Scaling High-Yield 3,6-Diaminopyrazine-2,5-Dicarboxylic Acid Production via Microreactor Technology
The pharmaceutical and fine chemical industries are constantly seeking robust pathways for synthesizing complex heterocyclic intermediates, particularly those serving as precursors for advanced diagnostic agents. Patent CN116438164A introduces a groundbreaking methodology for the preparation of 3,6-diaminopyrazine-2,5-dicarboxylic acid (MB-301) and its key synthetic intermediates. This compound is critical for producing pyrazine derivatives like MB-102, which function as essential fluorescent tracers in medical imaging. The disclosed technology addresses long-standing inefficiencies in the field, specifically targeting the oxidative cyclization step that has historically plagued manufacturers with low yields and safety hazards. By leveraging precise flow control and alternative alkaline conditions, this innovation transforms a previously bottlenecked reaction into a high-efficiency process suitable for modern industrial demands.
For research and development teams, the significance of this patent lies in its ability to deliver high-purity intermediates with significantly reduced impurity profiles. The shift from traditional batch limitations to controlled simultaneous addition mechanisms ensures that transient intermediates are stabilized before degradation can occur. This level of process control is vital for maintaining the stringent purity specifications required in pharmaceutical applications. Furthermore, the substitution of hazardous reagents with safer inorganic alternatives aligns with global trends towards greener chemistry, making this route not only technically superior but also environmentally compliant for forward-thinking organizations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
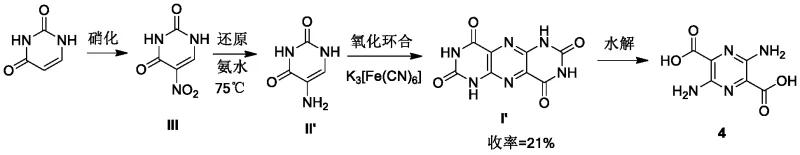
Historically, the synthesis of 3,6-diaminopyrazine-2,5-dicarboxylic acid has been hindered by severe operational drawbacks that limit its commercial viability. Early synthetic routes relied on highly toxic raw materials such as hydrogen cyanide and diphenyl disulfide, posing unacceptable safety risks for large-scale operations. Subsequent improvements, such as those described in DE 10 2016 205 615 A1, attempted to mitigate toxicity by using uracil derivatives but introduced new challenges. Specifically, the oxidative cyclization step in these conventional methods suffered from extremely low yields, often reported around 21%, due to the rapid degradation of transient intermediates into urea byproducts. Additionally, the reliance on ammonia water as a base at elevated temperatures created significant safety hazards due to volatilization and potential exposure risks.
The inefficiency of these legacy processes creates a substantial burden on supply chains, as low yields necessitate larger volumes of raw materials and generate excessive waste. For procurement managers, this translates to higher unit costs and unpredictable availability of critical intermediates. The inability to effectively control the reaction environment in standard batch reactors means that side reactions dominate, leading to complex purification requirements that further erode profit margins. Consequently, there has been a persistent demand for a method that can overcome these thermodynamic and kinetic barriers without compromising on safety or environmental standards.
The Novel Approach
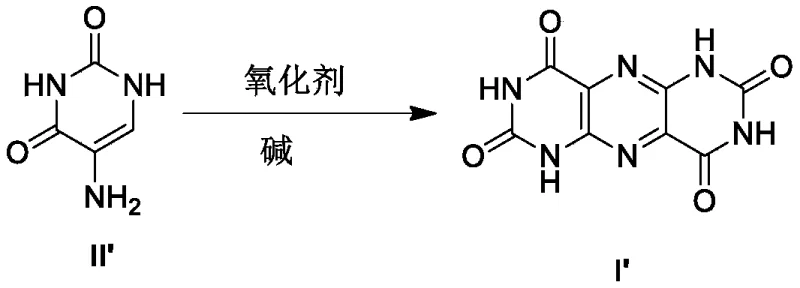
The innovative strategy outlined in CN116438164A fundamentally reengineers the oxidative cyclization step through the implementation of microreactor technology and optimized reagent addition protocols. Instead of sequential or uncontrolled mixing, the new method involves the simultaneous introduction of 5-aminouracil and metal oxidant solutions into a reactor at constant, controlled flow rates. This precise mixing regime ensures that the concentration of reactive species remains within an optimal window, effectively suppressing the degradation pathways that lead to urea formation. As a result, the yield of the pivotal pteridine-tetraketone intermediate surges from roughly 21% to approximately 90% under optimized conditions, representing a paradigm shift in process efficiency.
Beyond yield enhancement, this approach significantly improves the safety profile of the synthesis. By replacing volatile ammonia with stable inorganic bases such as potassium hydroxide or sodium carbonate during the reduction phase, the process eliminates the risk of noxious gas release. The use of aqueous systems and moderate temperatures further simplifies the engineering requirements for containment and ventilation. For supply chain leaders, this reliability means fewer interruptions due to safety incidents and a more consistent output of high-quality material. The adaptability of the method to both microreactor continuous flow and optimized stirred tank reactors provides flexibility for scaling from pilot studies to full commercial production without losing the benefits of the improved kinetics.
Mechanistic Insights into Oxidative Cyclization and Impurity Control
The core chemical advancement in this patent revolves around the stabilization of the oxidative cyclization mechanism using iron(III) based oxidants like potassium ferricyanide. In traditional setups, the slow addition of oxidants allows the intermediate species to linger in the reaction mixture, where they are susceptible to hydrolysis and rearrangement into urea. The patented process counters this by maintaining a high local concentration of oxidant relative to the substrate through simultaneous pumping. This kinetic control forces the reaction pathway towards the desired ring closure, forming the pyrimido[4,5-g]pteridine structure before degradation can compete. The reaction is typically conducted at low temperatures, ranging from -6°C to 25°C, which further stabilizes the transition states and minimizes thermal decomposition.
Impurity control is another critical aspect where this mechanism excels. The strict regulation of molar ratios, specifically maintaining the oxidant to substrate ratio between 3.0:1.0 and 3.2:1.0, ensures complete conversion while minimizing excess reagent carryover. The use of specific proton acceptors like KOH or Na2CO3 helps maintain the pH in a range that favors the deprotonation of the uracil ring, facilitating the nucleophilic attack required for cyclization. By avoiding ammonia, the process also prevents the formation of ammonium salts that can complicate downstream isolation. This clean reaction profile results in crude products with purity exceeding 98%, drastically reducing the load on purification units and ensuring that the final API intermediate meets rigorous regulatory standards for heavy metals and organic impurities.
How to Synthesize 3,6-Diaminopyrazine-2,5-Dicarboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to the preparation of precursor solutions and the calibration of flow equipment. The process begins with the reduction of 5-nitouracil to 5-aminouracil using sodium dithionite in the presence of an inorganic base, followed by the critical oxidative cyclization step. Operators must ensure that the feed solutions are cooled to between 0°C and 4°C before entering the mixing module to prevent premature reaction. The residence time in the microreactor is short, typically around 20 to 30 seconds, highlighting the speed and efficiency of the transformation. Detailed standardized operating procedures regarding pump calibration and temperature monitoring are essential to replicate the high yields observed in the patent examples consistently.
- Reduce 5-nitouracil using sodium dithionite and an inorganic base like sodium bicarbonate at 75°C to obtain 5-aminouracil with yields up to 92%.
- Perform oxidative cyclization by simultaneously pumping 5-aminouracil and potassium ferricyanide solutions into a microreactor at 0°C to -5°C, achieving yields around 84-90%.
- Hydrolyze the resulting pteridine-tetraketone intermediate with sodium hydroxide at 170-180°C to finalize the 3,6-diaminopyrazine-2,5-dicarboxylic acid structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain strategists, the adoption of this patented methodology offers tangible economic and operational benefits that extend beyond simple yield metrics. The drastic improvement in reaction efficiency directly correlates to a reduction in the cost of goods sold, as less raw material is wasted on byproducts. The elimination of hazardous reagents like hydrogen cyanide and volatile ammonia simplifies regulatory compliance and reduces the overhead costs associated with specialized safety infrastructure and waste disposal. These factors combine to create a more resilient supply chain capable of meeting high-volume demands without the bottlenecks typical of older, less efficient chemistries.
- Cost Reduction in Manufacturing: The increase in yield from approximately 21% to over 84% fundamentally alters the economics of production. By maximizing the conversion of starting materials into the desired pteridine intermediate, manufacturers can significantly lower the effective cost per kilogram of the final product. Furthermore, the use of common inorganic bases and aqueous solvents reduces the reliance on expensive organic solvents or specialized catalysts that require complex removal steps. This simplification of the bill of materials allows for better margin protection and more competitive pricing strategies in the global market for fluorescent tracer precursors.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply continuity. By removing toxic and volatile substances from the process, the risk of production shutdowns due to safety incidents or regulatory inspections is markedly decreased. The robustness of the inorganic base system ensures that raw material sourcing is more stable, as these chemicals are commodity items with reliable global availability. This stability allows supply chain managers to forecast production schedules with greater confidence, ensuring that downstream customers receive their orders on time without unexpected delays caused by raw material shortages or processing hazards.
- Scalability and Environmental Compliance: The design of this process supports seamless scale-up from laboratory to industrial scales. Whether utilizing microreactor arrays for continuous manufacturing or adapted batch vessels with controlled addition, the chemistry remains consistent. This flexibility allows companies to respond quickly to market demand fluctuations. Additionally, the reduction in urea byproducts and the avoidance of toxic emissions align with increasingly strict environmental regulations. This proactive approach to green chemistry not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B partnerships.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is essential for stakeholders evaluating its integration into existing production lines. The following questions address common concerns regarding yield consistency, safety protocols, and scalability. The answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring that the information reflects the actual capabilities of the technology. This transparency helps build trust between technology providers and manufacturing partners.
Q: How does the new process improve yield compared to prior art?
A: The patented method utilizes simultaneous addition of reactants in a microreactor system, which suppresses the formation of urea byproducts. This increases the oxidative cyclization yield from approximately 21% in conventional methods to over 84-90%.
Q: What safety advantages does this synthesis route offer?
A: The process eliminates the use of volatile ammonia water in the reduction step by substituting it with stable inorganic bases like sodium bicarbonate. It also avoids highly toxic hydrogen cyanide used in older synthetic routes.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It supports both microreactor continuous flow modes and optimized batch processes with controlled addition rates, ensuring consistent quality and high throughput for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,6-Diaminopyrazine-2,5-Dicarboxylic Acid Supplier
As the demand for high-performance fluorescent tracers continues to grow, securing a reliable supply of high-purity 3,6-diaminopyrazine-2,5-dicarboxylic acid is paramount for pharmaceutical and diagnostic companies. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced process technologies like the one described in CN116438164A to deliver superior intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the necessary criteria for downstream synthesis.
We invite you to collaborate with us to optimize your supply chain for these critical materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this high-yield, safer synthesis method. Contact us today to discuss how we can support your R&D and commercial manufacturing goals with reliable, cost-effective solutions.
