Advanced Catalytic Synthesis of Pyrimidine Thioketones for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. Patent CN1803777A introduces a transformative approach to the synthesis of pyrimidine thioketone compounds, a critical class of intermediates used in the development of advanced therapeutic agents and specialty chemicals. This innovation specifically addresses the long-standing challenges associated with traditional cyclization methods by replacing corrosive and hazardous mineral acids with a novel, recyclable Lewis acid catalyst system. By utilizing Magnesium Trifluoromethanesulfonate [Mg(OTf)2], the process achieves exceptional reaction yields exceeding 85% and purity levels greater than 98.5% under significantly milder conditions. This technical breakthrough not only enhances the chemical efficiency of the synthesis but also aligns with modern green chemistry principles by drastically reducing the generation of hazardous waste streams. For R&D directors and procurement leaders, this patent represents a viable route to secure high-quality intermediates while optimizing the overall cost structure of the manufacturing supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrimidine thioketones has relied heavily on the use of strong mineral acids such as concentrated hydrochloric acid or traditional Lewis acids like zinc chloride and boron trifluoride etherate. These conventional catalytic systems present severe operational drawbacks that hinder efficient commercial production. The use of concentrated hydrochloric acid, for instance, necessitates high reaction temperatures and extended reaction times, often requiring up to 18 hours to reach completion, which severely limits throughput capacity. Furthermore, the highly corrosive nature of these acids causes significant degradation of reactor equipment, leading to frequent maintenance downtime and increased capital expenditure for corrosion-resistant materials. Traditional Lewis acids are also notoriously sensitive to moisture, requiring strict anhydrous conditions that complicate the handling of raw materials and increase energy consumption for drying processes. Additionally, these catalysts often suffer from deactivation in the presence of nitrogen-containing compounds, requiring excessive catalyst loading to drive the reaction, which subsequently complicates downstream purification and waste disposal.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes Magnesium Trifluoromethanesulfonate as a robust and water-tolerant catalyst that overcomes the inherent limitations of prior art. This new catalytic system enables the condensation of aromatic aldehydes, beta-diketones, and substituted thioureas to proceed smoothly at temperatures ranging from 20°C to 150°C, offering remarkable flexibility in process optimization. The mild reaction conditions significantly reduce the thermal stress on the reaction mixture, thereby minimizing the formation of thermal degradation by-products and ensuring a cleaner crude product profile. Unlike traditional acids that are consumed or neutralized during the workup, this magnesium-based catalyst can be recovered from the filtrate with a recovery rate of approximately 95%, allowing for multiple reuse cycles without significant loss of activity. This shift from a consumable, corrosive reagent to a recyclable catalytic species fundamentally changes the economic model of the synthesis, transforming a waste-intensive process into a sustainable, closed-loop operation that is highly attractive for large-scale manufacturing.
Mechanistic Insights into Mg(OTf)2-Catalyzed Cyclization
The efficacy of Magnesium Trifluoromethanesulfonate in this transformation stems from its unique electronic properties as a hard Lewis acid that effectively activates carbonyl groups without promoting unwanted side reactions. The magnesium center coordinates with the oxygen atoms of the beta-diketone or beta-ketoester, increasing the electrophilicity of the carbonyl carbon and facilitating the nucleophilic attack by the thiourea derivative. This activation mode is superior to protonic acids because it avoids the excessive protonation of the thiourea nitrogen atoms, which can otherwise inhibit the nucleophilicity required for the cyclization step. The reaction proceeds through a concerted mechanism where the aldehyde, activated beta-dicarbonyl, and thiourea condense to form the dihydropyrimidine ring system with high regioselectivity. The stability of the triflate anion ensures that the catalyst remains active throughout the reaction cycle, even in the presence of trace moisture that would typically quench other Lewis acids. This mechanistic robustness allows for a broader substrate scope, accommodating various electronic substituents on the aromatic aldehyde without compromising yield or purity.
From an impurity control perspective, the mild nature of the Mg(OTf)2 catalyzed process is instrumental in achieving the reported purity levels of over 98.5%. Harsh acidic conditions often lead to polymerization of the aldehyde or hydrolysis of the ester groups, generating complex impurity profiles that are difficult to separate. By maintaining a neutral to mildly acidic environment, the new process suppresses these degradation pathways, resulting in a reaction mixture where the desired pyrimidine thioketone is the dominant species. The high selectivity reduces the burden on the purification stage, allowing for simple recrystallization from solvents like ethanol or ethyl acetate to achieve pharmaceutical-grade quality. Furthermore, the ability to recover the catalyst from the aqueous phase after filtration ensures that no heavy metal residues or corrosive salts remain in the final product, which is a critical requirement for regulatory compliance in API manufacturing. This level of control over the reaction environment provides R&D teams with the confidence to scale the process without fearing unexpected impurity spikes.
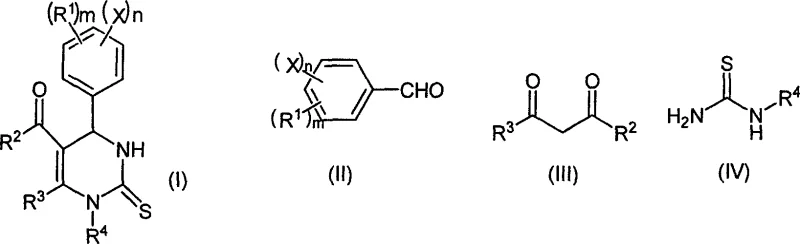
How to Synthesize Pyrimidine Thioketone Efficiently
The implementation of this synthesis route involves a straightforward operational protocol that is well-suited for standard chemical manufacturing facilities. The process begins with the precise charging of aromatic aldehyde, beta-diketone or beta-ketoester, and substituted thiourea into a reactor containing a suitable organic solvent such as acetonitrile, ethanol, or ethyl acetate. The Magnesium Trifluoromethanesulfonate catalyst is then added in a catalytic amount, typically ranging from 0.005 to 0.20 molar equivalents relative to the aldehyde, ensuring cost-effective usage. The mixture is heated to the desired temperature, often between 50°C and 80°C for optimal balance between reaction rate and energy consumption, and stirred until HPLC monitoring confirms the complete consumption of the starting materials. Upon completion, the product precipitates or is isolated via filtration, washed with ice water to remove soluble impurities, and recrystallized to obtain the final high-purity solid. The detailed standardized synthesis steps, including specific stoichiometric ratios and workup procedures, are outlined in the guide below.
- Combine aromatic aldehyde, beta-diketone or beta-ketoester, and substituted thiourea with Mg(OTf)2 catalyst in an organic solvent.
- Heat the reaction mixture to a temperature between 20°C and 150°C and maintain stirring for 0.5 to 24 hours until completion.
- Filter the reaction product, wash the filter cake with ice water, and recrystallize from ethanol or ethyl acetate to obtain high-purity solids.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Mg(OTf)2 catalyzed route offers substantial strategic advantages that extend beyond simple yield improvements. The primary value driver is the significant reduction in manufacturing costs achieved through the elimination of expensive corrosion-resistant equipment and the minimization of waste treatment expenses. Since the catalyst is recyclable and the reaction generates almost no three wastes, the operational expenditure associated with environmental compliance and hazardous waste disposal is drastically lowered. The mild reaction conditions also translate to enhanced safety profiles, reducing the risk of thermal runaways and allowing for the use of standard glass-lined or stainless steel reactors rather than specialized tantalum or hastelloy vessels. These factors combine to create a more resilient and cost-effective supply chain that is less vulnerable to regulatory changes regarding waste disposal and worker safety.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental shift from stoichiometric acid consumption to catalytic turnover. By recovering and reusing the Magnesium Trifluoromethanesulfonate catalyst, the raw material cost per kilogram of product is significantly reduced compared to processes that consume large quantities of mineral acids. Additionally, the high purity of the crude product minimizes the need for extensive chromatographic purification or multiple recrystallization steps, leading to lower solvent consumption and reduced processing time. The elimination of neutralization steps further reduces the consumption of bases and the generation of salt waste, contributing to a leaner and more efficient manufacturing cost structure that improves overall margin potential.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents and the capacity of waste treatment facilities. This synthesis method utilizes readily available raw materials such as common aromatic aldehydes and beta-ketoesters, which are widely sourced from the global chemical market, ensuring stable input availability. The robustness of the catalyst against moisture and air simplifies storage and handling requirements, reducing the risk of supply disruptions due to reagent degradation. Furthermore, the simplified workup procedure shortens the production cycle time, allowing manufacturers to respond more quickly to fluctuations in market demand and maintain healthier inventory levels without the need for excessive safety stock.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges related to heat transfer and mixing, but the mild exothermic nature of this reaction makes it inherently safer to scale up from laboratory to plant scale. The low waste generation profile aligns perfectly with increasingly stringent environmental regulations, future-proofing the manufacturing site against tighter emission standards. The ability to operate in a variety of common organic solvents provides flexibility in solvent recovery and recycling infrastructure, further enhancing the sustainability credentials of the production process. This environmental compatibility ensures long-term operational licenses and reduces the risk of production halts due to compliance violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrimidine thioketone synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing production lines or for qualifying new suppliers who utilize this advanced methodology. The insights provided here cover catalyst performance, environmental impact, and scalability considerations.
Q: What are the advantages of using Mg(OTf)2 over traditional Lewis acids?
A: Magnesium Trifluoromethanesulfonate offers superior stability against hydrolysis compared to traditional Lewis acids like ZnCl2 or BF3. It operates under milder conditions, reduces equipment corrosion, and allows for efficient catalyst recovery and reuse, significantly lowering long-term operational costs.
Q: How does this process impact environmental compliance?
A: The process generates almost no three wastes due to the high selectivity of the catalyst and the ability to recover the catalyst from the filtrate. This minimizes the need for extensive waste treatment and neutralization steps required by strong mineral acids.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the method is highly scalable. The mild reaction conditions (20-150°C) and the use of common organic solvents like ethanol or acetonitrile make it safe and feasible for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidine Thioketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in maintaining a competitive edge in the global fine chemical market. Our technical team has thoroughly analyzed the Mg(OTf)2 catalyzed pathway and confirmed its potential for delivering high-purity pyrimidine thioketones with exceptional consistency. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the low impurity profiles promised by this novel synthesis method, guaranteeing that every batch meets the exacting standards required by international pharmaceutical and agrochemical clients.
We invite you to collaborate with us to leverage this advanced technology for your specific supply chain needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient process for your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your project timelines. Let us help you optimize your supply chain with a solution that balances technical excellence with commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
