Scalable Manufacturing of Prasugrel Intermediates via Novel Non-Grignard Acylation Technology
Introduction to Advanced Antithrombotic Intermediate Synthesis
The pharmaceutical landscape for antithrombotic agents continues to evolve, driven by the demand for safer and more efficient manufacturing processes for key active ingredients. Central to the production of Prasugrel, a potent P2Y12 inhibitor used in the treatment of acute coronary syndromes, is the critical building block known as 1-cyclopropyl-2-(2-fluorophenyl) ethyl ketone. Recent advancements detailed in patent CN102140059B introduce a transformative synthetic methodology that departs from hazardous conventional practices. This innovation leverages N,N'-carbonyldiimidazole (CDI) mediated activation to achieve high-yield condensation under remarkably mild conditions. By circumventing the need for sensitive organometallic reagents, this technology offers a robust pathway for the reliable pharmaceutical intermediate supplier community to enhance production safety and consistency.
The strategic importance of this intermediate cannot be overstated, as it forms the core scaffold of the final therapeutic molecule. The structural integrity and purity of this ketone directly influence the downstream processing and final quality of the antithrombotic API. As depicted in the structural analysis below, the connection between the cyclopropyl carbonyl group and the fluorobenzyl moiety is the defining feature that necessitates precise chemical construction.
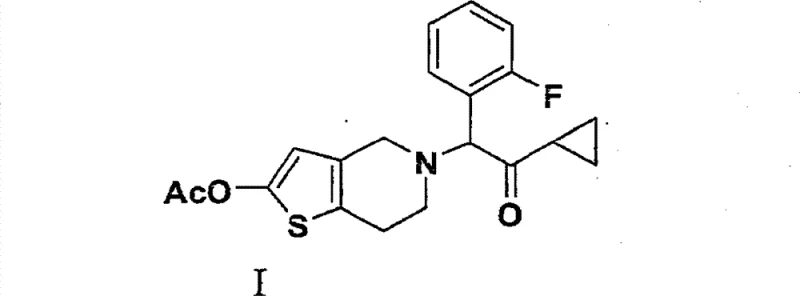
Implementing this novel route allows manufacturers to address long-standing bottlenecks in the supply chain of cardiovascular medications. The shift towards greener chemistry principles is not merely regulatory compliance but a competitive advantage that ensures long-term viability. For R&D directors and procurement specialists, understanding the mechanistic nuances of this CDI-based approach is essential for evaluating potential technology transfers and scaling strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-cyclopropyl-2-(2-fluorophenyl) ethyl ketone has relied heavily on Grignard chemistry, a technique fraught with operational complexities and safety hazards. Traditional protocols, such as those cited in prior art WO2009062044 and CN101177430A, typically involve the generation of organomagnesium species from o-fluorobenzyl bromide or chloride. These reactions mandate the use of strictly anhydrous diethyl ether or tetrahydrofuran (THF) as solvents, requiring rigorous exclusion of moisture and oxygen to prevent reagent decomposition.
Furthermore, the starting materials employed in these legacy routes, specifically o-fluorobenzyl halides, possess strong lachrymatory properties, posing severe health risks to operators and necessitating specialized containment equipment. The exothermic nature of Grignard formation combined with the flammability of ether solvents creates a high-risk environment unsuitable for modern, large-scale commercial production. Additionally, the disposal of magnesium salts and the recovery of volatile organic solvents contribute to a heavier environmental footprint, complicating waste management protocols for manufacturing facilities.
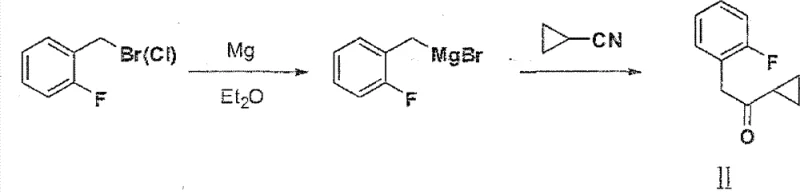
The Novel Approach
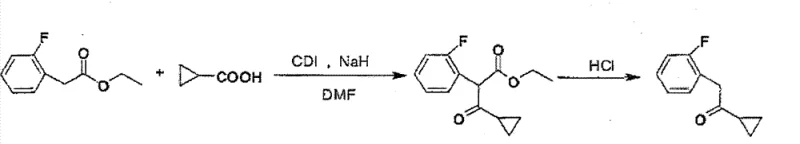
In stark contrast, the methodology disclosed in CN102140059B revolutionizes the synthesis by employing a condensation strategy between ethyl 2-(2-fluorophenyl)acetate and cyclopropanecarboxylic acid. This approach eliminates the need for pyrophoric reagents and inert atmospheres, allowing reactions to proceed in polar aprotic solvents like DMF under ambient or mildly cooled conditions. The use of CDI as a coupling agent activates the carboxylic acid efficiently, facilitating nucleophilic attack by the ester enolate generated in situ.
This paradigm shift significantly simplifies the operational workflow, removing the necessity for complex drying trains and explosion-proof infrastructure associated with ether handling. The reaction profile is much gentler, reducing the risk of thermal runaways and improving overall process safety. Moreover, the avoidance of halogenated benzyl starting materials mitigates operator exposure to toxic vapors, aligning the process with modern occupational health and safety standards. The resulting workflow is not only chemically elegant but also economically superior due to reduced utility consumption and waste treatment costs.
Mechanistic Insights into CDI-Mediated Acylation and Decarboxylation
The core of this innovative synthesis lies in the activation of cyclopropanecarboxylic acid by N,N'-carbonyldiimidazole (CDI). Upon mixing in DMF, CDI reacts with the carboxylic acid to form a reactive acyl imidazole intermediate, releasing imidazole as a by-product. This activated species is highly electrophilic, primed for nucleophilic attack. Simultaneously, the addition of a strong base such as sodium hydride (NaH) or potassium tert-butoxide (t-BuOK) deprotonates the alpha-carbon of ethyl 2-(2-fluorophenyl)acetate, generating a stable enolate ion.
The nucleophilic enolate then attacks the carbonyl carbon of the acyl imidazole, forming a beta-keto ester intermediate. This step is crucial as it constructs the carbon-carbon bond between the cyclopropyl and fluorobenzyl fragments without the need for metal catalysis. The reaction is typically conducted at low temperatures (below 5°C) during base addition to control exotherms and minimize side reactions, followed by stirring at room temperature to ensure complete conversion. The mildness of CDI prevents the degradation of sensitive functional groups, ensuring high fidelity in the molecular architecture.
Following the condensation, the process involves a hydrolysis and decarboxylation sequence. The crude beta-keto ester is subjected to acidic conditions using concentrated hydrochloric acid in an ethanol-water mixture under reflux. This step cleaves the ester moiety and facilitates the loss of carbon dioxide, yielding the target ketone, 1-cyclopropyl-2-(2-fluorophenyl) ethyl ketone. The acidic workup also serves to quench any remaining basic reagents and solubilize inorganic salts, which are subsequently removed through aqueous washing. This two-stage mechanism ensures high purity by effectively separating the desired ketone from unreacted starting materials and urea-like by-products derived from CDI.
How to Synthesize 1-cyclopropyl-2-(2-fluorophenyl) ethyl ketone Efficiently
Executing this synthesis requires careful attention to reagent stoichiometry and temperature control during the activation phase. The protocol begins with the dissolution of the acid and ester components in dry DMF, followed by the batch-wise addition of CDI to manage gas evolution. Once the activated intermediate is formed, the system is cooled, and the base is introduced slowly to generate the enolate. The subsequent reaction period allows for the coupling to reach completion, monitored typically by HPLC or TLC. Following the coupling, the acidic hydrolysis step is critical for converting the intermediate into the final ketone structure.
- Activate cyclopropanecarboxylic acid using CDI in DMF solvent at room temperature.
- Generate the enolate of ethyl 2-(2-fluorophenyl)acetate using NaH or t-BuOK at low temperature (below 5°C) and react with the activated acid.
- Perform acidic hydrolysis and decarboxylation using HCl in ethanol/water under reflux to yield the final ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this non-Grignard methodology represents a significant opportunity for cost reduction in pharmaceutical intermediates manufacturing. By eliminating the requirement for expensive, anhydrous-grade solvents and specialized reactor linings capable of handling corrosive halides, the capital expenditure (CAPEX) and operational expenditure (OPEX) are substantially optimized. The simplified workup procedure, involving standard aqueous extractions and distillation, reduces cycle times and increases throughput capacity without the need for complex purification columns.
- Cost Reduction in Manufacturing: The removal of Grignard reagents eliminates the cost associated with magnesium turnings and the rigorous drying of solvents, which are energy-intensive processes. Furthermore, the use of commodity chemicals like cyclopropanecarboxylic acid and ethyl esters, which are readily available in bulk, stabilizes raw material pricing and reduces dependency on niche suppliers. The higher atom economy and reduced waste generation also lower the costs associated with environmental compliance and hazardous waste disposal.
- Enhanced Supply Chain Reliability: Traditional routes relying on o-fluorobenzyl bromide face supply volatility due to the hazardous nature of the material and strict transportation regulations. The novel route utilizes stable, non-hazardous carboxylic acids and esters that are easier to store and transport, ensuring a more resilient supply chain. This stability minimizes the risk of production stoppages caused by raw material shortages or regulatory delays in shipping dangerous goods.
- Scalability and Environmental Compliance: The mild reaction conditions allow for seamless scale-up from pilot plants to multi-ton commercial reactors without the engineering challenges posed by exothermic Grignard formations. The process generates less hazardous waste, primarily consisting of imidazole and salt solutions, which are easier to treat than heavy metal residues or halogenated organic waste. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the sustainability profile of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this critical antithrombotic intermediate. These insights are derived directly from the patented methodology and practical manufacturing considerations, providing clarity for stakeholders evaluating this technology for their supply chains.
Q: Why is the CDI-mediated route superior to traditional Grignard methods for this ketone?
A: Traditional Grignard methods require strictly anhydrous and oxygen-free conditions with flammable solvents like diethyl ether, posing significant safety risks. The novel CDI route operates under mild conditions without the need for inert atmospheres, significantly reducing operational hazards and infrastructure costs.
Q: What are the critical quality attributes for this prasugrel intermediate?
A: High purity is essential to ensure the efficacy of the final antithrombotic API. The process utilizes robust purification steps including aqueous washes and distillation to remove residual solvents and by-products, ensuring the material meets stringent pharmaceutical specifications.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the elimination of hazardous lachrymators (like o-fluorobenzyl bromide) and the use of common solvents like DMF and ethanol make this process highly scalable. The mild reaction conditions allow for easier temperature control and safer handling in multi-ton reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-cyclopropyl-2-(2-fluorophenyl) ethyl ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality intermediates play in the global fight against cardiovascular disease. Our technical team has extensively analyzed the CDI-mediated pathway described in CN102140059B and possesses the expertise to implement this superior route at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our state-of-the-art rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, guaranteeing performance in downstream API synthesis.
We invite pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology for their Prasugrel programs. By choosing our services, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring your project moves forward with the most efficient and reliable supply solution available.
