Advanced Metal-Free Synthesis of 2-Fluoroindole Compounds for Commercial Pharma Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing fluorinated heterocyclic scaffolds, which are pivotal in modern drug design. Patent CN112724065B introduces a groundbreaking synthetic strategy for 2-fluoroindole compounds that addresses critical bottlenecks in current manufacturing processes. This innovation utilizes o-aminochalcone derivatives as key starting materials, reacting them with ethyl bromodifluoroacetate in the presence of a base and organic solvent under a nitrogen atmosphere. The significance of this patent lies in its ability to bypass the need for pre-synthesized indole substrates and expensive transition metal catalysts, thereby offering a streamlined, one-pot approach that is highly attractive for the commercial production of high-purity pharmaceutical intermediates. By operating under mild thermal conditions ranging from 50-100°C, this method not only enhances safety profiles but also significantly reduces energy consumption, positioning it as a superior alternative for sustainable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-fluoroindole compounds has been fraught with significant technical and economic challenges that hinder efficient commercial scale-up. Traditional routes often rely heavily on the use of precious transition metal catalysts such as palladium, copper, silver, or ruthenium to activate C-H bonds or facilitate cross-coupling reactions. These metal-dependent processes are not only costly due to the high price of the catalysts but also introduce severe complications regarding product purity, as residual heavy metals must be rigorously removed to meet stringent pharmaceutical regulatory standards. Furthermore, conventional strategies frequently require the prior synthesis of the indole core followed by a separate, often harsh, fluorination step, which leads to poor atom economy, multiple purification stages, and lower overall yields. The harsh reaction conditions associated with direct fluorination, such as the use of aggressive fluorinating agents or extreme temperatures, further exacerbate safety risks and limit the compatibility with sensitive functional groups, ultimately restricting the structural diversity accessible to medicinal chemists.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112724065B presents a transformative, metal-free pathway that fundamentally simplifies the synthetic landscape for 2-fluoroindoles. This novel approach leverages the intrinsic reactivity of o-aminochalcone compounds, allowing for simultaneous ring closure and fluorine incorporation in a single operational step. By employing ethyl bromodifluoroacetate as both the carbon and fluorine source in the presence of a mild inorganic base like potassium phosphate, the reaction proceeds efficiently without the need for exotic ligands or toxic metals. This methodology dramatically reduces the complexity of the supply chain by utilizing readily available starting materials and common solvents such as acetonitrile. The result is a cleaner reaction profile with fewer by-products, facilitating easier downstream processing and significantly lowering the cost of goods sold (COGS) for manufacturers. This shift from multi-step, metal-catalyzed sequences to a direct, base-mediated cyclization represents a paradigm shift in how these valuable heterocycles can be produced at scale.
Mechanistic Insights into Base-Mediated Cyclization and Fluorination
The mechanistic elegance of this synthesis lies in the synergistic interaction between the base, the difluoroacetate source, and the o-aminochalcone substrate under inert atmosphere conditions. The base, preferably potassium phosphate, acts as a crucial promoter that deprotonates the amine functionality or activates the difluoroacetate species, initiating a nucleophilic attack that triggers the cascade leading to indole ring formation. Unlike transition metal catalysis which relies on oxidative addition and reductive elimination cycles, this metal-free mechanism avoids the generation of metal-complexed intermediates that are difficult to separate. The introduction of the fluorine atom occurs concurrently with the cyclization event, ensuring that the fluorine is positioned precisely at the 2-position of the indole ring with high regioselectivity. This concerted process minimizes the formation of isomeric impurities that are common in stepwise fluorination protocols, thereby enhancing the overall purity of the crude reaction mixture. The use of a nitrogen atmosphere further protects the reactive intermediates from oxidation, ensuring consistent reproducibility and high yields across different batches, which is critical for maintaining quality control in GMP environments.
From an impurity control perspective, the absence of transition metals eliminates an entire class of potential contaminants that typically require specialized scavenging resins or complex extraction protocols to remove. The primary by-products in this reaction are generally inorganic salts and benign organic fragments that can be easily separated through simple aqueous workups or filtration. The patent data indicates that the reaction tolerates a wide range of substituents on the chalcone backbone, including electron-withdrawing and electron-donating groups, without significant degradation in performance. This robustness suggests that the mechanism is not overly sensitive to steric or electronic variations, making it a versatile platform for generating diverse libraries of fluorinated indoles. The ability to achieve high conversion rates within 10-15 hours at moderate temperatures further underscores the kinetic efficiency of this pathway, allowing manufacturers to maximize reactor throughput and minimize cycle times.
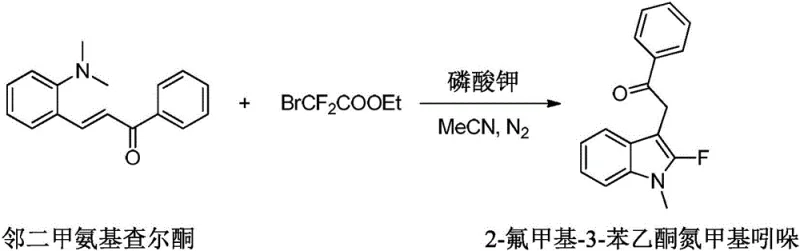
How to Synthesize 2-Fluoroindole Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure optimal yield and safety. The process begins with the precise weighing of o-aminochalcone substrates and ethyl bromodifluoroacetate, which are then combined with a stoichiometric amount of base such as potassium phosphate in a pressure-resistant sealed reaction vessel. Acetonitrile is introduced as the solvent of choice due to its excellent solubility profile and stability under the reaction conditions. The system is purged with nitrogen to create an inert environment before being heated to a controlled temperature range of 50-100°C. Monitoring the reaction progress via TLC or GC allows operators to determine the exact endpoint, typically around 12 hours, ensuring complete conversion without over-reaction. Upon completion, the mixture is cooled, and the product is isolated through a straightforward filtration and washing sequence with ethyl acetate, followed by solvent removal and purification via silica gel column chromatography using a petroleum ether and dichloromethane eluent system.
- Combine o-aminochalcone, ethyl bromodifluoroacetate, base (e.g., potassium phosphate), and organic solvent (e.g., acetonitrile) in a sealed vessel under nitrogen.
- Stir the reaction mixture at 50-100°C for 10-15 hours to facilitate cyclization and fluorination.
- Cool to room temperature, filter, wash with ethyl acetate, remove solvent, and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis technology offers profound strategic advantages that extend far beyond simple chemical transformation. The most immediate impact is seen in the drastic reduction of raw material costs, as the elimination of precious metal catalysts removes a significant line item from the bill of materials. Furthermore, the reliance on commodity chemicals like acetonitrile and potassium phosphate ensures a stable and resilient supply chain that is less susceptible to the geopolitical volatility often associated with rare earth metals or specialized ligands. The simplified workup procedure, which avoids complex metal scavenging steps, translates directly into reduced processing time and lower utility consumption, thereby enhancing the overall operational efficiency of the manufacturing facility. These factors collectively contribute to a more competitive cost structure, enabling companies to offer high-quality intermediates at more attractive price points while maintaining healthy margins.
- Cost Reduction in Manufacturing: The exclusion of expensive transition metals such as palladium and ruthenium fundamentally alters the cost dynamics of producing 2-fluoroindole derivatives. By replacing these high-value catalysts with inexpensive inorganic bases and readily available organic reagents, manufacturers can achieve substantial cost savings on every kilogram produced. Additionally, the simplified purification process reduces the consumption of specialized resins and solvents required for metal removal, further driving down operational expenditures. This economic efficiency allows for greater flexibility in pricing strategies and improves the viability of projects that may have previously been marginally profitable due to high synthesis costs.
- Enhanced Supply Chain Reliability: Sourcing high-purity transition metal catalysts can often be a bottleneck, subject to long lead times and supply disruptions. In contrast, the reagents required for this novel method, including o-aminochalcones and ethyl bromodifluoroacetate, are widely available from multiple global suppliers, ensuring a continuous and reliable flow of materials. The robustness of the reaction conditions also means that production schedules are less likely to be impacted by minor variations in reagent quality or environmental factors. This reliability is crucial for meeting tight delivery deadlines and maintaining trust with downstream pharmaceutical clients who depend on just-in-time inventory models for their own production lines.
- Scalability and Environmental Compliance: The green chemistry credentials of this process are a major asset for companies aiming to reduce their environmental footprint. The absence of toxic heavy metals simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The mild reaction temperatures and use of standard solvents make the process inherently safer and easier to scale from gram-scale laboratory experiments to multi-ton commercial production without requiring specialized high-pressure or high-temperature equipment. This scalability ensures that supply can be rapidly ramped up to meet market demand without the need for significant capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of this route for their specific target molecules and for procurement teams assessing the long-term viability of the supply chain.
Q: What are the advantages of this metal-free synthesis over traditional methods?
A: Unlike traditional methods requiring expensive transition metals like palladium or ruthenium, this process is metal-free, significantly reducing raw material costs and eliminating the need for complex heavy metal removal steps, which streamlines purification and ensures higher product purity.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction operates under mild conditions, typically between 50-100°C in a nitrogen atmosphere using common organic solvents like acetonitrile, making it safer and more energy-efficient compared to harsh fluorination protocols.
Q: Is this method suitable for large-scale production?
A: Yes, the use of readily available o-aminochalcone substrates and simple workup procedures involving filtration and washing makes this protocol highly scalable for industrial manufacturing without compromising yield or safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoroindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methods disclosed in CN112724065B and are fully equipped to leverage this technology for our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are designed to handle complex organic syntheses with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 2-fluoroindole intermediate meets the highest industry standards. We understand that consistency and quality are non-negotiable in the pharmaceutical sector, and our dedicated technical team is committed to delivering products that exceed expectations.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your specific project requirements. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume needs, along with specific COA data and route feasibility assessments. Whether you are developing a new API or optimizing an existing supply chain, our expertise in fluorinated heterocycles positions us as the ideal partner to drive your success forward. Contact us today to discuss how we can support your journey from molecule to medicine with efficiency and precision.
