Advanced Three-Step Synthesis of Benzo[c]quinolizin-3-ones for Pharmaceutical Applications
Advanced Three-Step Synthesis of Benzo[c]quinolizin-3-ones for Pharmaceutical Applications
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex heterocyclic scaffolds, particularly those serving as potent enzyme inhibitors. Patent CN1128148C introduces a groundbreaking process for the preparation of (1H)-benzo[c]quinolizin-3-ones derivatives, a class of compounds renowned for their efficacy as 5α-reductase inhibitors. These molecules are critical in treating pathologies such as benign prostatic hyperplasia, male pattern baldness, and prostate cancer. The disclosed innovation represents a significant leap forward by condensing the synthesis into merely three robust steps, utilizing starting materials that are either commercially available or easily preparable. This reduction in synthetic complexity addresses long-standing challenges in process chemistry, offering a streamlined alternative to the cumbersome multi-step protocols previously described in international applications like PCT/EP97/00552. By focusing on operational simplicity and high atom economy, this methodology provides a compelling value proposition for manufacturers aiming to optimize their production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of general formula (I) compounds was fraught with inefficiencies and operational hazards. As detailed in the background art, traditional routes necessitated a convoluted sequence beginning with the protection of the amide group in compound II as an N-Boc derivative. This was followed by a reduction step and a subsequent reaction with a silyloxydiene prepared in situ, which inherently introduces variability and moisture sensitivity issues. The pathway further required hydrolysis to yield intermediate compounds of general formula III, as depicted in the structural representation below. Finally, the introduction of the crucial double bond at the b-position demanded harsh oxidative conditions, typically employing dichlorodicyanoquinone (DDQ) or mercuric acetate. These reagents are not only expensive but also pose significant environmental and safety disposal challenges, creating bottlenecks in waste management and increasing the overall cost of goods sold for the final active pharmaceutical ingredient.
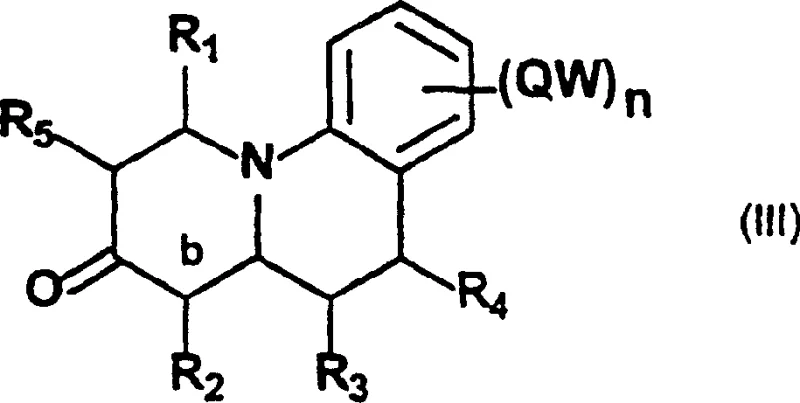
The Novel Approach
In stark contrast to the legacy methodologies, the present invention offers a direct and elegant solution that bypasses the need for protecting groups and toxic oxidants. The core of this novel strategy involves the N-alkylation of a thioamide derivative using a vinyl ketone, followed by a methylation and cyclization sequence. This approach leverages the nucleophilicity of the thioamide nitrogen to attack the vinyl ketone under mild basic conditions, forming the carbon-nitrogen bond essential for the ring system. The entire transformation is achieved in just three steps, drastically reducing the time and resources required for production. By eliminating the need for silylenol ethers and heavy metal oxidants, the process aligns perfectly with modern green chemistry principles. The reaction scheme below illustrates the seamless transition from simple starting materials (Formula 2 and 3) to the complex target molecule (Formula I), highlighting the efficiency that makes this a preferred route for reliable pharmaceutical intermediate suppliers seeking to enhance their manufacturing capabilities.
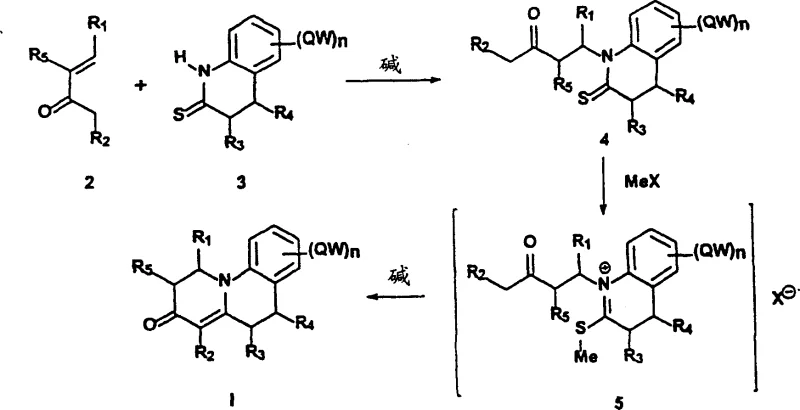
Mechanistic Insights into Thioamide N-Alkylation and Cyclization
The success of this synthetic route hinges on the precise control of reaction conditions during the initial N-alkylation step. The patent explicitly notes that the nature of the base is paramount; strong nucleophilic bases must be avoided as they induce the polymerization of the vinyl ketone starting material, leading to tarry byproducts and reduced yields. Conversely, weak bases are insufficient to deprotonate the thioamide effectively. The optimal solution involves using strong but non-nucleophilic bases such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) or potassium carbonate complexed with 18-crown-6. These bases facilitate the formation of the reactive thioamide anion without attacking the electrophilic vinyl group. The reaction is typically conducted in polar aprotic solvents like tetrahydrofuran (THF) at temperatures ranging from 0°C to 30°C. Maintaining this temperature window is critical to suppress side reactions while ensuring complete conversion to the N-alkylated thioamide intermediate (Compound 4).
Following the alkylation, the mechanism proceeds through a sulfur methylation step using dimethyl sulfate, which activates the thioamide sulfur for the subsequent intramolecular cyclization. This generates a reactive sulfonium salt intermediate (Compound 5), which is generally not isolated but treated directly with a base. The final cyclization is driven by the deprotonation of the alpha-carbon adjacent to the carbonyl group, triggering a nucleophilic attack on the activated iminium carbon. This cascade effectively closes the ring system to form the benzo[c]quinolizin-3-one core. From an impurity control perspective, the avoidance of mercury and DDQ eliminates heavy metal residues and quinone-related impurities, simplifying the purification process. The use of chromatography or crystallization becomes more straightforward, ensuring that the final product meets the stringent purity specifications required for clinical applications.
How to Synthesize Benzo[c]quinolizin-3-ones Efficiently
The practical implementation of this synthesis requires careful attention to reagent addition and temperature control to maximize yield and safety. The process begins with the suspension of the thioamide and base in anhydrous THF under an inert atmosphere, followed by the intermittent addition of the vinyl ketone to manage exothermicity. Once the N-alkylated intermediate is formed, the solvent may be swapped or the reaction mixture directly treated with methylating agents in toluene under reflux. The final cyclization step is rapid and efficient when performed with DBU. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below which outlines the exact experimental conditions validated in the patent examples.
- Perform N-alkylation of a thioamide derivative with a vinyl ketone in an organic solvent like THF, using a strong non-nucleophilic base such as DBU or K2CO3 with 18-crown-6 at 0-30°C.
- Methylate the resulting N-alkylated thioamide at the sulfur atom using dimethyl sulfate in a solvent like toluene under reflux conditions to generate an intermediate salt.
- Treat the intermediate salt directly with a strong non-nucleophilic base to induce cyclization, yielding the final benzo[c]quinolizin-3-one derivative without isolating the salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this three-step synthesis offers substantial strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike the conventional route which relies on specialized silylating agents and protected amino acid derivatives, this method utilizes commodity chemicals like ethyl vinyl ketone and substituted dihydroquinoline-thiones. These starting materials are widely available from multiple global vendors, mitigating the risk of supply disruptions and allowing for competitive sourcing strategies. Furthermore, the elimination of multiple isolation and purification steps between intermediates reduces the consumption of solvents and silica gel, directly lowering the variable costs associated with manufacturing. This leaner process flow translates into a more resilient supply chain capable of responding quickly to market demand fluctuations for 5α-reductase inhibitor intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive and hazardous reagents such as mercuric acetate and DDQ, which carry high procurement costs and significant waste disposal fees. By replacing these with inexpensive bases like potassium carbonate and dimethyl sulfate, the direct material costs are significantly reduced. Additionally, the reduction in unit operations—from multi-step protection/deprotection sequences to a direct three-step flow—lowers labor costs and equipment occupancy time. The avoidance of heavy metals also removes the need for costly scavenging resins or specialized wastewater treatment processes, contributing to a lower overall cost of production without compromising quality.
- Enhanced Supply Chain Reliability: Reliability in the pharmaceutical supply chain is often compromised by the dependency on custom-synthesized building blocks. This new methodology relies on robust, commercially available starting materials that have established supply chains. The simplified reaction sequence reduces the cumulative yield loss typical of long linear syntheses, ensuring higher throughput per batch. This efficiency means that manufacturers can maintain lower safety stock levels while still meeting delivery commitments. The robustness of the chemistry, particularly the tolerance for various substituents on the aromatic ring, allows for a flexible production schedule where different analogues can be produced on the same equipment line with minimal changeover time.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this route is inherently scalable due to its reliance on standard reaction types like alkylation and methylation. The absence of cryogenic conditions or high-pressure hydrogenation simplifies the engineering requirements for plant scale-up. From an environmental standpoint, the process aligns with increasingly strict regulatory frameworks by eliminating mercury waste and reducing the E-factor (mass of waste per mass of product). The use of common solvents like THF and toluene facilitates recycling and recovery, further enhancing the sustainability profile of the manufacturing operation and ensuring compliance with global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction scope and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this process into existing manufacturing workflows.
Q: How does this new synthesis route improve upon conventional methods for benzo[c]quinolizin-3-ones?
A: Conventional methods require multiple steps including N-Boc protection, reduction, reaction with silyloxydienes, and harsh oxidation using mercury or DDQ. The new patent CN1128148C reduces this to only three steps using commercially available starting materials, significantly simplifying the workflow and reducing waste.
Q: What are the critical reaction conditions for the N-alkylation step?
A: The choice of base is crucial. Strong nucleophilic bases cause polymerization of the vinyl ketone, while weak bases fail to drive the reaction. The patent specifies using strong but non-nucleophilic bases like DBU or K2CO3 with 18-crown-6 in THF at controlled temperatures (0-30°C) to ensure high efficiency.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. It utilizes readily available raw materials and avoids expensive or hazardous reagents like mercuric acetate. The reduction in step count from multi-step legacy routes to a concise three-step sequence enhances throughput and supply chain reliability for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo[c]quinolizin-3-ones Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the competitive landscape of pharmaceutical intermediates. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN1128148C and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this three-step process are realized at an industrial level. Our state-of-the-art facilities are designed to handle the specific solvent systems and reagents required for this synthesis, while our rigorous QC labs guarantee that every batch meets stringent purity specifications necessary for downstream API synthesis.
We invite potential partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this optimized synthetic route, we can help you achieve significant reductions in lead time and production costs. Please contact our technical procurement team today to request specific COA data for our benzo[c]quinolizin-3-ones derivatives and to discuss route feasibility assessments for your project pipeline. Let us be your trusted partner in delivering high-quality chemical solutions efficiently.
