Mastering Controllable Carbazole Bromination for Commercial Scale-up of complex Electronic Chemicals
The landscape of organic functional materials is undergoing a significant transformation, driven by the relentless demand for high-performance electronic components. At the forefront of this evolution is the precise synthesis of carbazole derivatives, which serve as critical building blocks for OLED displays and organic photovoltaics. Patent CN103772269A introduces a groundbreaking controllable preparation method for carbazole bromo-compounds that addresses long-standing challenges in regioselectivity and process safety. This technology leverages N-bromosuccinimide (NBS) in a nitrogen atmosphere to achieve near-theoretical yields while eliminating the hazards associated with traditional liquid bromine handling. For industry leaders seeking a reliable electronic chemical supplier, this patent represents a pivotal shift towards greener, more efficient manufacturing paradigms that align with modern sustainability goals.
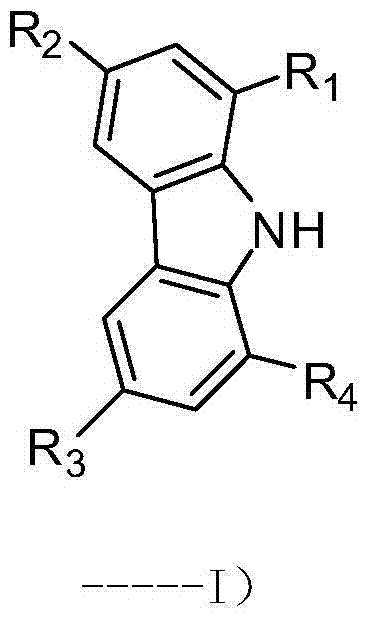
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the bromination of carbazole has been plagued by significant technical and safety hurdles that impede efficient commercial scale-up of complex electronic chemicals. Traditional protocols often rely on elemental liquid bromine, a highly volatile and corrosive reagent that poses severe risks to operator safety and reactor integrity. These methods typically require harsh conditions, such as high temperatures or the use of toxic Lewis acid catalysts like iron(III) chloride, which necessitate complex downstream purification steps to remove heavy metal residues. Furthermore, the reactivity of liquid bromine is difficult to control, frequently leading to a mixture of mono-, di-, and tri-brominated by-products that drastically reduce the overall yield of the desired tetra-substituted target. The environmental burden of disposing of excess bromine and acidic waste streams further complicates the regulatory compliance for manufacturers operating under strict environmental standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN103772269A offers a refined and controllable alternative that fundamentally reshapes the synthesis workflow. By utilizing N-bromosuccinimide (NBS) as the brominating agent in N,N-dimethylformamide (DMF), the process achieves exceptional selectivity through precise stoichiometric control rather than brute-force reactivity. The reaction proceeds under mild conditions, specifically at low temperatures ranging from 0°C to 5°C, which effectively suppresses side reactions and ensures the formation of the specific target isomer. This approach not only simplifies the workup procedure—allowing for simple water precipitation and filtration—but also eliminates the need for hazardous quenching steps required to neutralize excess elemental bromine. The result is a streamlined process that delivers high-purity intermediates suitable for sensitive optoelectronic applications without the baggage of toxic waste.
Mechanistic Insights into NBS-Mediated Electrophilic Substitution
The core of this technological advancement lies in the mechanistic precision of the electrophilic aromatic substitution facilitated by the NBS-DMF system. In this environment, the polar aprotic solvent DMF plays a crucial role in stabilizing the transition state and enhancing the electrophilicity of the bromine species generated from NBS. The reaction initiates with the formation of a bromonium ion equivalent, which attacks the electron-rich positions on the carbazole ring, specifically targeting the 3 and 6 positions first due to their higher electron density. By carefully modulating the molar ratio of NBS to carbazole, chemists can arrest the reaction at specific stages, allowing for the selective synthesis of 3-bromo, 3,6-dibromo, 1,3,6-tribromo, or 1,3,6,8-tetrabromo derivatives. This level of control is unattainable with elemental bromine, where the reaction kinetics are too rapid and exothermic to manage effectively without generating a statistical distribution of products.
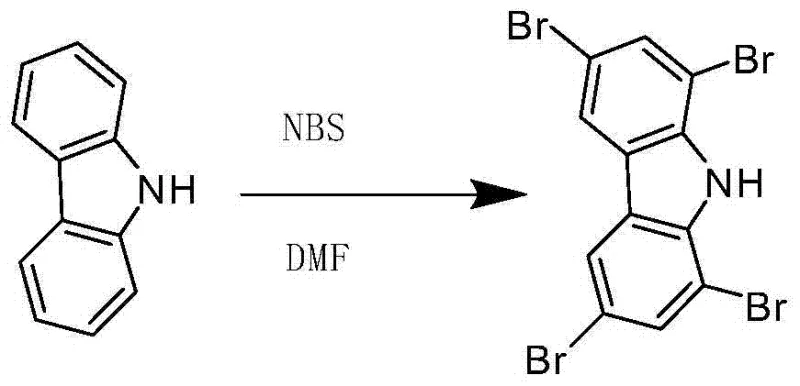
Furthermore, the impurity profile of the final product is significantly improved due to the mild nature of the reagents and the absence of metal catalysts. In traditional catalytic systems, trace metals can coordinate with the nitrogen atom of the carbazole or the pi-system, leading to difficult-to-remove complexes that degrade the performance of the final OLED material. The metal-free nature of this NBS-mediated process ensures that the impurity spectrum is limited primarily to unreacted starting material or succinimide by-products, both of which are easily removed via the described recrystallization steps using dichloromethane. This purity is paramount for R&D directors focusing on the longevity and efficiency of organic light-emitting diodes, where even parts-per-million levels of impurities can act as quenching sites that reduce device lifetime.
How to Synthesize 1,3,6,8-Tetrabromocarbazole Efficiently
Implementing this synthesis route requires strict adherence to the specified parameters to ensure reproducibility and safety on an industrial scale. The process begins with the preparation of two distinct solutions under a nitrogen atmosphere to prevent oxidation and moisture interference, which could degrade the NBS reagent. The precise control of temperature during the addition phase is critical, as is the subsequent stirring time at room temperature to ensure complete conversion. While the general principles are straightforward, the exact operational details regarding agitation speeds, addition rates, and crystallization parameters are vital for maximizing yield and minimizing batch-to-batch variability. For a comprehensive guide on the standardized operational procedures, please refer to the technical breakdown below.
- Dissolve carbazole in DMF to prepare Solution A and NBS in DMF for Solution B with a specific volume ratio of 20: 3.
- Control the reaction temperature at 5°C and slowly add Solution B to Solution A under nitrogen atmosphere.
- React at room temperature for 10 hours, then precipitate with water, filter, and recrystallize with dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, adopting this patented methodology offers substantial cost reduction in electronic chemical manufacturing by fundamentally altering the input cost structure. The elimination of elemental bromine removes the need for specialized corrosion-resistant reactors and extensive safety infrastructure, thereby lowering capital expenditure and maintenance costs associated with equipment degradation. Additionally, the high atom economy of the NBS reaction means that raw material utilization is optimized, reducing the volume of waste that requires expensive hazardous disposal. For supply chain heads, the reliability of NBS as a stable, solid reagent contrasts sharply with the logistical challenges of transporting and storing volatile liquid bromine, ensuring greater continuity of supply and reduced risk of production stoppages due to regulatory shipping constraints.
- Cost Reduction in Manufacturing: The transition to this metal-free process eliminates the expensive and time-consuming steps required to remove transition metal catalysts, which traditionally involve specialized scavengers or multiple washing cycles. By simplifying the purification workflow to a straightforward precipitation and recrystallization, manufacturers can significantly reduce solvent consumption and energy usage associated with distillation and drying. This streamlined approach translates directly into lower operating expenses per kilogram of produced intermediate, enhancing the overall margin profile for high-purity OLED material production without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing N-bromosuccinimide as the primary brominating agent mitigates the supply risks associated with hazardous liquid chemicals that are subject to strict transportation regulations and seasonal availability fluctuations. The stability of the solid reagent allows for bulk purchasing and long-term storage without the degradation issues common to liquid bromine, providing procurement managers with greater flexibility in inventory management. This stability ensures that production schedules can be maintained consistently, reducing lead time for high-purity electronic chemicals and preventing delays that could impact downstream device manufacturing timelines.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup procedure make this process inherently safer and easier to scale from pilot plant to full commercial production volumes. The absence of toxic heavy metal waste streams simplifies environmental compliance reporting and reduces the liability associated with hazardous waste disposal, aligning with global initiatives for greener chemistry. This environmental advantage not only future-proofs the manufacturing site against tightening regulations but also enhances the brand value of the final electronic products by ensuring they are produced via sustainable and responsible chemical pathways.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbazole bromination technology. These insights are derived directly from the patent specifications and are designed to clarify the operational benefits and chemical capabilities of the method for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production lines or new product development pipelines.
Q: How does this method improve upon traditional liquid bromine bromination?
A: Unlike traditional methods using volatile and corrosive liquid bromine which yield mixed by-products, this patent utilizes N-bromosuccinimide (NBS) for precise stoichiometric control, resulting in higher purity and easier separation without toxic heavy metal catalysts.
Q: What is the regioselectivity capability of this synthesis route?
A: By adjusting the molar ratio of NBS to carbazole and strictly controlling the reaction temperature between 0°C and 5°C, the process can selectively produce mono-, di-, tri-, or tetra-brominated carbazole derivatives with high specificity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method employs mild reaction conditions, common solvents like DMF, and a simple water precipitation workup, making it highly scalable and environmentally friendly compared to high-temperature pyroreaction methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,6,8-Tetrabromocarbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of precise chemical synthesis in the development of next-generation optoelectronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent to industrial reality is seamless and efficient. We are committed to delivering stringent purity specifications and maintaining rigorous QC labs to verify that every batch of carbazole derivative meets the exacting standards required for high-performance OLED and organic semiconductor applications. Our infrastructure is designed to handle complex chemistries safely, providing a secure foundation for your supply chain.
We invite you to collaborate with us to optimize your material sourcing strategy and leverage these advanced synthetic routes for your product portfolio. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate how our capabilities can support your long-term growth and innovation goals in the electronic materials sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
