Advanced Synthesis of Chiral Camptothecin Intermediates for Commercial Anticancer Drug Production
Advanced Synthesis of Chiral Camptothecin Intermediates for Commercial Anticancer Drug Production
The pharmaceutical industry continuously seeks robust and scalable pathways for producing complex chiral intermediates essential for next-generation oncology therapeutics. Patent CN101121719A introduces a groundbreaking methodology for the preparation of (S)-4-ethyl-6,6-disubstituted alkoxy(thio)-7,8-dihydro-4-hydroxy-1H-pyrano[3,4-f]indolizine-3,10(4H)-dione compounds, which serve as critical precursors for 20(S)-camptothecin derivatives. This technology addresses the longstanding challenges associated with synthesizing the E-ring lactone moiety found in potent topoisomerase I inhibitors such as Topotecan and Irinotecan. By leveraging a concise three-step sequence involving catalytic hydrogenation, reductive cyclization, and asymmetric hydroxylation, this process offers a compelling alternative to traditional biocatalytic or multi-step chemical routes. For R&D directors and procurement specialists, understanding the nuances of this patent is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with consistent stereochemical integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of camptothecin analogues has been plagued by inefficient routes that struggle to balance cost, yield, and stereoselectivity. One prominent conventional strategy, exemplified by the Imura method, relies on the derivatization of a racemic precursor followed by enzymatic selective hydrolysis to isolate the desired enantiomer. As illustrated in the reaction scheme below, this approach is inherently flawed due to its reliance on expensive enzymes and a protracted reaction sequence that drastically reduces overall throughput. The necessity for biological catalysts introduces variability in batch-to-batch consistency and imposes strict storage and handling conditions that complicate supply chain logistics for large-scale manufacturing facilities.
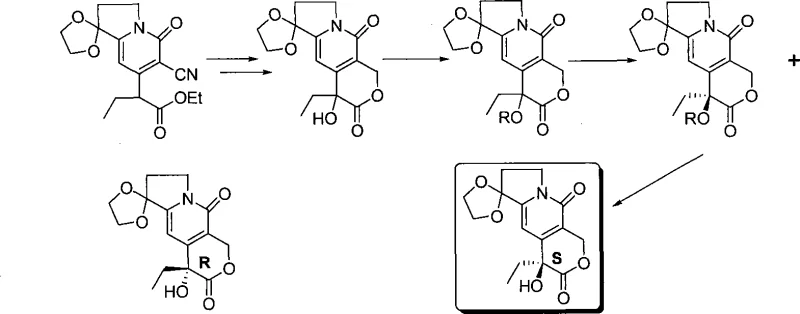
Furthermore, alternative chemical approaches, such as the method developed by Jew et al., attempt to bypass enzymatic steps but introduce their own set of significant drawbacks. This pathway typically involves a four-step sequence including reduction, dehydration, asymmetric dihydroxylation, and oxidation. The critical bottleneck in this route is the asymmetric dihydroxylation step, which requires costly chiral ligands that are difficult to recover and recycle. The cumulative effect of these multiple transformation steps results in substantial material loss and increased waste generation, making it economically unviable for the cost reduction in pharmaceutical intermediate manufacturing required by modern generic drug producers.
The Novel Approach
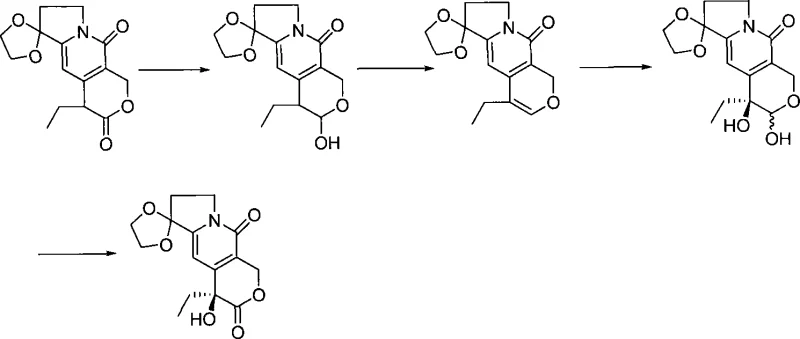
In stark contrast to these legacy methods, the novel approach detailed in CN101121719A streamlines the synthesis into a highly efficient three-step protocol that eliminates the need for expensive biocatalysts or complex chiral ligand systems. The process initiates with a straightforward catalytic hydrogenation of a cyano-ester precursor to generate a key formyl intermediate, utilizing readily available Raney nickel. This is followed by a clever one-pot reduction and ring-closing reaction that constructs the core pyrano-indolizine skeleton with high fidelity. The final step employs a chiral oxaziridine oxidant to install the crucial hydroxyl group with excellent stereocontrol. This strategic simplification not only shortens the production timeline but also significantly lowers the barrier to entry for commercial scale-up of complex pharmaceutical intermediates, ensuring a more stable and predictable supply for downstream API production.
Mechanistic Insights into Asymmetric Hydroxylation and Cyclization
The core innovation of this technology lies in the precise orchestration of the reductive cyclization and the subsequent asymmetric hydroxylation. In the second step, the aldehyde intermediate undergoes reduction using mild hydride sources like sodium borohydride or potassium borohydride in alcoholic solvents at controlled low temperatures ranging from -40°C to 30°C. This conditions the molecule for an immediate intramolecular attack, closing the lactone ring to form the tricyclic ketone structure. The choice of solvent, preferably methanol or ethanol, and the careful control of acidity during the workup are critical to preventing the hydrolysis of the sensitive lactone moiety, thereby preserving the structural integrity of the intermediate before the final stereochemical installation.
The final asymmetric hydroxylation step is the determinant of optical purity, utilizing a chiral oxidant such as (+)-camphorsulfonyloxaziridine in the presence of a strong non-nucleophilic base like potassium hexamethyldisilazide (KHMDS). The reaction is conducted at cryogenic temperatures, typically between -78°C and -40°C, to maximize the kinetic resolution and minimize racemization. The base deprotonates the alpha-position of the ketone, generating an enolate that attacks the chiral oxidant from the less hindered face, effectively transferring the oxygen atom with high facial selectivity. This mechanism ensures that the resulting (S)-enantiomer is produced with an enantiomeric excess (ee) consistently above 84%, providing a robust foundation for the synthesis of high-purity OLED material precursors or active pharmaceutical ingredients where chirality dictates biological activity.
How to Synthesize (S)-4-ethyl-6,6-disubstituted Alkoxy Compounds Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters to ensure optimal yield and stereochemical outcome. The process begins with the hydrogenation of the cyano starting material in a ternary solvent system of water, pyridine, and acetic acid, followed by the reductive cyclization in methanol. The final asymmetric oxidation demands rigorous temperature control and anhydrous conditions to maintain the activity of the strong base and chiral oxidant. For process chemists looking to replicate this success, the detailed standardized synthesis steps outlined below provide a clear roadmap for translating this laboratory-scale innovation into a GMP-compliant manufacturing process.
- Perform catalytic hydrogenation of the cyano-ester precursor using Raney nickel in a water-pyridine-acetic acid solvent system to generate the formyl intermediate.
- Execute a one-pot reduction and ring-closing reaction using sodium borohydride or potassium borohydride in methanol at low temperatures (-20°C to -30°C).
- Conduct asymmetric hydroxylation using a chiral oxaziridine oxidant and a strong organic base like KHMDS at cryogenic temperatures (-78°C) to establish the (S)-configuration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route represents a strategic opportunity to optimize the sourcing of critical oncology intermediates. By shifting away from enzyme-dependent or ligand-heavy processes, manufacturers can mitigate the risks associated with the volatility of biocatalyst pricing and the scarcity of specialized chiral reagents. This transition facilitates a more resilient supply chain, as the reagents required—such as Raney nickel, borohydrides, and standard organic solvents—are commodity chemicals with established global availability. Consequently, this leads to substantial cost savings in the overall production budget, allowing for more competitive pricing strategies in the final API market without compromising on quality or regulatory compliance standards.
- Cost Reduction in Manufacturing: The elimination of expensive enzymes and chiral ligands directly translates to a lower bill of materials. By utilizing robust chemical catalysts like Raney nickel and achieving high yields in the cyclization step (up to 92%), the process minimizes raw material waste. This efficiency drives down the cost per kilogram of the intermediate, enabling significant margin improvements for generic drug manufacturers who operate on thin margins and require aggressive cost reduction in pharmaceutical intermediate manufacturing to remain competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Dependence on proprietary biological agents often creates single points of failure in the supply chain. This chemical synthesis route relies on widely available inorganic and organic reagents, reducing the risk of supply disruptions. The simplified three-step sequence also shortens the lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond more rapidly to fluctuations in market demand. This reliability is crucial for maintaining continuous API production schedules and avoiding costly downtime in downstream formulation facilities.
- Scalability and Environmental Compliance: The streamlined nature of this process inherently reduces the environmental footprint by minimizing the number of unit operations and solvent exchanges. Fewer steps mean less energy consumption and reduced generation of hazardous waste streams, aligning with increasingly stringent environmental regulations. Furthermore, the use of standard chemical reagents simplifies the scale-up process from pilot plant to commercial tonnage, ensuring that the technology can be seamlessly transferred to large-scale reactors without the engineering complexities often associated with biocatalytic processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational benefits and chemical feasibility of the method. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or long-term supply partnerships.
Q: What are the primary advantages of this synthesis method over enzymatic routes?
A: Unlike enzymatic methods which suffer from lengthy routes and expensive biocatalysts, this chemical approach utilizes standard reagents like Raney nickel and chiral oxaziridines, significantly reducing raw material costs and simplifying the operational workflow for large-scale manufacturing.
Q: What level of stereoselectivity can be achieved with this protocol?
A: The patent data indicates that the asymmetric hydroxylation step yields the desired (S)-enantiomer with an enantiomeric excess (ee) of approximately 84% to 90%, which is sufficient for downstream processing into high-purity antitumor agents like Topotecan and Irinotecan.
Q: Which catalysts are preferred for the initial hydrogenation step?
A: While both Palladium-Carbon and Raney Nickel are viable, the patent specifically highlights Raney Nickel as the preferred catalyst due to its superior activity in converting the cyano group to the formyl group within the specific water-pyridine-acetic acid solvent mixture.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-ethyl-6,6-disubstituted Alkoxy Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving anticancer therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN101121719A can be executed with precision at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (S)-4-ethyl-6,6-disubstituted alkoxy compound meets the exacting standards required for clinical and commercial applications, providing our partners with the confidence needed to advance their drug development pipelines.
We invite global pharmaceutical companies and contract research organizations to collaborate with us on optimizing this synthesis route for their specific needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current supply chain constraints. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can enhance your production efficiency and secure your supply of this vital pharmaceutical building block.
