacturers can source alternative solvents like dichloromethane, ethyl acetate, or nitrobenzene without compromising yield, providing flexibility in procurement strategies and mitigating supply risks.
- Scalability and Environmental Compliance: Scaling chemical processes often exacerbates waste management challenges, but this technology is inherently designed for scalability with minimal environmental impact. The "three wastes" (waste gas, waste water, waste residue) are basically eliminated compared to traditional methods, aligning with increasingly stringent global environmental regulations. The ability to recover the catalyst and the simplicity of the filtration-based isolation make this process ideal for large-scale commercial production ranging from pilot plants to multi-ton facilities. This environmental compliance ensures long-term operational continuity without the threat of regulatory shutdowns or costly remediation projects.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using Yb(OTf)3 over traditional Aluminum Chloride catalysts?
A: Yb(OTf)3 acts as a true catalyst requiring only 0.02 to 1.0 molar equivalents, whereas Aluminum Chloride often requires stoichiometric amounts. Additionally, Yb(OTf)3 can be recovered and reused with high efficiency, significantly reducing waste generation and post-processing complexity.
Q: What is the typical purity and yield achievable with this method?
A: According to patent data, this method consistently achieves product yields generally above 80% and purity levels exceeding 99%, minimizing the need for extensive downstream purification processes.
Q: Can the catalyst be recycled for industrial scale production?
A: Yes, the patent specifies a recovery method where the filtrate is concentrated and dehydrated under vacuum at 190°C, allowing the Yb(OTf)3 catalyst to be recovered with rates reaching up to 96%, making it highly suitable for continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-5-Amidobenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of the Yb(OTf)3 catalytic process are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-hydroxy-5-amidobenzophenone meets the highest quality standards required for API synthesis. Our commitment to technical excellence allows us to deliver consistent, high-performance intermediates that support your drug development timelines.
We invite you to collaborate with us to optimize your supply chain for these essential chemical building blocks. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your project's efficiency and profitability. Let us be your partner in driving innovation and reliability in fine chemical manufacturing.
