Advanced Synthetic Route for Baloxavir Key Intermediate: Enhancing Efficiency and Scalability
Advanced Synthetic Route for Baloxavir Key Intermediate: Enhancing Efficiency and Scalability
The global demand for effective antiviral therapeutics has surged, placing intense scrutiny on the supply chains of critical active pharmaceutical ingredients (APIs) and their precursors. Among these, the synthesis of Baloxavir Marboxil, a groundbreaking cap-dependent endonuclease inhibitor, represents a significant challenge due to its complex heterocyclic structure and stringent stereochemical requirements. A pivotal development in this domain is detailed in Chinese Patent CN109912624B, which discloses a novel, highly efficient synthetic method for the key parent nucleus intermediate of Baloxavir. This patent introduces a strategic pathway that bypasses traditional bottlenecks, utilizing a direct condensation and one-step cyclization strategy to construct the core triazine-pyridone scaffold. For R&D directors and procurement specialists, understanding this technological leap is crucial, as it promises not only higher purity profiles but also a fundamentally more robust and cost-effective manufacturing landscape for this vital anti-influenza medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
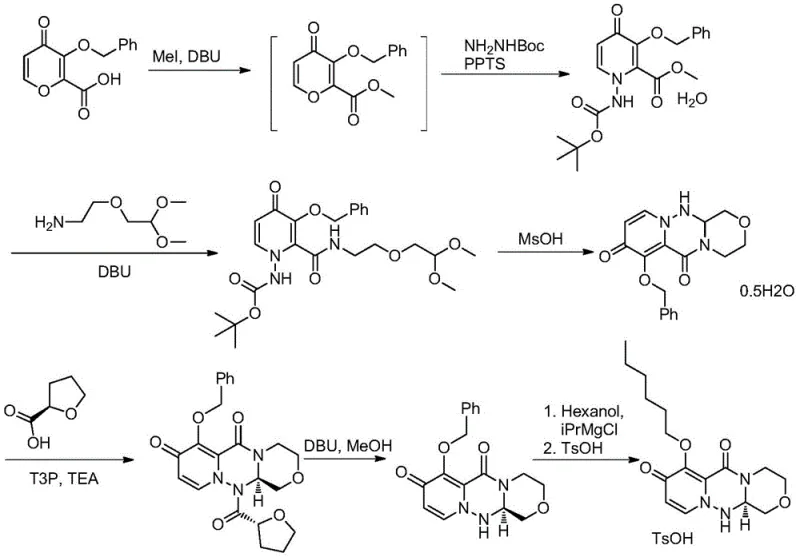
Prior art methodologies, such as those reported in Japanese Patent JP6212678, have historically plagued the production of Baloxavir intermediates with inefficiencies and safety hazards. The conventional routes typically rely on a multi-step sequence that necessitates the introduction and subsequent removal of benzyl protecting groups, adding unnecessary synthetic length and reducing overall atom economy. Furthermore, these legacy processes often employ virulent reagents like methyl iodide for esterification, posing severe occupational health risks and complicating waste disposal protocols. The requirement for excessive equivalents of expensive amines and the use of costly chiral resolving agents further inflate the material costs, rendering the process economically suboptimal for large-scale commercialization. These cumulative factors result in a low overall yield and a fragile supply chain vulnerable to regulatory and operational disruptions.
The Novel Approach
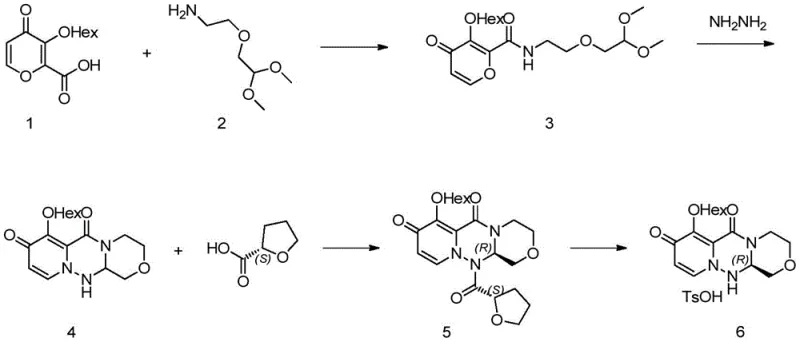
In stark contrast, the innovative route described in the patent data streamlines the synthesis by initiating with a 3-(n-hexyloxy)-4-oxo-4H-pyran-2-carboxylic acid derivative, effectively eliminating the need for late-stage alkyl group substitution. This strategic starting point allows for a direct condensation with a dimethoxyethoxy ethylamine, followed by a remarkable one-step cyclization with hydrazine hydrate to form the racemic core. The process then employs a crystallization-driven chiral resolution using the more affordable (S)-tetrahydrofuran-2-carboxylic acid, avoiding expensive chromatographic separations. Finally, a mild deprotection step yields the target salt. This consolidated approach drastically reduces the number of unit operations, minimizes solvent consumption, and enhances the safety profile by removing toxic alkylating agents, thereby establishing a superior framework for industrial manufacturing.
Mechanistic Insights into Hydrazine-Mediated Cyclization and Chiral Resolution
The cornerstone of this synthetic breakthrough lies in the efficient construction of the pyrido[2,1-f][1,2,4]triazine ring system via hydrazine-mediated cyclization. Mechanistically, the reaction involves the nucleophilic attack of hydrazine hydrate on the activated carbonyl species of the amide intermediate, facilitated by an acid catalyst such as p-toluenesulfonic acid. This triggers a cascade of intramolecular condensations that close the triazine ring with high fidelity. The choice of solvent, typically tetrahydrofuran or dichloromethane, and the precise control of temperature between 50-55°C are critical to driving the equilibrium towards the cyclic product while suppressing side reactions. This one-pot transformation is particularly elegant as it avoids the isolation of unstable intermediates, thereby preserving material throughput and ensuring a consistent impurity profile that is easier to manage downstream.
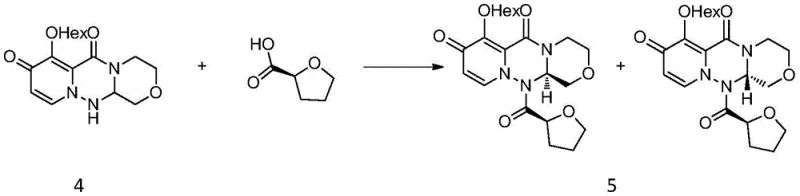
Following the cyclization, the control of stereochemistry is achieved through a diastereomeric salt formation or amide coupling strategy using (S)-tetrahydrofuran-2-carboxylic acid. This chiral auxiliary selectively interacts with one enantiomer of the racemic intermediate, altering its solubility properties sufficiently to allow for purification via recrystallization. The patent data highlights that this resolution step can be optimized using mixed solvent systems like acetone and petroleum ether, which promote the precipitation of the desired diastereomer while leaving the unwanted isomer in the mother liquor. Subsequent removal of the chiral auxiliary under basic conditions using DBU in methanol proceeds cleanly without racemization, ensuring that the final product retains the high optical purity required for biological activity. This mechanism underscores a sophisticated understanding of physical organic chemistry applied to process optimization.
How to Synthesize Baloxavir Core Intermediate Efficiently
The synthesis of this critical pharmaceutical intermediate requires precise adherence to reaction parameters to maximize yield and purity. The process begins with the activation of the carboxylic acid starting material using coupling agents like EDCI, followed by amidation. The subsequent cyclization must be carefully monitored to ensure complete conversion before proceeding to the resolution stage. The final deprotection and salt formation steps are equally vital, requiring controlled cooling to induce proper crystallization of the tosylate salt. For a comprehensive breakdown of the specific reagents, stoichiometry, and workup procedures validated in the patent examples, please refer to the standardized technical guide below.
- Condense 3-(n-hexyloxy)-4-oxo-4H-pyran-2-carboxylic acid with 2-(2,2-dimethoxyethoxy)ethylamine using EDCI to form the amide intermediate.
- Perform a one-step cyclization with hydrazine hydrate under acidic catalysis to construct the triazine ring system.
- Resolve the racemic mixture using (S)-tetrahydrofuran-2-carboxylic acid to isolate the desired chiral intermediate via crystallization.
- Deprotect the chiral auxiliary using DBU and methanol, followed by salt formation with p-toluenesulfonic acid to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic advantages that extend beyond simple unit cost metrics. By eliminating the use of hazardous methyl iodide and reducing the total number of synthetic steps, the process inherently lowers the regulatory burden associated with handling controlled substances and managing complex waste streams. The simplification of the workflow means fewer quality control checkpoints and reduced risk of batch failure, leading to a more predictable and reliable supply of the intermediate. Furthermore, the use of commodity chemicals and solvents ensures that the supply chain is not dependent on niche or single-source vendors, thereby mitigating the risk of raw material shortages that can plague complex pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this route is driven primarily by the drastic reduction in processing time and material consumption. By avoiding the installation and removal of benzyl protecting groups, the process saves significant amounts of reagents, solvents, and energy that would otherwise be consumed in additional reaction and purification cycles. The substitution of expensive chiral resolving agents with the more accessible (S)-tetrahydrofuran-2-carboxylic acid further drives down the bill of materials. Additionally, the high yields reported in the patent examples, often exceeding ninety percent in key steps, mean that less starting material is wasted, directly improving the cost of goods sold (COGS) and enhancing the overall profitability of the production campaign.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthesis offers superior supply security. The reliance on stable, commercially available starting materials like 3-(n-hexyloxy)-4-oxo-4H-pyran-2-carboxylic acid ensures that production can be ramped up quickly without long lead times for custom synthesis of precursors. The simplified process flow reduces the likelihood of bottlenecks that often occur in multi-step syntheses involving sensitive intermediates. This stability allows for better inventory planning and ensures that downstream API manufacturing schedules can be met consistently, which is critical for maintaining the continuity of supply for essential antiviral medications in the global market.
- Scalability and Environmental Compliance: The environmental footprint of this process is significantly lighter than that of conventional methods, aligning with modern green chemistry principles and increasingly strict environmental regulations. The avoidance of toxic alkylating agents reduces the need for specialized containment and scrubbing systems, lowering capital expenditure for facility upgrades. The high atom economy and reduced solvent usage per kilogram of product make the process easier to scale from pilot plant to commercial tonnage without encountering heat transfer or mixing limitations. This scalability ensures that manufacturers can meet surging demand during flu seasons without compromising on environmental compliance or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Baloxavir intermediates using this patented methodology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on the feasibility and advantages of this specific synthetic route for industry stakeholders.
Q: How does this new synthetic route improve upon previous methods for Baloxavir intermediates?
A: The new route eliminates the need for toxic methyl iodide and avoids complex benzyl protection/deprotection sequences found in prior art, significantly shortening the synthetic timeline and reducing hazardous waste.
Q: What is the role of (S)-tetrahydrofuran-2-carboxylic acid in this process?
A: It serves as a cost-effective chiral resolving agent that allows for the separation of enantiomers through crystallization, ensuring high optical purity without requiring expensive chromatographic techniques.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like dichloromethane and tetrahydrofuran, operates at mild temperatures, and achieves high yields (over 90% in key steps), making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baloxavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key pharmaceutical intermediates like the Baloxavir core structure. Our team of expert chemists has extensively analyzed emerging synthetic technologies, including the route described in CN109912624B, to ensure our manufacturing capabilities are aligned with the most efficient and safe industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver products with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify identity and purity, ensuring that every batch meets the exacting requirements of global regulatory bodies.
We invite pharmaceutical partners to collaborate with us to optimize their supply chains for antiviral drug production. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation and quality can support your long-term strategic goals in the competitive pharmaceutical marketplace.
