Revolutionizing Amino Acid Intermediates: High-Efficiency Synthesis of N-Acetyl-D,L-Methionine for Global Supply Chains
Revolutionizing Amino Acid Intermediates: High-Efficiency Synthesis of N-Acetyl-D,L-Methionine for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to enhance the efficiency of amino acid derivative production, particularly for critical intermediates like N-acetyl-D,L-methionine. A pivotal advancement in this domain is detailed in Chinese Patent CN103408474A, which outlines a highly efficient production method capable of achieving exceptional purity and yield metrics. This technology represents a significant departure from legacy crystallization techniques, addressing long-standing challenges regarding environmental impact and raw material utilization. By integrating a novel organic solvent treatment step following acylation, the process effectively isolates the target compound from inorganic salt byproducts without generating the voluminous acidic wastewater typical of older protocols. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for evaluating potential supply chain partners who can deliver high-purity pharmaceutical intermediates with consistent reliability. The methodology not only optimizes the reaction kinetics but also introduces a sustainable separation mechanism that aligns with modern green chemistry principles, making it a cornerstone technology for scalable manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
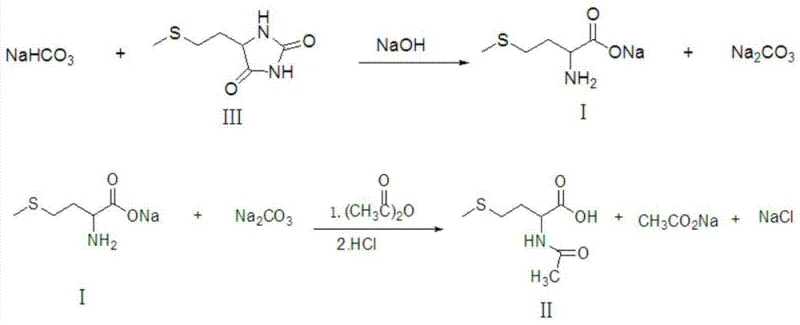
Historically, the synthesis of N-acetyl-D,L-methionine has been plagued by inefficiencies inherent in traditional acidification and cooling crystallization workflows. Conventional processes, such as those described in earlier patents like US6114163, typically involve neutralizing D,L-methionine with sodium hydroxide followed by reaction with acetic anhydride, and finally acidifying the mixture to induce crystallization. However, these methods suffer from critically low crystallization yields, often leaving a significant portion of the valuable product dissolved in the acidic mother liquor. Furthermore, the generation of large quantities of acidic wastewater containing complex organic residues poses a severe environmental burden, necessitating costly treatment procedures or leading to regulatory non-compliance risks. The inability to effectively recover the product from the mother liquor not only inflates production costs through raw material wastage but also complicates the purification landscape, often requiring additional ion-exchange resin steps that further erode profit margins. Consequently, manufacturers relying on these outdated techniques face substantial hurdles in maintaining competitive pricing while adhering to stringent environmental standards.
The Novel Approach
In stark contrast, the innovative methodology disclosed in patent CN103408474A introduces a paradigm shift by replacing direct acid crystallization with a sophisticated organic solvent precipitation strategy. This novel approach begins with the acylation of sodium D,L-methionine, followed by a controlled acidification to pH 2-4, but crucially diverges by employing vacuum evaporation to concentrate the mixture to a water content of less than 5%. The addition of specific organic solvents—ranging from alcohols like ethanol to esters like ethyl acetate—induces the precipitation of inorganic salts such as sodium chloride and sodium acetate, leaving the N-acetyl-D,L-methionine in the organic phase. This separation mechanism allows for the physical removal of salt byproducts via filtration or centrifugation, thereby preventing them from contaminating the final crystal lattice. The result is a process that boasts yields as high as 98% and purity levels reaching 99%, effectively solving the dual problems of low recovery rates and environmental pollution. By enabling the recycling of organic solvents and eliminating salty wastewater discharge, this technique offers a compelling value proposition for cost reduction in amino acid derivative manufacturing.
Mechanistic Insights into Acylation and Solvent-Based Separation
The core chemical transformation in this process relies on the Schotten-Baumann type acylation of the amino group on the methionine sodium salt. When D,L-methionine sodium reacts with acetic anhydride at temperatures between 25°C and 45°C, the nucleophilic nitrogen attacks the carbonyl carbon of the anhydride, forming the N-acetyl bond and releasing acetate ions. The stoichiometry is carefully controlled with a molar ratio of methionine sodium to acetic anhydride ranging from 1:1.0 to 1:1.5 to ensure complete conversion while minimizing excess reagent waste. Following this, the introduction of inorganic acids like sulfuric or hydrochloric acid protonates the carboxylate group, converting the soluble sodium salt into the free acid form of N-acetyl-D,L-methionine. However, the true mechanistic brilliance lies in the subsequent solubility differential exploitation; by reducing the water content to ≤5% via vacuum distillation at temperatures not exceeding 65°C, the polarity of the medium is drastically altered. This creates an environment where inorganic salts become insoluble in the added organic solvent, while the organic target molecule remains soluble, facilitating a clean phase separation that is thermodynamically driven and kinetically favorable.
Furthermore, the impurity control mechanism is intrinsically linked to this solvent-based purification step. In traditional aqueous crystallizations, inorganic salts often co-crystallize or form solvates with the product, necessitating multiple recrystallization cycles that degrade overall yield. In this patented process, the inorganic byproducts (NaCl and Sodium Acetate) are physically filtered off as solids before the final crystallization of the product occurs. This pre-emptive removal ensures that the final evaporation of the organic solvent yields crystals of exceptional purity, often exceeding 99% without the need for extensive downstream polishing. Additionally, the process incorporates a decolorization step using activated carbon on the organic solution, which effectively adsorbs colored polymeric impurities or trace organic side-products formed during the acylation. This comprehensive approach to impurity management ensures that the final API intermediate meets the rigorous specifications required for downstream enzymatic resolution into L-methionine, thereby safeguarding the quality of the entire synthetic sequence.
How to Synthesize N-Acetyl-D,L-Methionine Efficiently
Implementing this high-efficiency synthesis route requires precise control over reaction parameters and separation conditions to maximize the economic and technical benefits outlined in the patent literature. The process is designed to be adaptable, allowing for the use of commercially available sodium methionine or in-situ generated sodium methionine from hydantoin hydrolysis, providing flexibility in raw material sourcing. Operators must strictly adhere to the temperature constraints during the vacuum concentration phase to prevent thermal degradation of the heat-sensitive acetyl group, while ensuring that the water content is sufficiently reduced to trigger the desired solubility shifts upon solvent addition. The selection of the organic solvent is also critical, with ethanol and ethyl acetate demonstrating superior performance in balancing solubility profiles and ease of recovery. For a detailed breakdown of the operational parameters, including specific flow rates, mixing speeds, and filtration protocols, please refer to the standardized synthesis guide below which encapsulates the critical process steps for successful replication.
- Perform acylation of D,L-methionine sodium with acetic anhydride at 25-45°C with a molar ratio of 1: 1.0-1.5 to form the sodium salt intermediate.
- Adjust pH to 2-4 using inorganic acid, followed by vacuum evaporation at ≤65°C to reduce water content to ≤5%.
- Treat the concentrated mixture with organic solvents (e.g., ethanol, ethyl acetate) to precipitate inorganic salts, filter, decolorize, and evaporate solvent to obtain crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented manufacturing process offers profound advantages that extend far beyond simple yield improvements. By fundamentally altering the separation physics from aqueous crystallization to organic solvent extraction, the process eliminates the generation of hazardous acidic mother liquors, thereby drastically reducing waste treatment costs and environmental compliance liabilities. This shift translates directly into a more stable and predictable cost structure, as the volatility associated with waste disposal fees and regulatory penalties is effectively mitigated. Moreover, the ability to recycle the organic solvents used in the precipitation step creates a closed-loop system that minimizes raw material consumption, further enhancing the economic viability of large-scale production runs. For supply chain managers, this means a more resilient sourcing channel that is less susceptible to disruptions caused by environmental shutdowns or raw material scarcity.
- Cost Reduction in Manufacturing: The elimination of expensive ion-exchange resin columns and the reduction in raw material loss due to high crystallization yields significantly lower the cost of goods sold. By recovering nearly all the product from the reaction mixture through organic extraction rather than losing it in acidic waste streams, manufacturers can achieve substantial cost savings without compromising on quality. The process also reduces energy consumption by utilizing moderate temperatures for vacuum evaporation compared to the high-energy demands of distilling large volumes of water in traditional methods.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route against variations in raw material purity, such as the ability to utilize D,L-methionine with content as low as 94-98%, ensures a steady supply of intermediates even when feedstock quality fluctuates. This flexibility allows suppliers to maintain continuous production schedules without frequent stoppages for raw material qualification or process re-optimization. Consequently, buyers can rely on shorter lead times and more consistent delivery schedules, which is critical for maintaining just-in-time inventory strategies in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop experiments to multi-ton commercial production without the need for complex equipment modifications. The absence of salty wastewater discharge simplifies the environmental permitting process and reduces the footprint of the production facility's effluent treatment plant. This alignment with green chemistry principles not only future-proofs the supply chain against tightening environmental regulations but also enhances the corporate social responsibility profile of the end-product for global pharmaceutical clients.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this intermediate into their supply chains, we have compiled a set of frequently asked questions based on the specific technical disclosures within the patent documentation. These inquiries address common concerns regarding process scalability, impurity profiles, and raw material flexibility, providing clarity on how this technology compares to incumbent methods. Understanding these details is crucial for R&D teams assessing route scouting options and procurement officers negotiating supply agreements. The answers provided below are derived directly from the experimental data and process descriptions found in the source intellectual property, ensuring accuracy and relevance to real-world manufacturing scenarios.
Q: How does this process improve environmental compliance compared to traditional methods?
A: Unlike traditional methods that generate large volumes of acidic mother liquor requiring disposal, this patented process utilizes organic solvent extraction to separate inorganic salts, allowing for solvent recycling and eliminating salty wastewater discharge.
Q: What purity levels can be achieved with this acylation protocol?
A: The process consistently achieves product purity levels as high as 99%, with crystallization yields reaching 97-99%, significantly reducing raw material waste compared to conventional cooling crystallization techniques.
Q: Can low-grade D,L-methionine be used as a starting material?
A: Yes, the process is robust enough to handle D,L-methionine with purity as low as 94-98% or sodium methionate derived from hydantoin hydrolysis, offering flexibility in raw material sourcing and cost management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acetyl-D,L-Methionine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high-purity standards demonstrated in laboratory settings are faithfully reproduced at an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry benchmarks. Our commitment to quality assurance means that every kilogram of N-acetyl-D,L-methionine we supply is backed by comprehensive data packages that validate its suitability for downstream enzymatic resolution and API synthesis.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our implementation of this solvent-based precipitation technology can reduce your overall production costs. We encourage potential partners to contact us directly to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence rather than theoretical projections. Let us collaborate to build a more efficient, sustainable, and cost-effective supply chain for your critical amino acid intermediates.
